DOI: https://doi.org/10.1038/s41467-023-44478-3
PMID: https://pubmed.ncbi.nlm.nih.gov/38167880
تاريخ النشر: 2024-01-02
طلاءات طويلة الأمد متوسطة بواسطة شبكة الهيبارين على القثاطير داخل الأوعية الدموية لمكافحة التخثر التكيفية والعدوى البكتيرية
تم القبول: 14 ديسمبر 2023
(أ) التحقق من التحديثات
الملخص
تُعتبر العدوى المرتبطة بالبكتيريا والتخثر، وخاصةً العدوى المرتبطة بالقسطرة والتخثر المرتبط بالقسطرة، من المضاعفات التي تهدد الحياة. هنا، نستخدم تجميعًا مختصرًا من هيبارين الصوديوم مع السطح النشط الأمونيومي الرباعي العضوي لصنع مركب طلاء متعدد الوظائف. على عكس الطلاءات التقليدية ذات الاستخدام الواحد، يرتبط المركب بالأجهزة الطبية ذات الأشكال والتركيبات العشوائية من خلال عملية غمس سهلة، ويشكل بعد ذلك طلاءات قوية لعلاج العدوى المرتبطة بالقسطرة والتخثر في الوقت نفسه. من خلال قوتها وفصلها التكيفي، لا تُظهر الطلاءات فقط استقرارًا جيدًا تحت الظروف القاسية، بل تقلل أيضًا بشكل كبير من التصاق الجلطات.
جلطة الأوردة العميقة مع معدل وفيات مرتفع
النتائج
تشكيل طلاء HS/DAC المستقر
سلسلة الألكيل
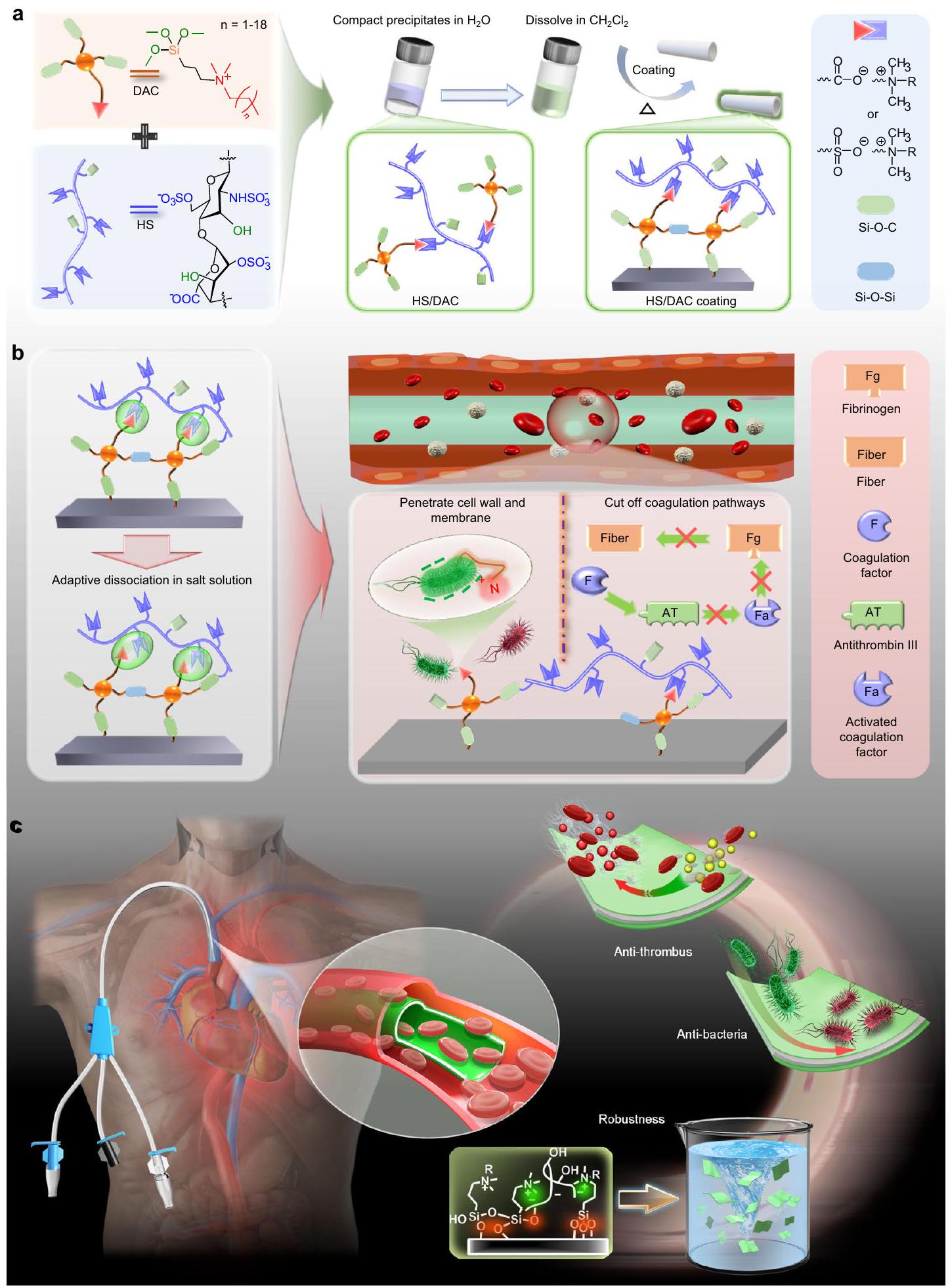
رسم توضيحي للإجراء المستخدم لتحضير مركب HS/DAC و HS/DAC
طلاء.
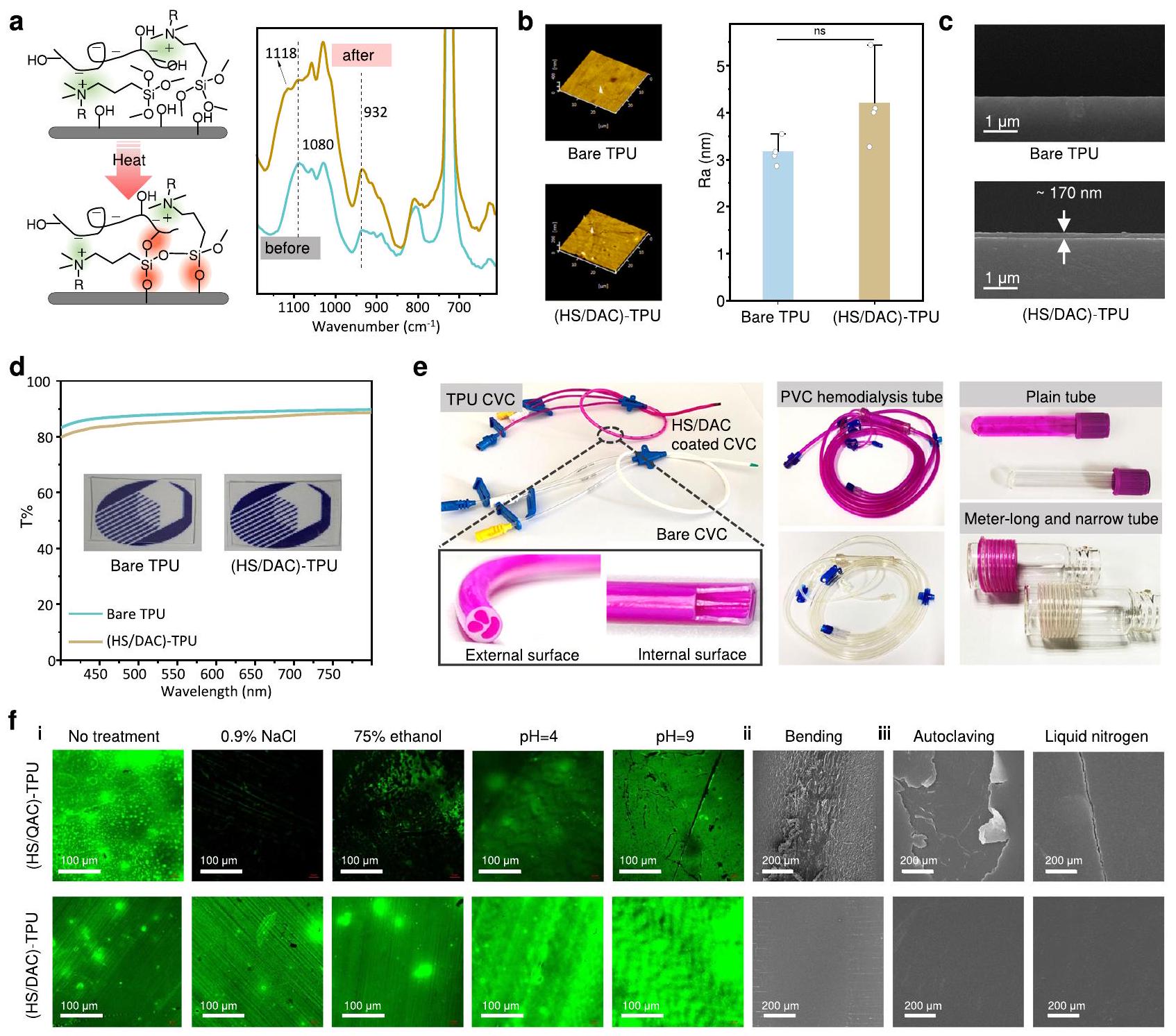
كان الطلاء سليمًا (الشكل 2f iii). قمنا بمزيد من الاستكشاف لاستقرار طلاء HS/DAC على المدى الطويل في التدفق
الخاصية المضادة للتخثر لطلاء HS/DAC
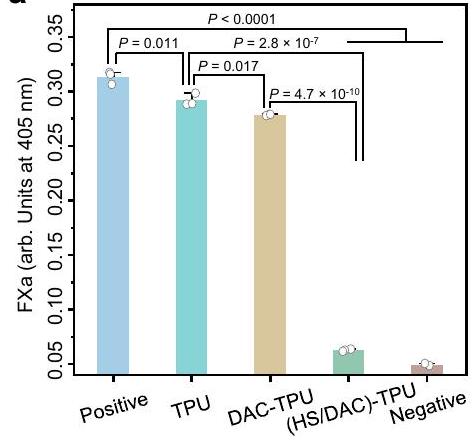
مع عينات لمدة ساعتين لدراسة التصاق Fg. أشارت صور المجهر الضوئي الماسح بالليزر (CLSM) إلى أن Fg كان أقل التصاقًا في القسطرة المطلية بـ HS/DAC مقارنة بالقسطرة العارية (الشكل 3d). على العكس من ذلك، لوحظت اختلافات ضئيلة في كمية ألبومين مصل البقر (BSA) من خلال اختبار حمض البيكينشونيك (BCA) عندما تم نقع العينات في محلول PBS من BSA (الشكل 3c). يُعتقد أن هذه الظاهرة المختلفة مرتبطة بتنشيط Fg. تؤدي التغيرات الشكلية في بنية Fg أثناء تنشيط Fg لتشكيل شبكة من الفيبرين إلى تعرض مواقع ربط متعددة. يمكن أن ترتبط هذه المواقع بمختلف البروتينات، مما يتسبب في امتصاص البروتين. وبالتالي، تم التحقيق في تأثير طلاء HS/DAC على تنشيط Fg بعد ساعتين من الدوران في الدم الكامل من خلال تقييم تعرض الـ







تشارك في تشكيل الجلطة
تأثير التحفيز بالملح، قمنا بدراسة الخصائص الفيزيائية والكيميائية لطلاء (HS/DAC) قبل وبعد المعالجة في محلول الملح. تم تمييز التكيف الذاتي لـ (HS/DAC)-TPU من خلال زوايا تماس الماء الثابتة، FTIR وXPS. بعد الحضانة مع محلول PBS، انخفضت زاوية التماس لـ (HS/DAC)-TPU بشكل ملحوظ، وانتقل نطاق مجموعة الكربوكسيل من 1609 إلى
خصائص مضادة للبكتيريا والتوافق الحيوي لطلاء HS/DAC في المختبر وفي الجسم الحي
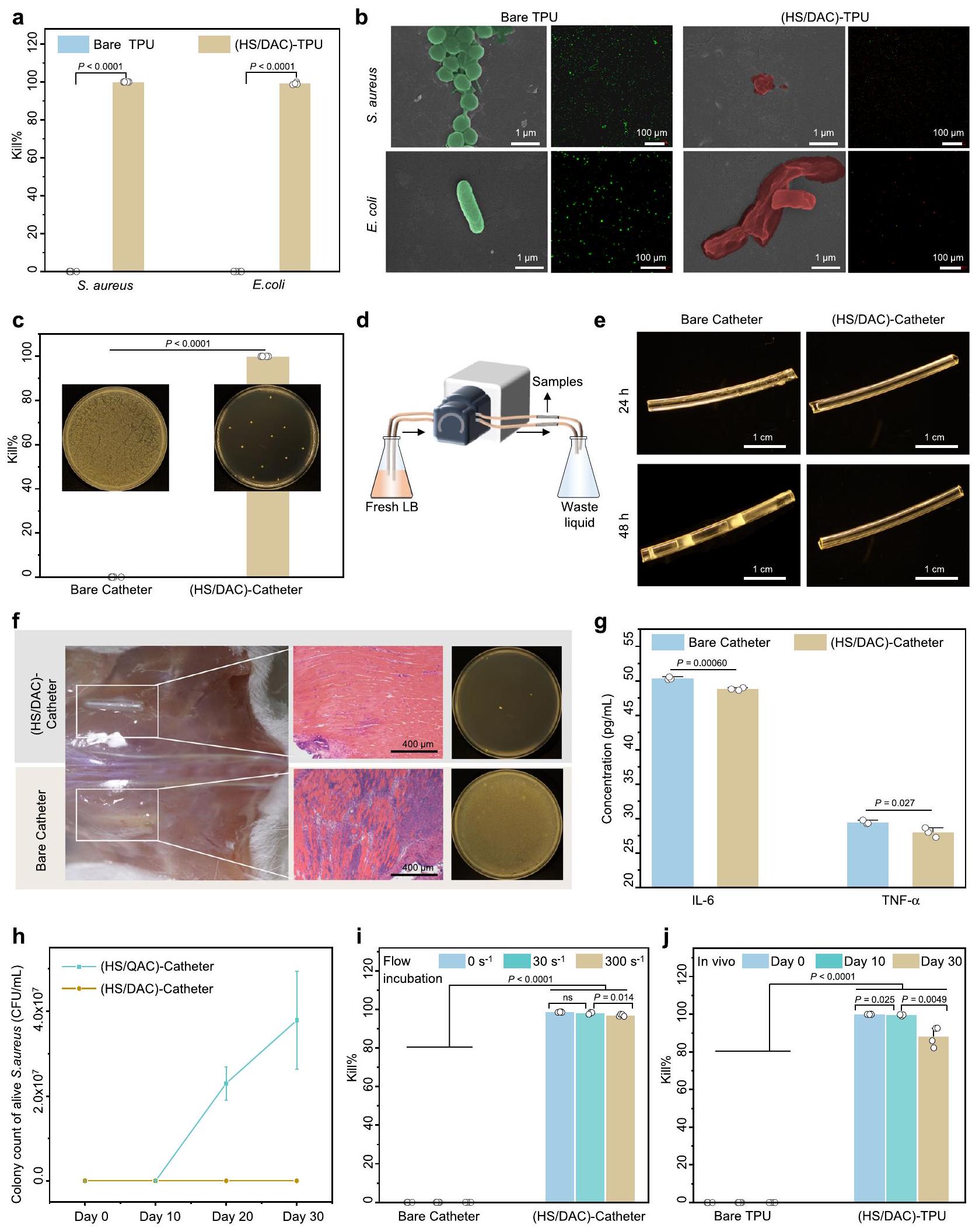
تؤكد بشكل أكبر تنظيم الالتهاب الذي تسببه العدوى البكتيرية. كما هو موضح في الشكل 4g، كانت العوامل المكتشفة منخفضة بشكل ملحوظ في القسطرة المغلفة بـ HS/DAC، وهو ما يتوافق مع نتائج الفحص النسيجي.
عدوى مرتبطة بالقسطرة في المختبر وفي الجسم الحي. مقارنة بقسطرة (HS/QAC)، حافظت قسطرة (HS/DAC) على نشاطها المضاد للبكتيريا الأصلي حتى بعد شهر واحد من العلاج في
تمت تكرار القياسات ثلاث مرات بشكل مستقل مع نتائج مشابهة.
مرتفعًا حتى 97% حتى عند التدفق في دم صناعي بمعدل قص عالي لمدة 30 يومًا. علاوة على ذلك، تم استكشاف الاستقرار المضاد للبكتيريا على المدى الطويل في الجسم الحي بشكل أكبر في نموذج زراعة تحت الجلد في الجرذان. أظهر اختبار عد مستعمرات أطباق الأجار أن طلاء HS/DAC أظهر معدل قتل بكتيري جيد.
طلاء HS/DAC ضد البكتيريا المرتبطة بالقسطرة
تم اكتشافها في دم خط الدورة الدموية للقسطرة العارية الملقحة بالبكتيريا (الشكل 5d iii والشكل التكميلي 20b، d). بالإضافة إلى ذلك، مقارنةً بالجلطة التي تشكلت على القسطرة العارية، فإن القسطرة العارية الملقحة بالبكتيريا عززت تشكيل الألياف وأغشية الفيبرين (الشكل 5d iv)، والتي ترتبط بتكوين الأغشية الحيوية.
نقاش
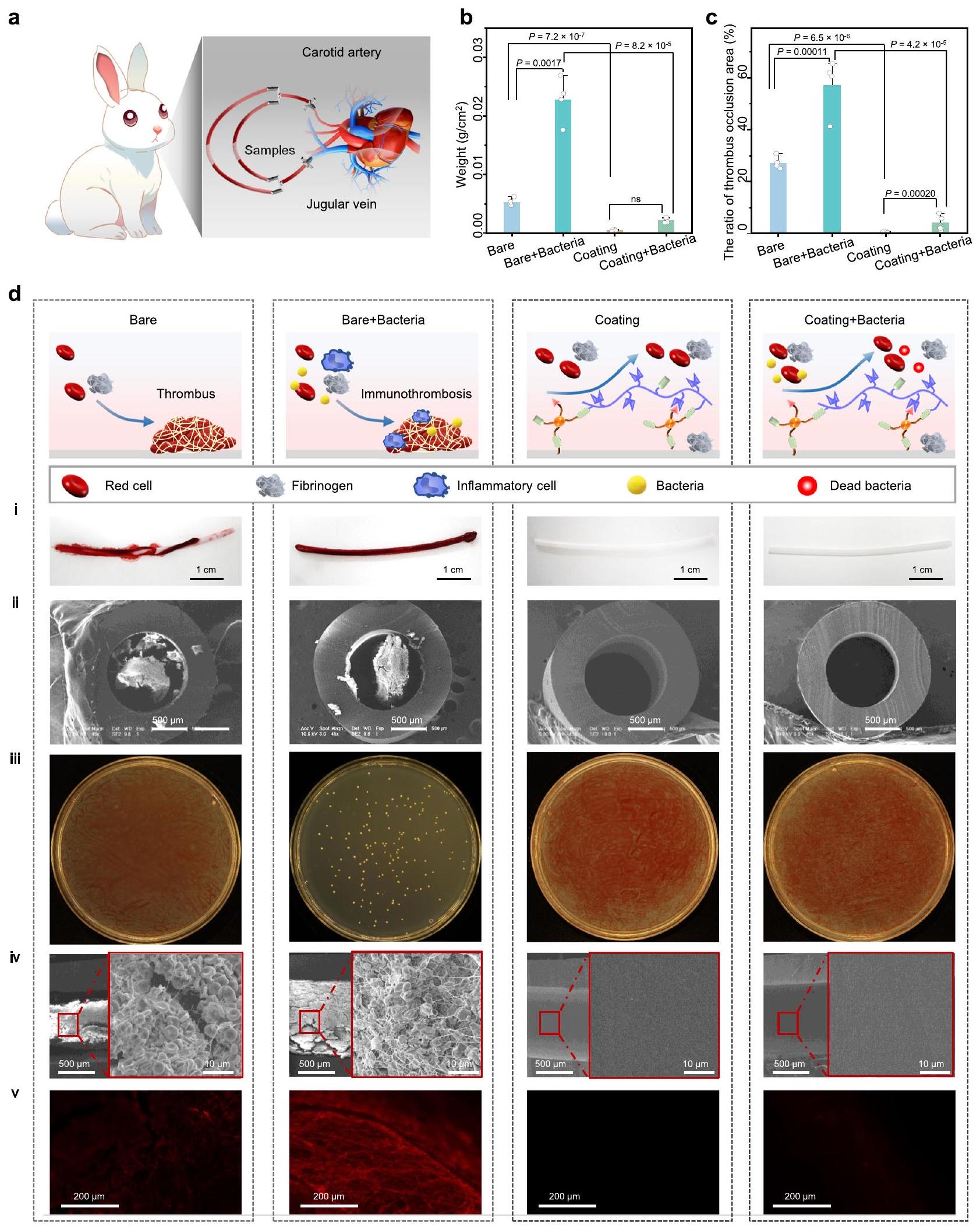
تم تقليل تكوين الجلطات المرتبطة بالعدوى والعدوى المرتبطة بالقسطرة بشكل كبير في نموذج أرنب خارج الجسم.
طرق
تحضير العينات المغلفة بـ HS/DAC
استقرار طلاء HS/DAC
الخثار المرتبط بالعدوى وبكتيريميا القسطرة المرتبطة بطبقة HS/DAC
التحليل الإحصائي
ملخص التقرير
توفر البيانات
References
- Parada, G. et al. Ultrathin and robust hydrogel coatings on cardiovascular medical devices to mitigate thromboembolic and infectious complications. Adv. Healthc. Mater. 9, 2001116 (2020).
- Woods, E., Percival, S. L. eds. Biofilms in infection prevention and control: a Healthcare Handbook. America: Academic Press. (2014).
- Verso, M. & Agnelli, G. Venous thromboembolism associated with long-term use of central venous catheters in cancer patients. J. Clin. Oncol. 21, 3665-3675 (2003).
- Zakhour, R., Chaftari, A. M. & Raad, I. I. Catheter-related infections in patients with haematological malignancies: novel preventive and therapeutic strategies. Lancet Infect. Dis. 16, 241-250 (2016).
- Carre, Y. et al. Randomized study of antiseptic application technique in healthy volunteers before vascular access insertion (TApAS trial). J. Infect. 81, 532-539 (2020).
- Citla Sridhar, D., Abou-Ismail, M. Y. & Ahuja, S. P. Central venous catheter-related thrombosis in children and adults. Thromb. Res. 187, 103-112 (2020).
- Budnik, I. & Brill, A. Immune factors in deep vein thrombosis initiation. Trends Immunol. 39, 610-623 (2018).
- Vertes, A., Hitchins, V. & Phillips, K. S. Analytical challenges of microbial biofilms on medical devices. Anal. Chem. 84, 3858-3866 (2012).
- Gahlot, R., Nigam, C., Kumar, V., Yadav, G. & Anupurba, S. Catheterrelated bloodstream infections. Int J. Crit. Illn. Inj. Sci. 4, 162-167 (2014).
- Fletcher, S. Catheter-related bloodstream infection. BJA Educ. 5, 49-51 (2005).
- Ding, M. et al. Stimuli-responsive nanocarriers for bacterial biofilm treatment. Rare Met. 41, 482-498 (2022).
- Hou, Z. et al. Precisely structured nitric-oxide-releasing copolymer brush defeats broad-spectrum catheter-associated biofilm infections in vivo. ACS Cent. Sci. 6, 2031-2045 (2020).
- Hanna, R. & Raad, I. I. Diagnosis of catheter-related bloodstream infection. Curr. Infect. Dis. Rep. 7, 413-419 (2005).
- Stark, K. & Massberg, S. Interplay between inflammation and thrombosis in cardiovascular pathology. Nat. Rev. Cardiol. 18, 666-682 (2021).
- Mehall, J. R. et al. Fibrin sheath enhances central venous catheter infection. Crit. Care Med. 30, 908-912 (2002).
- Yang, X. et al. Bacterial endotoxin activates the coagulation cascade through gasdermin D-dependent phosphatidylserine exposure. Immunity 51, 983-996 (2019).
- Wang, R., Chua, K. L. & Neoh, K. G. Bifunctional coating with sustained release of 4-amide-piperidine-C12 for long-term prevention of bacterial colonization on silicone. ACS Biomater. Sci. Eng. 1, 405-415 (2015).
- Golabchi, A. et al. Zwitterionic polymer/polydopamine coating reduce acute inflammatory tissue responses to neural implants. Biomaterials 225, 119519 (2019).
- Bujold, K. E., Halstead, E. S. & Xavier, F. Continuous low-dose heparin infusion for catheter-related thrombosis prophylaxis in critically-Ill children. Blood 136, 9-10 (2020).
- Zhang, D. et al. Dealing with the foreign-body response to implanted biomaterials: strategies and applications of new materials. Adv. Funct. Mater. 31, 2007226 (2020).
- Ding, X. et al. Versatile antibacterial materials: an emerging arsenal for combatting bacterial pathogens. Adv. Funct. Mater. 28, 1802140 (2018).
- Zander, Z. K. et al. Post-fabrication QAC-functionalized thermoplastic polyurethane for contact-killing catheter applications. Biomaterials 178, 339-350 (2018).
- Lavery, K. S. et al. Anti-thrombotic technologies for medical devices. Adv. Drug Deliv. Rev. 112, 2-11 (2017).
- Semak, V., Fischer, M. B. & Weber, V. Biomimetic principles to develop blood compatible surfaces. Int J. Artif. Organs 40, 22-30 (2017).
- Reviakine, I. et al. Stirred, shaken, or stagnant: What goes on at the blood-biomaterial interface. Blood Rev. 31, 11-21 (2017).
- Li, P. et al. Preparation of phospholipid-based polycarbonate urethanes for potential applications of blood-contacting implants. Regen. Biomater. 7, 491-504 (2020).
- Zhou, J. et al. Robust and versatile coatings engineered via simultaneous covalent and noncovalent interactions. Angew. Chem. Int. Ed. 60, 20225-20230 (2021).
- Zhou, J. et al. Polyphenol-mediated assembly for particle engineering. Acc. Chem. Res. 53, 1269-1278 (2020).
- Hu, B. et al. Polyphenol-binding amyloid fibrils self-assemble into reversible hydrogels with antibacterial activity. ACS Nano 12, 3385-3396 (2018).
- Su, H. et al. Paclitaxel-promoted supramolecular polymerization of peptide conjugates. J. Am. Chem. Soc. 141, 11997-12004 (2019).
- Xie, L. et al. Programmed surface on poly(aryl-ether-ether-ketone) initiating immune mediation and fulfilling bone regeneration sequentially. Innovation 2, 100148 (2021).
- Öhman, M. & Persson, D. ATR-FTIR Kretschmann spectroscopy for interfacial studies of a hidden aluminum surface coated with a silane film and epoxy I. Characterization by IRRAS and ATR-FTIR. Surf. Interfaces 44, 133-143 (2012).
- Yu, H. et al. Water-insoluble polymeric guanidine derivative and application in the preparation of antibacterial coating of catheter. ACS Appl. Mater. Interfaces 10, 39257-39267 (2018).
- Yu, H. et al. Fabrication of polylysine based antibacterial coating for catheters by facile electrostatic interaction. Chem. Eng. J. 360, 1030-1041 (2019).
- Liu, L. et al. One-step hydrophobization of tannic acid for antibacterial coating on catheters to prevent catheter-associated infections. Biomater. Sci. 7, 5035-5043 (2019).
- Wang, X. et al. Electrostatic assembly functionalization of poly (
glutamic acid) for biomedical antibacterial applications. J. Mater. Sci. Technol. 59, 14-25 (2020). - Li, C. et al. Thrombosis-responsive thrombolytic coating based on thrombin-degradable tissue plasminogen activator (t-PA) nanocapsules. Adv. Funct. Mater. 27, 1703934 (2017).
- Biran, R. & Pond, D. Heparin coatings for improving blood compatibility of medical devices. Adv. Drug Deliv. Rev. 112, 12-23 (2017).
- Lin, F. et al. Porous polymers as universal reversal agents for heparin anticoagulants through an inclusion-sequestration mechanism. Adv. Mater. 34, 2200549 (2022).
- Karlsson, R. et al. Dissecting structure-function of 3-O-sulfated heparin and engineered heparan sulfates. Sci. Adv. 7, eabl6026 (2021).
- Jaffer, I. H. et al. Medical device-induced thrombosis: what causes it and how can we prevent it? J. Thromb. Haemost. 13, S72-S81 (2015).
- Afshar-Kharghan, V. & Thiagarajan, P. Leukocyte adhesion and thrombosis. Curr. Opin. Hematol. 13, 34-39 (2006).
- Yao, M. et al. Microgel reinforced zwitterionic hydrogel coating for blood-contacting biomedical devices. Nat. Commun. 13, 5339 (2022).
- Chae, K. et al. Antibacterial infection and immune-evasive coating for orthopedic implants. Sci. Adv. 6, eabb0025 (2020).
- Sekhavat Pour, Z., Makvandi, P. & Ghaemy, M. Performance properties and antibacterial activity of crosslinked films of quaternary ammonium modified starch and poly(vinyl alcohol). Int. J. Biol. Macromol. 80, 596-604 (2015).
- Timsit, J. F. et al. New materials and devices for preventing catheterrelated infections. Ann. Intensive Care. 1, 34 (2011).
- Boersma, R. S. et al. Thrombotic and infectious complications of central venous catheters in patients with hematological malignancies. Ann. Oncol. 19, 433-442 (2008).
- Yang, C. et al. Brush-like polycarbonates containing dopamine, cations, and PEG providing a broad-spectrum, antibacterial, and antifouling surface via one-step coating. Adv. Mater. 26, 7346-7351 (2014).
- Chen, Z. et al. Alternatingly amphiphilic antimicrobial oligoguanidines: structure-property relationship and usage as the coating material with unprecedented hemocompatibility. Chem. Mater. 34, 3670-3682 (2022).
- Cai, W. et al. Carboxyl-ebselen-based layer-by-layer films as potential antithrombotic and antimicrobial coatings. Biomaterials 32, 7774-7784 (2011).
- Pinese, C. et al. Simple and specific grafting of antibacterial peptides on silicone catheters. Adv. Healthc. Mater. 5, 3067-3073 (2016).
- Park, S. K. et al. Polysaccharide-derivative coated intravascular catheters with superior multifunctional performance via simple and biocompatible method. Chem. Eng. J. 433, 134565 (2022).
- Yu, Y. et al. A tough, slippery, and anticoagulant doublenetwork hydrogel coating. ACS Appl. Polym. Mater. 4, 5941-5951 (2022).
- Long, L. et al. A hydrophobic cationic polyphenol coating for versatile antibacterial and hemostatic devices. Chem. Eng. J. 444, 135426 (2022).
- Keum, H. et al. Prevention of bacterial colonization on catheters by a one-step coating process involving an antibiofouling polymer in water. ACS Appl. Mater. Interfaces 9, 19736-19745 (2017).
- Yan, S. et al. Nonleaching bacteria-responsive antibacterial surface based on a unique hierarchical architecture. ACS Appl. Mater. Interfaces 8, 24471-24481 (2016).
- Fan, Y. et al. Immobilization of nano Cu-MOFs with polydopamine coating for adaptable gasotransmitter generation and copper ion delivery on cardiovascular stents. Biomaterials 204, 36-45 (2019).
- Zhang, B. et al. Epigallocatechin gallate mediated sandwich-like coating for mimicking endothelium with sustained therapeutic nitric oxide generation and heparin release. Biomaterials 269, 120418 (2021).
الشكر والتقدير
مساهمات المؤلفين
المصالح المتنافسة
معلومات إضافية
المواد التكميلية متاحة على
https://doi.org/10.1038/s41467-023-44478-3.
يجب توجيه المراسلات والطلبات للحصول على المواد إلى شيفانغ لوان أو هينغتشونغ شي.
http://www.nature.com/reprints
© المؤلف(ون) 2024
المختبر الوطني الرئيسي لفيزياء وكيمياء البوليمرات، معهد تشانغتشون للكيمياء التطبيقية، الأكاديمية الصينية للعلوم، تشانغتشون 130022، الصين. جامعة العلوم والتكنولوجيا في الصين، هيفي 230026، الصين. المختبر الرئيسي للبحث وتقييم استقلاب الأدوية في إدارة الغذاء والدواء الوطنية، المختبر الرئيسي لمقاطعة قوانغدونغ لفحص الأدوية الجديدة، كلية العلوم الصيدلانية، جامعة الطب الجنوبي، قوانغتشو 510515، الصين.
ساهم هؤلاء المؤلفون بالتساوي: لين ليو، هوان يوي. البريد الإلكتروني:sfluan@ciac.ac.cn؛shihc@ciac.ac.cn
DOI: https://doi.org/10.1038/s41467-023-44478-3
PMID: https://pubmed.ncbi.nlm.nih.gov/38167880
Publication Date: 2024-01-02
Heparin-network-mediated long-lasting coatings on intravascular catheters for adaptive antithrombosis and antibacterial infection
Accepted: 14 December 2023
(A) Check for updates
Abstract
Bacteria-associated infections and thrombosis, particularly catheter-related bloodstream infections and catheter-related thrombosis, are life-threatening complications. Herein, we utilize a concise assembly of heparin sodium with organosilicon quaternary ammonium surfactant to fabricate a multifunctional coating complex. In contrast to conventional one-time coatings, the complex attaches to medical devices with arbitrary shapes and compositions through a facile dipping process and further forms robust coatings to treat catheterrelated bloodstream infections and thrombosis simultaneously. Through their robustness and adaptively dissociation, coatings not only exhibit good stability under extreme conditions but also significantly reduce thrombus adhesion by
deep vein thrombosis with high mortality
Results
Formation of stable HS/DAC coating
(alkyl chain

a llustration of the procedure used to prepare the HS/DAC complex and HS/DAC
coating.
the coating was intact (Fig. 2f iii). We further explored the long-term stability of HS/DAC coating in flowing
Anti-thrombotic property of the HS/DAC coating

with samples for 2 h to study Fg adhesion. Confocal laser scanning microscopy (CLSM) images indicated that Fg was less adhered in the HS/DAC-coated catheter than the bare catheter (Fig. 3d). On the contrary, negligible differences were observed in the amount of bovine serum albumin (BSA) by bicinchoninic acid (BCA) assay when samples soaked in PBS solution of BSA (Fig. 3c). It is speculated that this different phenomenon is related to the activation of Fg. Conformational changes in Fg structure during Fg activation to form a fibrin network result in exposure of multiple binding sites. These binding sites can bind to various proteins, causing protein adsorption. Hence, the effect of HS/DAC coating on Fg activation after 2 h of circulation in whole blood was investigated by assessing the exposure of the








participate in the formation of thrombus
salt-triggered effect, we examined the physicochemical properties of the HS/DAC coating before and after treatment in salt solution. The self-adaptivity of (HS/DAC)-TPU was characterized by static water contact angles, FTIR and XPS. After incubation with PBS solution, the contact angle of (HS/DAC)-TPU decreased significantly, and the band of the carboxyl group shifted from 1609 to
Antibacterial and biocompatibility properties of the HS/DAC coating in vitro and in vivo

further confirm the inflammation regulation prompted by bacterial infection. As shown in Fig. 4g, the detected factors were significantly reduced in the HS/DAC-coated catheter, which is consistent with the histological assay results.
catheter-related infection in vitro and in vivo. Compared to the (HS/QAC)-catheter, the (HS/DAC)-catheter still maintained its original antibacterial activity even after one month of treatment in
independent samples). Measurements were repeated three times independently with similar results.
high as 97% even if flowing in artificial blood with high shear rate for 30 days. Furthermore, the long-term antibacterial stability in vivo was further explored in a rat subcutaneous implantation model. Agar plate colony counting assay demonstrated that the HS/DAC coating exhibited a good bactericidal rate (
HS/DAC coating against catheter-related bacteremia
were detected in the blood of the circulation line of bare catheter inoculated bacteria (Fig. 5d iii and Supplementary Fig. 20b, d). In addition, compared to the thrombus formed on the bare catheter, the bare catheter inoculated with bacteria promoted the formation of fiber and fibrin sheaths (Fig. 5d iv), which are associated with biofilm formation
Discussion

the formation of infection-related thrombi and catheter-related infection was significantly reduced in an ex vivo rabbit model.
Methods
Preparation of HS/DAC-coated samples
Stability of the HS/DAC coating
Anti-infection-related thrombus and catheter-related bacteremia of HS/DAC coating
Statistical analysis
Reporting summary
Data availability
References
- Parada, G. et al. Ultrathin and robust hydrogel coatings on cardiovascular medical devices to mitigate thromboembolic and infectious complications. Adv. Healthc. Mater. 9, 2001116 (2020).
- Woods, E., Percival, S. L. eds. Biofilms in infection prevention and control: a Healthcare Handbook. America: Academic Press. (2014).
- Verso, M. & Agnelli, G. Venous thromboembolism associated with long-term use of central venous catheters in cancer patients. J. Clin. Oncol. 21, 3665-3675 (2003).
- Zakhour, R., Chaftari, A. M. & Raad, I. I. Catheter-related infections in patients with haematological malignancies: novel preventive and therapeutic strategies. Lancet Infect. Dis. 16, 241-250 (2016).
- Carre, Y. et al. Randomized study of antiseptic application technique in healthy volunteers before vascular access insertion (TApAS trial). J. Infect. 81, 532-539 (2020).
- Citla Sridhar, D., Abou-Ismail, M. Y. & Ahuja, S. P. Central venous catheter-related thrombosis in children and adults. Thromb. Res. 187, 103-112 (2020).
- Budnik, I. & Brill, A. Immune factors in deep vein thrombosis initiation. Trends Immunol. 39, 610-623 (2018).
- Vertes, A., Hitchins, V. & Phillips, K. S. Analytical challenges of microbial biofilms on medical devices. Anal. Chem. 84, 3858-3866 (2012).
- Gahlot, R., Nigam, C., Kumar, V., Yadav, G. & Anupurba, S. Catheterrelated bloodstream infections. Int J. Crit. Illn. Inj. Sci. 4, 162-167 (2014).
- Fletcher, S. Catheter-related bloodstream infection. BJA Educ. 5, 49-51 (2005).
- Ding, M. et al. Stimuli-responsive nanocarriers for bacterial biofilm treatment. Rare Met. 41, 482-498 (2022).
- Hou, Z. et al. Precisely structured nitric-oxide-releasing copolymer brush defeats broad-spectrum catheter-associated biofilm infections in vivo. ACS Cent. Sci. 6, 2031-2045 (2020).
- Hanna, R. & Raad, I. I. Diagnosis of catheter-related bloodstream infection. Curr. Infect. Dis. Rep. 7, 413-419 (2005).
- Stark, K. & Massberg, S. Interplay between inflammation and thrombosis in cardiovascular pathology. Nat. Rev. Cardiol. 18, 666-682 (2021).
- Mehall, J. R. et al. Fibrin sheath enhances central venous catheter infection. Crit. Care Med. 30, 908-912 (2002).
- Yang, X. et al. Bacterial endotoxin activates the coagulation cascade through gasdermin D-dependent phosphatidylserine exposure. Immunity 51, 983-996 (2019).
- Wang, R., Chua, K. L. & Neoh, K. G. Bifunctional coating with sustained release of 4-amide-piperidine-C12 for long-term prevention of bacterial colonization on silicone. ACS Biomater. Sci. Eng. 1, 405-415 (2015).
- Golabchi, A. et al. Zwitterionic polymer/polydopamine coating reduce acute inflammatory tissue responses to neural implants. Biomaterials 225, 119519 (2019).
- Bujold, K. E., Halstead, E. S. & Xavier, F. Continuous low-dose heparin infusion for catheter-related thrombosis prophylaxis in critically-Ill children. Blood 136, 9-10 (2020).
- Zhang, D. et al. Dealing with the foreign-body response to implanted biomaterials: strategies and applications of new materials. Adv. Funct. Mater. 31, 2007226 (2020).
- Ding, X. et al. Versatile antibacterial materials: an emerging arsenal for combatting bacterial pathogens. Adv. Funct. Mater. 28, 1802140 (2018).
- Zander, Z. K. et al. Post-fabrication QAC-functionalized thermoplastic polyurethane for contact-killing catheter applications. Biomaterials 178, 339-350 (2018).
- Lavery, K. S. et al. Anti-thrombotic technologies for medical devices. Adv. Drug Deliv. Rev. 112, 2-11 (2017).
- Semak, V., Fischer, M. B. & Weber, V. Biomimetic principles to develop blood compatible surfaces. Int J. Artif. Organs 40, 22-30 (2017).
- Reviakine, I. et al. Stirred, shaken, or stagnant: What goes on at the blood-biomaterial interface. Blood Rev. 31, 11-21 (2017).
- Li, P. et al. Preparation of phospholipid-based polycarbonate urethanes for potential applications of blood-contacting implants. Regen. Biomater. 7, 491-504 (2020).
- Zhou, J. et al. Robust and versatile coatings engineered via simultaneous covalent and noncovalent interactions. Angew. Chem. Int. Ed. 60, 20225-20230 (2021).
- Zhou, J. et al. Polyphenol-mediated assembly for particle engineering. Acc. Chem. Res. 53, 1269-1278 (2020).
- Hu, B. et al. Polyphenol-binding amyloid fibrils self-assemble into reversible hydrogels with antibacterial activity. ACS Nano 12, 3385-3396 (2018).
- Su, H. et al. Paclitaxel-promoted supramolecular polymerization of peptide conjugates. J. Am. Chem. Soc. 141, 11997-12004 (2019).
- Xie, L. et al. Programmed surface on poly(aryl-ether-ether-ketone) initiating immune mediation and fulfilling bone regeneration sequentially. Innovation 2, 100148 (2021).
- Öhman, M. & Persson, D. ATR-FTIR Kretschmann spectroscopy for interfacial studies of a hidden aluminum surface coated with a silane film and epoxy I. Characterization by IRRAS and ATR-FTIR. Surf. Interfaces 44, 133-143 (2012).
- Yu, H. et al. Water-insoluble polymeric guanidine derivative and application in the preparation of antibacterial coating of catheter. ACS Appl. Mater. Interfaces 10, 39257-39267 (2018).
- Yu, H. et al. Fabrication of polylysine based antibacterial coating for catheters by facile electrostatic interaction. Chem. Eng. J. 360, 1030-1041 (2019).
- Liu, L. et al. One-step hydrophobization of tannic acid for antibacterial coating on catheters to prevent catheter-associated infections. Biomater. Sci. 7, 5035-5043 (2019).
- Wang, X. et al. Electrostatic assembly functionalization of poly (
glutamic acid) for biomedical antibacterial applications. J. Mater. Sci. Technol. 59, 14-25 (2020). - Li, C. et al. Thrombosis-responsive thrombolytic coating based on thrombin-degradable tissue plasminogen activator (t-PA) nanocapsules. Adv. Funct. Mater. 27, 1703934 (2017).
- Biran, R. & Pond, D. Heparin coatings for improving blood compatibility of medical devices. Adv. Drug Deliv. Rev. 112, 12-23 (2017).
- Lin, F. et al. Porous polymers as universal reversal agents for heparin anticoagulants through an inclusion-sequestration mechanism. Adv. Mater. 34, 2200549 (2022).
- Karlsson, R. et al. Dissecting structure-function of 3-O-sulfated heparin and engineered heparan sulfates. Sci. Adv. 7, eabl6026 (2021).
- Jaffer, I. H. et al. Medical device-induced thrombosis: what causes it and how can we prevent it? J. Thromb. Haemost. 13, S72-S81 (2015).
- Afshar-Kharghan, V. & Thiagarajan, P. Leukocyte adhesion and thrombosis. Curr. Opin. Hematol. 13, 34-39 (2006).
- Yao, M. et al. Microgel reinforced zwitterionic hydrogel coating for blood-contacting biomedical devices. Nat. Commun. 13, 5339 (2022).
- Chae, K. et al. Antibacterial infection and immune-evasive coating for orthopedic implants. Sci. Adv. 6, eabb0025 (2020).
- Sekhavat Pour, Z., Makvandi, P. & Ghaemy, M. Performance properties and antibacterial activity of crosslinked films of quaternary ammonium modified starch and poly(vinyl alcohol). Int. J. Biol. Macromol. 80, 596-604 (2015).
- Timsit, J. F. et al. New materials and devices for preventing catheterrelated infections. Ann. Intensive Care. 1, 34 (2011).
- Boersma, R. S. et al. Thrombotic and infectious complications of central venous catheters in patients with hematological malignancies. Ann. Oncol. 19, 433-442 (2008).
- Yang, C. et al. Brush-like polycarbonates containing dopamine, cations, and PEG providing a broad-spectrum, antibacterial, and antifouling surface via one-step coating. Adv. Mater. 26, 7346-7351 (2014).
- Chen, Z. et al. Alternatingly amphiphilic antimicrobial oligoguanidines: structure-property relationship and usage as the coating material with unprecedented hemocompatibility. Chem. Mater. 34, 3670-3682 (2022).
- Cai, W. et al. Carboxyl-ebselen-based layer-by-layer films as potential antithrombotic and antimicrobial coatings. Biomaterials 32, 7774-7784 (2011).
- Pinese, C. et al. Simple and specific grafting of antibacterial peptides on silicone catheters. Adv. Healthc. Mater. 5, 3067-3073 (2016).
- Park, S. K. et al. Polysaccharide-derivative coated intravascular catheters with superior multifunctional performance via simple and biocompatible method. Chem. Eng. J. 433, 134565 (2022).
- Yu, Y. et al. A tough, slippery, and anticoagulant doublenetwork hydrogel coating. ACS Appl. Polym. Mater. 4, 5941-5951 (2022).
- Long, L. et al. A hydrophobic cationic polyphenol coating for versatile antibacterial and hemostatic devices. Chem. Eng. J. 444, 135426 (2022).
- Keum, H. et al. Prevention of bacterial colonization on catheters by a one-step coating process involving an antibiofouling polymer in water. ACS Appl. Mater. Interfaces 9, 19736-19745 (2017).
- Yan, S. et al. Nonleaching bacteria-responsive antibacterial surface based on a unique hierarchical architecture. ACS Appl. Mater. Interfaces 8, 24471-24481 (2016).
- Fan, Y. et al. Immobilization of nano Cu-MOFs with polydopamine coating for adaptable gasotransmitter generation and copper ion delivery on cardiovascular stents. Biomaterials 204, 36-45 (2019).
- Zhang, B. et al. Epigallocatechin gallate mediated sandwich-like coating for mimicking endothelium with sustained therapeutic nitric oxide generation and heparin release. Biomaterials 269, 120418 (2021).
Acknowledgements
Author contributions
Competing interests
Additional information
supplementary material available at
https://doi.org/10.1038/s41467-023-44478-3.
Correspondence and requests for materials should be addressed to Shifang Luan or Hengchong Shi.
http://www.nature.com/reprints
© The Author(s) 2024
State Key Laboratory of Polymer Physics and Chemistry, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, Changchun 130022, China. University of Science and Technology of China, Hefei 230026, China. NMPA Key Laboratory for Research and Evaluation of Drug Metabolism, Guangdong Provincial Key Laboratory of New Drug Screening, School of Pharmaceutical Sciences, Southern Medical University, Guangzhou 510515, China.
These authors contributed equally: Lin Liu, Huan Yu. e-mail: sfluan@ciac.ac.cn; shihc@ciac.ac.cn
