DOI: https://doi.org/10.1186/s43094-024-00667-5
تاريخ النشر: 2024-07-29
أحماض أوميغا-3 الدهنية: مراجعة علمية شاملة لمصادرها ووظائفها وفوائدها الصحية
الملخص
خلفية في السنوات الأخيرة، زاد الوعي العام بالأنظمة الغذائية الصحية بشكل كبير، مما أدى إلى زيادة استهلاك المكملات الغذائية. من بين هذه المكملات، أصبحت أحماض أوميغا-3 الدهنية شائعة بشكل خاص.
الخلفية
(1) زيادة في تناول الطاقة مقارنة بإنفاق الطاقة.
(2) زيادة في تناول الدهون المشبعة الغذائية، وأحماض أوميغا-6 الدهنية، والأحماض الدهنية المتحولة مقارنة بأحماض أوميغا-3 الدهنية.
(3) نظام غذائي بمحتوى أقل من الكربوهيدرات المعقدة والألياف.
(4) نظام غذائي بنسب أكبر من الحبوب مقارنة بالفواكه والخضروات.
(5) انخفاض في تناول البروتينات ومضادات الأكسدة والكالسيوم.
كانت الأدلة الأولية على الفوائد الصحية لأحماض أوميغا-3 الدهنية، وهي حمض الإيكوسابنتاينويك (EPA) وحمض الدوكوساهيكسانويك (DHA)، تأتي من الإسكيمو في غرينلاند، الذين تناولوا نظامًا غذائيًا غنيًا بالأسماك وكان لديهم معدلات منخفضة من التصلب المتعدد، والربو، والسكري من النوع الأول، وأمراض القلب التاجية. أظهرت العديد من الدراسات أن الدهون أوميغا-3 ضرورية لجسم الإنسان لتخليق أغشية الخلايا في الجسم وتأثيرها على وظيفة مستقبلات الخلايا في هذه الأغشية. تعمل كعنصر أساسي من الهرمونات التي تنظم التخثر، والالتهاب، وانقباض واسترخاء جدران الشرايين. علاوة على ذلك، يمكن أن ترتبط بمستقبلات الخلايا التي تنظم النشاط الجيني. تشمل هذه الفوائد الوقاية من أمراض القلب والسكتة الدماغية، وإدارة الذئبة، والإكزيما، والتهاب المفاصل الروماتويدي، ووظيفة أحماض أوميغا-3 كعوامل وقائية ضد السرطان وأمراض أخرى [5-7].
تشمل الأحماض الدهنية غير المشبعة العالية (PUFAs) حمض ألفا-لينولينيك (ALA)، وحمض الدوكوساهيكسانويك (DHA)، وحمض الإيكوسابنتاينويك (EPA) و
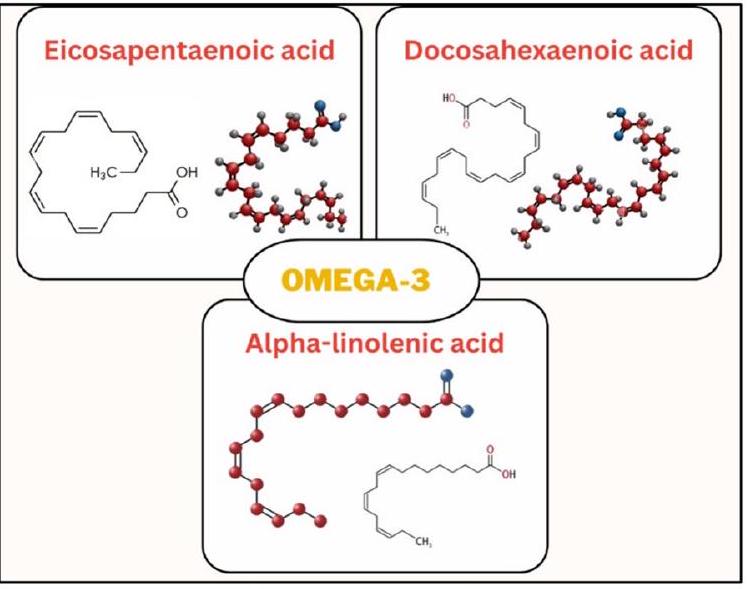
النص الرئيسي
أنواع أحماض أوميغا-3 الدهنية
مصادر أوميغا-3
تخليق أوميغا-3
تؤثر التوازن الهرموني، وتثبيط التغذية الناتج النهائي، والحالة الغذائية على أنشطة
دلتا-6 ودلتا-5 ديساتوراز. لتحويل EPA إلى DHA (22:6n-3)، يجب إضافة كربونين إلى EPA لتشكيل DHA.
الفوائد الصحية العلاجية لأوميغا-3

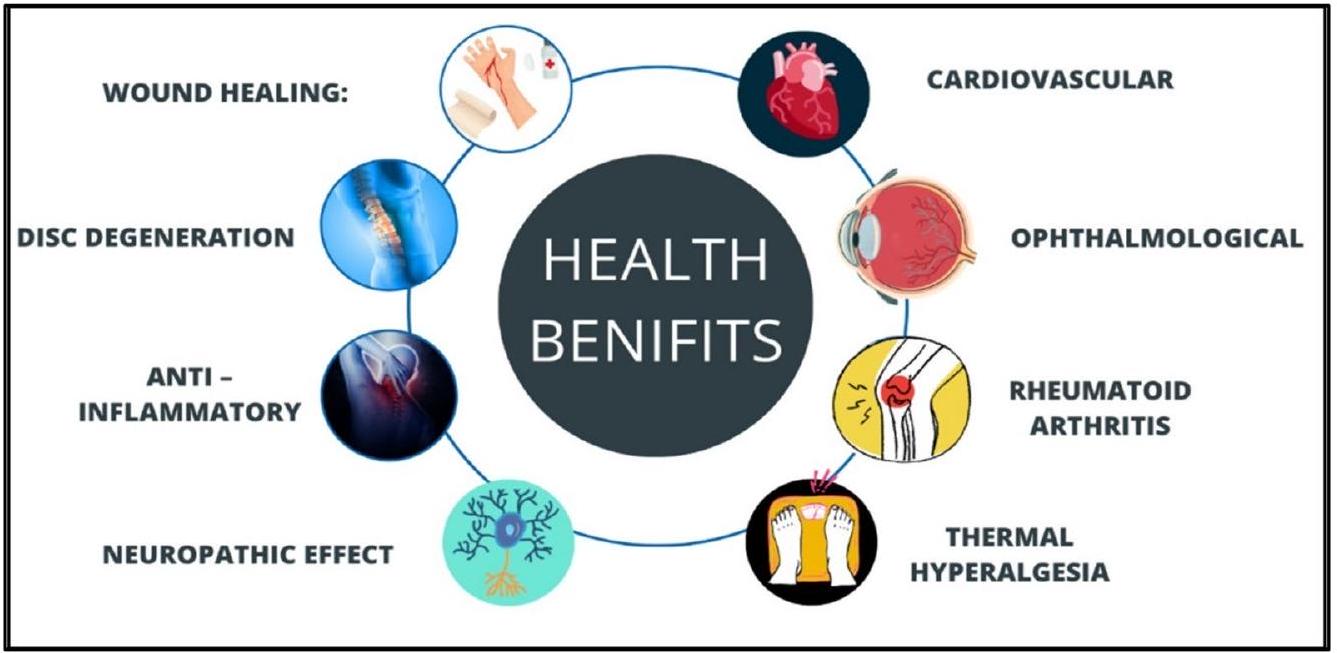
آلية الأحماض الدهنية أوميغا-3
آثار مكملات أوميغا-3 على النتائج الصحية
| رقم السجل | مرض | آلية العمل | المستقبلات المعنية | المزايا المقدمة | المراجع |
| 1 | أمراض القلب والأوعية الدموية | تغيير تركيبة الفوسفوليبيد في غشاء الميتوكوندريا | مستقبلات البروتين G المرتبطة (GPRs)، مستقبلات المنشطات المفعلة للبروكسيسوم (PPARs) | يقلل من استهلاك الأكسجين في الميتوكوندريا، يخفض ضغط الدم، يحسن وظيفة الأوعية الدموية | [30-34] |
| ٢ | مرض عيون | تغيير خصائص الطور الدهني، السماكة، السيولة والنفاذية. تجديد الرودوبسين والتحويل الضوئي | تم دمج GPRs و PPARs مباشرة في أغشية الشبكية | من خلال تنظيم التهاب سطح العين وتعزيز ملفات الدهون في الدموع، يخفف DHA من الأعراض والمؤشرات المرتبطة بمتلازمة جفاف العين. | [٢٩، ٣٥-٣٧] |
| ٣ | فرط الألم | تقوم أحماض أوميغا-3 بتثبيط تعبير الإنزيمات والبروتينات السيتوكينية المسببة للالتهابات. آلية تعديل قنوات الأيونات. | مستقبلات PGE2، قنوات الأيون TRPV1 و TRPM8 | تقليل الالتهاب وانخفاض الحساسية غير الطبيعية للألم | [٣٨-٤٠] |
| ٤ | مرض عصبي | يقلل من السيتوكينات الالتهابية | TNF-ألفا، إنترلوكين-1 وإنترلوكين-6 | تعديل مركزي
|
[٣٩-٤١] |
| ٥ | مضاد للالتهابات | أدى DHA إلى تثبيط تلامس الخلايا البيضاء مع خلايا البطانة، وتعبير جزيئات الالتصاق، وتوليد السيتوكينات الالتهابية الكلاسيكية. | مستقبل PGE2، TNF-ألفا، إنترلوكين وCOX-2 | يقلل الالتهاب والألم في المفاصل ويخفف التورم | [16] |
| ٦ | تنكس القرص الفقري | أوميغا 3 ستقلل من السيتوكينات الالتهابية وتتفاعل مع المستقبلات على خلايا القرص. التخليق، تكسير الغضروف، والموت الخلوي المبرمج. | مستقبل PGE2، TNF-ألفا، إنترلوكين | يقلل الالتهاب والتدهور كما يعزز صحة الأقراص وإصلاح الأنسجة | [42,43] |
| ٧ | شفاء الجروح | تعديل الالتهاب. تعزيز تكوين الأوعية الدموية. تعزيز تخليق الكولاجين وتنظيمه | السيتوكينات الالتهابية مثل IL-6 و TNF-a. IL-10. تكاثر الكيراتينوسيت. مستقبلات البروتين G المرتبطة (GPCRs) | تعزيز عملية الشفاء بشكل أسرع وأكثر كفاءة. تحسين تدفق الدم يسرع الشفاء | [٣٧، ٤٤] |
| ٨ | متلازمة الأيض | خفض الدهون الثلاثية. تحسين حساسية الأنسولين. تقليل الالتهاب. تعديل ضغط الدم. | مستقبلات البروتين المنشط لبيروكسيسوم (PPARs). GPR120 و GPR40 | يقلل من مستويات الدهون الثلاثية في الدم. يزيد من استجابة الخلايا تجاه الأنسولين، مما يؤدي إلى تحسين التحكم في مستوى السكر في الدم. | [45] |
| 9 | التطور العصبي | كتل بناء الغشاء تحفيز الأعصاب. تعديل الالتهاب العصبي. تنظيم التعبير الجيني | مستقبلات البروتين G المرتبطة (GPCRs). مستقبلات حمض الريتينويك (RARs). مستقبلات المنشطات المتعددة للبروكسيسوم (PPARs) | يضمن تناول DHA الهيكل والوظيفة العصبية السليمة، مما يسهل نقل الإشارات ومعالجة المعلومات. | [٤٦، ٤٧] |
مشكلة الاستقرار
يحدث أكسدة الأحماض الدهنية المتعددة غير المشبعة طويلة السلسلة أوميغا-3 في ثلاثة
مراحل
مرحلة الانتشار
- هجوم الأكسجين يتفاعل الجذير الدهني مع الأكسجين لتكوين جذير بيروكسيلي (LOO).
- تفاعل سلسلة الجذري البيروكسيلي يستخلص ذرة هيدروجين من جزيء دهني آخر، مما يولد جذري دهني آخر وينشر تفاعل السلسلة.
- تكوين الهيدروبيروكسيد يتحد ذرة الهيدروجين المستخرجة مع الأكسجين لتكوين هيدروبيروكسيد (LOOH)، وهو المنتج الأساسي للأكسدة.
مرحلة إنهاء
- جمع الجذور يمكن لجذريين أن يتحدا، مما ينهي سلسلة التفاعل. بدلاً من ذلك، يمكن لمضادات الأكسدة أن تلتقط الجذور، مما يمنع المزيد من الانتشار.
ردود الفعل الثانوية
طرق لتعزيز استقرار تغليف زيت أوميغا-3
استقرار الأطعمة التي تحتوي على أحماض دهنية غير مشبعة طويلة السلسلة أوميغا-3 من خلال التعبئة في جو معدل
تطبيقات زيت أوميغا-3 في الصياغة والتطوير وتحديات التوافر الحيوي
أنظمة التوصيل المتقدمة
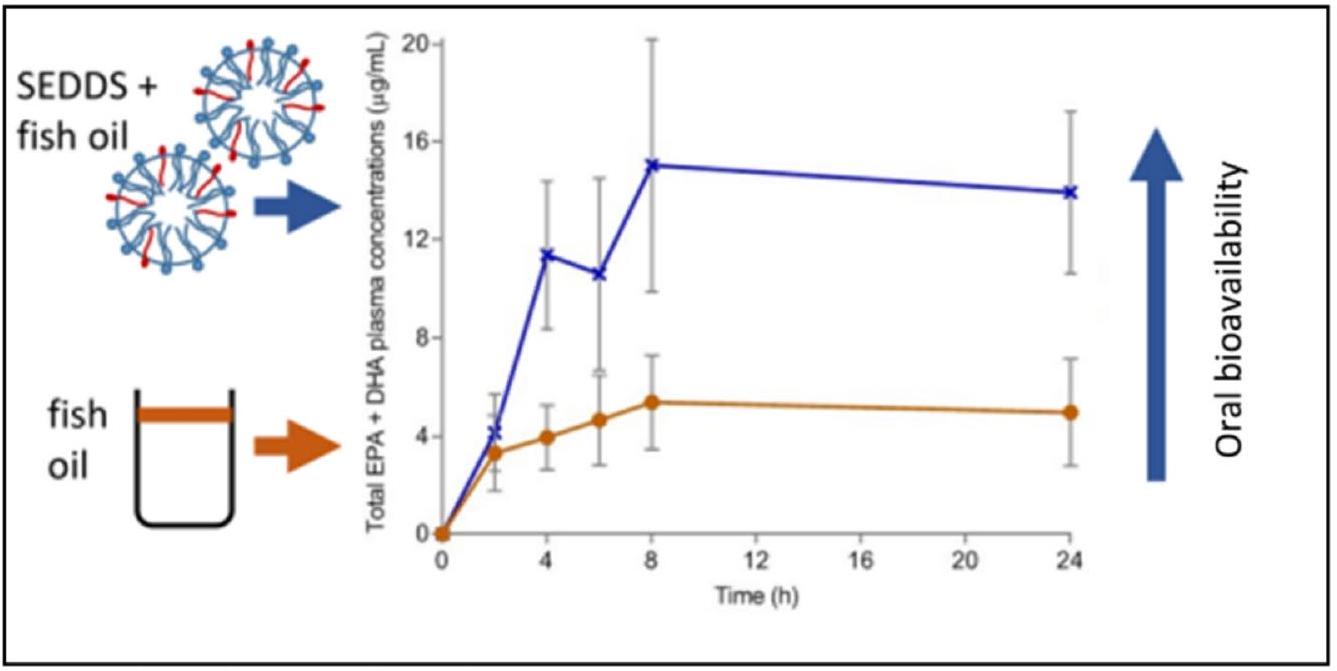
تمت دراسة أنظمة توصيل الأدوية الذاتية النانوإموليشن (SNEDDS) لتعزيز توافر DHA الحيوي بواسطة Alhakamy Na وآخرين للتغلب على القيود المفروضة بسبب ذوبانه الضعيف. كان حجم القطرات الأمثل 111.5 نانومتر، وإمكانات زيتا -23.53 مللي فولت، مما عزز امتصاص واستقرار التركيبة. أدى إعطاء SNEDDS إلى زيادة بمقدار 3.32 مرة في مستويات DHA في البلازما مقارنة بالتشتت المائي، مما يبرز فعالية هذا النهج في التوصيل [53].
تقنيات الميكروكبسولة يمكن أن تمنع أكسدة الأحماض الدهنية أوميغا-3، وتزيد من مدة صلاحيتها، وتخفي النكهات غير المرغوب فيها. كما أنها توفر طريقة فعالة لتثبيت وتحسين إدارة المكونات النشطة بيولوجيًا. تعتبر عمليات التجفيف بالرش والتجمع المعقد من أكثر الطرق التجارية شيوعًا للميكروكبسولة. وقد أجرت دراسة بواسطة ناتاليا كاستيجون وزملائها فحصت تأثيرات عملية الميكروكبسولة على تركيبة الأحماض الدهنية وأفادت بأن الدهون أوميغا-3 الميكروكبسولة ذات أحجام جزيئية تتراوح من 1.5 إلى
موافقة إدارة الغذاء والدواء للاستخدام الآمن لزيوت أوميغا 3
على مدى العشرين عامًا الماضية، قامت إدارة الغذاء والدواء الأمريكية (FDA) وغيرها من الهيئات التنظيمية الدولية بتقييم بعض مصادر زيت DHA أو الغني بـ DHA لإمكانية تضمين DHA في الأطعمة المخصصة للاستهلاك البشري. تعتبر GRN 137 واحدة من إشعارات GRAS الأمريكية ذات الصلة (FDA، 2008). تضمنت كل إشعار GRAS تفاصيل وبيانات من دراسات سريرية لدعم سلامة المكونات المقترحة من DHA للاستخدام في الأطعمة المخصصة للاستهلاك البشري.
لقد وافقت إدارة الغذاء والدواء الأمريكية على استخدام بعض الادعاءات الصحية المؤهلة بشأن قدرة الأطعمة والمكملات الغذائية التي تحتوي على أحماض أوميغا-3 الدهنية، EPA وDHA، على خفض ضغط الدم وتقليل خطر ارتفاع ضغط الدم وأمراض القلب التاجية. تم إصدار الحكم استجابةً لطلب تم تقديمه في عام 2014 من قبل المنظمة العالمية لأحماض أوميغا-3 EPA وDHA (GOED).
لخفض حدوث الأحداث القلبية الوعائية لدى بعض المرضى الذين يعانون من أمراض القلب والأوعية الدموية (CVD) أو الذين لديهم مخاطر عالية للإصابة بها، قامت إدارة الغذاء والدواء الأمريكية (FDA) بتفويض استخدام جديد للإيكوسابنت إيثيل (فاسسيبا)، وهو دواء يعد نسخة مصفاة بشكل كبير من حمض الإيكوسابنتاينويك (EPA)، وهو حمض دهني أوميغا-3 موجود في الأسماك. العلامة التجارية EPA/DHA إيبانوفا من أسترازينيكا ونسختان جنريك، والتي تستهدف جميعها فرط ثلاثي الغليسريد (مستويات ثلاثي الغليسريد من
تملكها شركة ليندساي غولدبرغ ذ.م.م وشركة آكر بيو مارين إيه إس إيه [60].
تفاعلات أوميغا-3 مع الأدوية
نقص أوميغا-3
نصف العمر
تناول أوميغا-3
الأحماض (حوالي 40 ملغ للأطفال والمراهقين وحوالي 90 ملغ للبالغين) [7].
الخاتمة
الاختصارات
| وكالة حماية البيئة | حمض الإيكوسابنتاينويك |
| DHA | حمض الدوكوساهيكسانويك |
| ألا | حمض ألفا-linolenic |
| الأحماض الدهنية المتعددة غير المشبعة | أحماض دهنية متعددة غير مشبعة عالية |
| OM3FAs | أحماض أوميغا-3 الدهنية |
| LPL | ليباز البروتين الدهني |
| مستقبلات البيروكسيزوم المتعددة | مستقبلات محفزات تكاثر البيروكسيسوم |
| القط 1 | أسيتيل كارنيتين ترانسفيراز 1 |
| جي بي آر | مستقبلات البروتين G المرتبطة |
شكر وتقدير
مساهمات المؤلفين
تمويل
توفر البيانات والمواد
الإعلانات
موافقة الأخلاقيات والموافقة على المشاركة
المصالح المتنافسة
نُشر على الإنترنت: 29 يوليو 2024
References
- Theinel MH, Nucci MP, Alves AH, Dias OFM, Mamani JB, Garrigós MM, Oliveira FA, Rego GNA, Valle NME, Cianciarullo G, Gamarra LF (2023) The effects of omega-3 polyunsaturated fatty acids on breast cancer as a preventive measure or as an adjunct to conventional treatments. Nutrients 15:1310. https://doi.org/10.3390/nu15061310
- Sharifi E, Yazdani Z, Najafi M, Hosseini-khah Z, Jafarpour A, Rafiei A (2022) The combined effect of fish oil containing omega-3 fatty acids and Lactobacillus plantarum on colorectal cancer. Food Sci Nutr 10:4411-4418. https://doi.org/10.1002/fsn3.3037
- Liang P, Henning SM, Guan J, Grogan T, Elashoff D, Cohen P, Aronson WJ (2020) Effect of dietary omega-3 fatty acids on castrate-resistant prostate cancer and tumor-associated macrophages. Prostate Cancer Prostatic Dis 23:127-135. https://doi.org/10.1038/s41391-019-0168-8
- Simopoulos AP (2008) The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp Biol Med 233:674-688. https://doi.org/10.3181/0711-MR-311
- Ratanabanangkoon P, Guzman H, Almarsson O, Berkovitz D, Tokarcyzk S, Straughn AB, Chen H (2008) A high-throughput approach towards a novel formulation of fenofibrate in omega-3 oil. Eur J Pharm Sci 33:351-360. https://doi.org/10.1016/j.ejps.2008.01.005
- Elagizi A, Lavie CJ, O’Keefe E, Marshall K, O’Keefe JH, Milani RV (2021) An update on omega-3 polyunsaturated fatty acids and cardiovascular health. Nutrients 13:204. https://doi.org/10.3390/nu13010204
- The Nutrition Source. Omega-3 fatty acids: an essential contribution [Internet]. 2023 [cited 2024 Jun 27]. Available from: https://www.hsph. harvard.edu/nutritionsource/what-should-you-eat/fats-and-cholesterol/ types-of-fat/omega-3-fats/
- Djuricic I, Calder PC (2021) Beneficial outcomes of omega-6 and omega-3 polyunsaturated fatty acids on human health: an update for 2021. Nutrients 13:2421. https://doi.org/10.3390/nu13072421
- Wikipedia contributors. Omega-3 fatty acid [Internet]. Wikipedia. 2024 [cited 2024 Jun 27]. Available from: https://en.wikipedia.org/wiki/Omega3_fatty_acid
- Tur JA, Bibiloni MM, Sureda A, Pons A (2012) Dietary sources of omega 3 fatty acids: public health risks and benefits. Br J Nutr 107:S23-S52. https:// doi.org/10.1017/S0007114512001456
- Office of Dietary Supplements. Omega-3 fatty acids [Internet]. [Cited 2024 Jun 27]. Available from: https://ods.od.nih.gov/factsheets/Omega 3FattyAcids-HealthProfessional/
- Miller MR, Nichols PD, Carter CG (2008) Omega 3 Oil sources for use in aquaculture-alternatives to the unsustainable harvest of wild fish. Nutr Res Rev 21:85-96. https://doi.org/10.1017/S0954422408102414
- Davidson MH, Kling D, Maki KC (2011) Novel developments in omega-3 fatty acid-based strategies. Curr Opin Lipidol 22:437-444. https://doi.org/ 10.1097/MOL.0b013e32834bd642
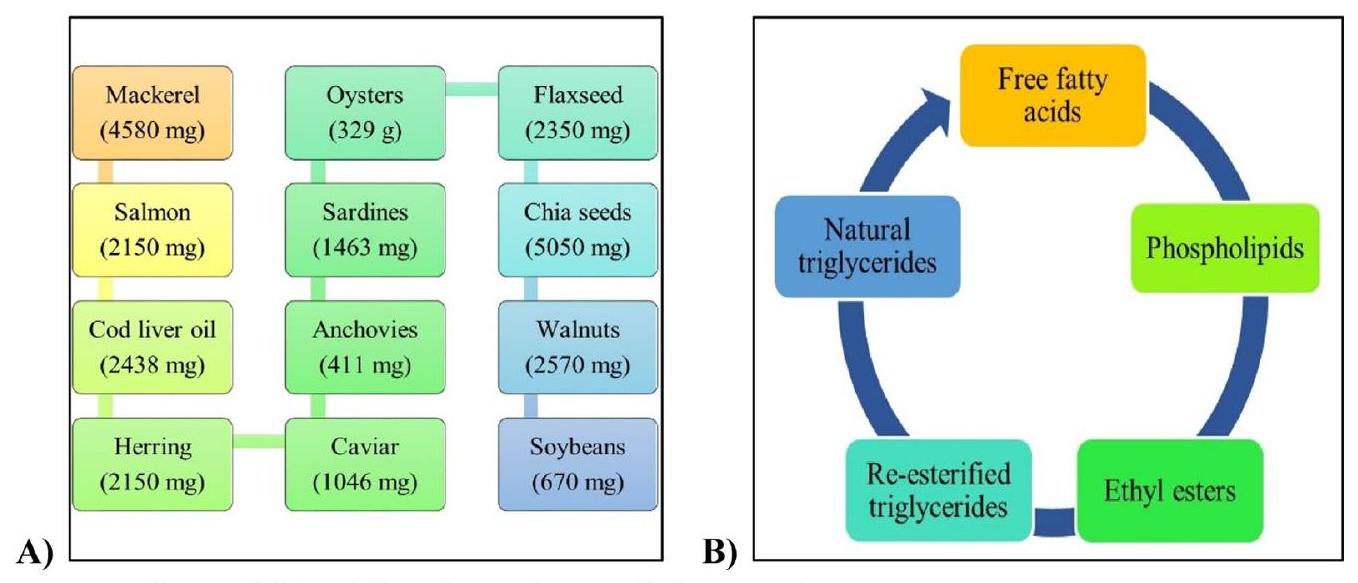
- Ms FH. 12 foods that are very high in omega- 3 [Internet]. Healthline (2023) Available from: https://www.healthline.com/nutrition/12-omega-3-rich-foods#1.-Mackerel-(4,580-mg-per-serving)
- Cunningham E (2012) Are krill oil supplements a better source of
fatty acids than fish oil supplements. J Acad Nutr Diet 112:344. https://doi.org/ 10.1016/j.jand.2011.12.016 - Calder PC (2013) Omega-3 polyunsaturated fatty acids and inflammatory processes: nutrition or pharmacology. Br J Clin Pharmacol 75:645-662. https://doi.org/10.1111/j.1365-2125.2012.04374.x
- Backes J, Anzalone D, Hilleman D, Catini J (2016) The clinical relevance of omega-3 fatty acids in the management of hypertriglyceridemia. Lipids Health Dis 15:118. https://doi.org/10.1186/s12944-016-0286-4
18 Noreen EE, Sass MJ, Crowe ML, Pabon VA, Brandauer J, Averill LK (2010) Effects of supplemental fish oil on resting metabolic rate, body composition, and salivary cortisol in healthy adults. J Int Soc Sports Nutr. https:// doi.org/10.1186/1550-2783-7-31 - Bays HE, Tighe AP, Sadovsky R, Davidson MH (2008) Prescription omega-3 fatty acids and their lipid effects: physiologic mechanisms of action and clinical implications. Expert Rev Cardiovasc Ther 6:391-409. https://doi. org/10.1586/14779072.6.3.391
- Logan SL, Spriet LL (2015) Omega-3 fatty acid supplementation for 12 weeks increases resting and exercise metabolic rate in healthy community-dwelling older females. PLOS ONE 10:e0144828. https://doi. org/10.1371/journal.pone. 0144828
- Seo T, Blaner WS, Deckelbaum RJ (2005) Omega-3 fatty acids: molecular approaches to optimal biological outcomes. Curr Opin Lipidol 16:11-18. https://doi.org/10.1097/00041433-200502000-00004
- Kota B, Huang T, Roufogalis B (2005) An overview on biological mechanisms of PPARs. Pharmacol Res 51:85-94. https://doi.org/10.1016/j.phrs. 2004.07.012
- Nabavi SF, Bilotto S, Russo GL, Orhan IE, Habtemariam S, Daglia M, Devi KP, Loizzo MR, Tundis R, Nabavi SM (2015) Omega-3 polyunsaturated fatty acids and cancer: lessons learned from clinical trials. Cancer Metastasis Rev 34:359-380. https://doi.org/10.1007/s10555-015-9572-2
- Ishihara T, Yoshida M, Arita M (2019) Omega-3 fatty acid-derived mediators that control inflammation and tissue homeostasis. Int Immunol 31:559-567. https://doi.org/10.1093/intimm/dxz001
- Svensson RU et al (2016) Inhibition of acetyl-CoA carboxylase suppresses fatty acid synthesis and tumour growth of non-small-cell lung cancer in preclinical models. Nat Med 22:1108-1119. https://doi.org/10.1038/nm. 4181
- Chew EY, Clemons TE, Agrón E, Launer LJ, Grodstein F, Bernstein PS (2015) Effect of omega-3 fatty acids, lutein/zeaxanthin, or other nutrient supplementation on cognitive function. JAMA 314:791. https://doi.org/10.1001/ jama.2015.9677
- Lim W, Gammack J, Van Niekerk J, Dangour A (2005) Omega 3 fatty acid for the prevention of dementia. In: Lim W-S (ed) Cochrane database syst rev. John Wiley & Sons Ltd, Chichester, UK. https://doi.org/10.1002/14651 858.CD005379
- Tully AM, Roche HM, Doyle R, Fallon C, Bruce I, Lawlor B, Coakley D, Gibney MJ (2003) Low serum cholesteryl ester-docosahexaenoic acid levels in Alzheimer’s disease: a case-control study. Br J Nutr 89:483-489. https:// doi.org/10.1079/BJN2002804
- SanGiovanni JP, Chew EY (2005) The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog Retin Eye Res 24:87-138. https://doi.org/10.1016/j.preteyeres.2004.06.002
- Sakamoto A, Saotome M, Iguchi K, Maekawa Y (2019) Marine-derived omega-3 polyunsaturated fatty acids and heart failure: current understanding for basic to clinical relevance. Int J Mol Sci 20:4025. https://doi. org/10.3390/ijms20164025
- Nodari S, Metra M, Milesi G, Manerba A, Cesana BM, Gheorghiade M, Dei Cas
(2009) The role of n-3 PUFAs in preventing the arrhythmic risk in patients with idiopathic dilated cardiomyopathy. Cardiovasc Drugs Ther 23:5-15. https://doi.org/10.1007/s10557-008-6142-7
32 London B, Albert C, Anderson ME et al (2007) Omega-3 fatty acids and cardiac arrhythmias: prior studies and recommendations for future research. Circulation. https://doi.org/10.1161/CIRCULATIONAHA. 107. 712984 - Albert CM (1998) Fish consumption and risk of sudden cardiac death. JAMA 279:23. https://doi.org/10.1001/jama.279.1.23
- AbuMweis S, Jew S, Tayyem R, Agraib L (2018) Eicosapentaenoic acid and docosahexaenoic acid containing supplements modulate risk factors for cardiovascular disease: a meta-analysis of randomised placebo-control human clinical trials. J Hum Nutr Diet 31:67-84. https://doi.org/10.1111/ jhn. 12493
- Zhang AC, Singh S, Craig JP, Downie LE (2020) Omega-3 fatty acids and eye health: opinions and self-reported practice behaviors of optometrists in Australia and New Zealand. Nutrients 12:1179. https://doi.org/10.3390/ nu12041179
- Bhargava R, Pandey K, Ranjan S, Mehta B, Malik A (2023) Omega-3 fatty acids supplements for dry eye-Are they effective or ineffective. Indian J Ophthalmol 71:1619-1625. https://doi.org/10.4103/IJO.IJO_2789_22
- McDaniel JC, Rausch J, Tan A (2020) Impact of omega-3 fatty acid oral therapy on healing of chronic venous leg ulcers in older adults: Study protocol for a randomized controlled single-center trial. Trials 21:93. https://doi.org/10.1186/s13063-019-3970-7
39. Silva RV, Oliveira JT, Santos BLR, Dias FC, Martinez AMB, Lima CKF, Miranda ALP (2017) Long-chain omega-3 fatty acids supplementation accelerates nerve regeneration and prevents neuropathic pain behavior in mice. Front Pharmacol. https://doi.org/10.3389/fphar.2017.00723
40. Unda SR, Villegas EA, Toledo ME, AsisOnell G, Laino CH (2020) Beneficial effects of fish oil enriched in omega-3 fatty acids on the development and maintenance of neuropathic pain. J Pharm Pharmacol 72:437-447. https://doi.org/10.1111/jphp. 13213
41. Galan Arriero I, Serrano-Munoz D, Gómez-Soriano J, Goicoechea C, Taylor J, Velasco A (2017) The role of omega-3 and omega-9 fatty acids for the treatment of neuropathic pain after neurotrauma. Biochim et Biophys Acta (BBA) – Biomembranes 1859:1629-1635. https://doi.org/10.1016/j. bbamem.2017.05.003
42. NaPier Z, Kanim LEA, Arabi Y, Salehi K, Sears B, Perry M et al (2019) Omega-3 fatty acid supplementation reduces intervertebral disc degeneration. Med Sci Monit 25:9531-9537. https://doi.org/10.12659/MSM. 918649
43. Chong EW-T (2008) Dietary
44. Ishak WMW, Katas H, Yuen NP, Abdullah MA, Zulfakar MH (2019) Topical application of omega-3-, omega-6-, and omega-9-rich oil emulsions for cutaneous wound healing in rats. Drug Deliv Transl Res 9:418-433. https://doi.org/10.1007/s13346-018-0522-8
45. Garcia Lopez S, Villanueva Arriaga RE, Nájera Medina O, Rodríguez López CP et al (2016) one month of omega-3 fatty acid supplementation improves lipid profiles, glucose levels and blood pressure in overweight schoolchildren with metabolic syndrome. J Pediatr Endocrinol Metab. https://doi.org/10.1515/jpem-2015-0324
46 Bernardi JR, Escobar RS, Ferreira CF, Silveira PP (2012) Fetal and neonatal levels of omega-3: effects on neurodevelopment, nutrition, and growth. Sci World J. https://doi.org/10.1100/2012/202473
47. Nevins JEH, Donovan SM, Snetselaar L, Dewey KG, Novotny R (2021) Omega-3 fatty acid dietary supplements consumed during pregnancy and lactation and child neurodevelopment: a systematic review. J Nutr 151:3483-3494. https://doi.org/10.1093/jn/nxab238
48 Shen SC, Gong C, Jin KQ, Zhou L, Xiao Y, Ma L (2022) Omega-3 fatty acid supplementation and coronary heart disease risks: a meta-analysis of randomized controlled clinical trials. Front Nutr. https://doi.org/10.3389/ fnut.2022.809311
49 Dong Y, Zhu H, Chen L, Huang Y, Christen W, Cook NR, Copeland T, Mora S, Buring JE, Lee IM, Costenbader KH, Manson JAE (2022) Effects of vitamin D3 and marine omega-3 fatty acids supplementation on biomarkers of systemic inflammation: 4-year findings from the VITAL randomized trial. Nutrients. https://doi.org/10.3390/nu14245307
50. Arab-Tehrany E, Jacquot M, Gaiani C, Imran M, Desobry S, Linder M (2012) Beneficial effects and oxidative stability of omega-3 long-chain polyunsaturated fatty acids. Trends Food Sci Technol 25:24-33. https://doi.org/ 10.1016/j.tifs.2011.12.002
51. Jensen HA, Sørensen A-DM, Hansen LT, Bøknæs N, Mejlholm O, Jacobsen C (2023) Effect of artificial light on the lipid oxidation of whole, cooked Northern shrimp (Pandalus borealis) during frozen storage. Food Chemistry Advances 3:100369. https://doi.org/10.1016/j.focha.2023.100369
52. Bremmell KE, Briskey D, Meola TR, Mallard A, Prestidge CA, Rao A (2020) A self-emulsifying omega-3 ethyl ester formulation (AquaCelle) significantly improves eicosapentaenoic and docosahexaenoic acid bioavailability in healthy adults. Eur J Nutr 59:2729-2737. https://doi.org/10.1007/ s00394-019-02118-x
53. Alhakamy NA, Aldawsari HM, Hosny KM, Ahmad J, Akhter S, Kammoun AK, Alghaith AF, Asfour HZ, Al-Rabia MW, Md S (2020) Formulation design and pharmacokinetic evaluation of docosahexaenoic acid containing self-nanoemulsifying drug delivery system for oral administration. Nanomater Nanotechnol 10:184798042095098. https://doi.org/10.1177/18479 80420950988
54. Castejon N, Luna P, Senorans FJ (2021) Microencapsulation by spray drying of omega-3 lipids extracted from oilseeds and microalgae: effect on polyunsaturated fatty acid composition. LWT 148:111789. https://doi.org/ 10.1016/j.lwt.2021.111789
55. Hamed S, Elshafei K, El-Sayed H, Abo-Elwafa G, Afifi S, Zahran H (2020) Formulation of multi-functional omega-3 oil rich microcapsules by spray drying methodology. Egypt J Chem. https://doi.org/10.21608/ejchem. 2020.43946.2891
56. Jagtap AA, Badhe YS, Hegde MV, Zanwar AA (2021) Development and characterization of stabilized omega-3 fatty acid and micronutrient emulsion formulation for food fortification. J Food Sci Technol 58:996-1004. https://doi.org/10.1007/s13197-020-04614-z
57. Sreedhar R, Kumar VS, Bhaskaran Pillai AK, Mangalathillam S (2019) Omega-3 fatty acid based nanolipid formulation of atorvastatin for treating hyperlipidemia. Adv Pharm Bull 9:271-280. https://doi.org/10.15171/ apb.2019.031
58. Oklahoma State University. Foods, health and omega-3 oils [Internet] (2017) Available from: https://extension.okstate.edu/fact-sheets/foods-health-and-omega-3-oils.html
59. GRAS Notice 000732: Docosahexaenoic acid oil produced in Schizochytrium sp. | FDA [Internet]. Available from: https://www.fda.gov/ media/112824
60. Harvard Health (2020) FDA approves fish oil-based drug for heart attack and stroke prevention [Internet]. Harvard Health. Available from: https:// www.health.harvard.edu/heart-health/fda-approves-fish-oil-based-drug-for-heart-attack-and-stroke-prevention
61. Grebow J (2020) FDA approves three more omega- 3 drugs. Is this good or bad news for supplements? Nutr Outlook [Internet]. 2020 Nov 15. Available from: https://www.nutritionaloutlook.com/view/fda-approves-three-more-omega-3-drugs-good-or-bad-news-supplements
62. Wachira JK, Larson MK, Harris WS (2014) n-3 Fatty acids affect haemostasis but do not increase the risk of bleeding: clinical observations and mechanistic insights. Br J Nutr 111:1652-1662. https://doi.org/10.1017/ S000711451300425X
ملاحظة الناشر
- *المراسلات:
راجاشري س. ماساريدي
راجاشريمساريدي@كليفرم.edu
1 قسم الصيدلة، كلية كليه الصيدلة، كاهير
جامعة، أكاديمية كيه إل إي للتعليم العالي والبحث، بيلغافي، كارناتاكا 590001، الهند
DOI: https://doi.org/10.1186/s43094-024-00667-5
Publication Date: 2024-07-29
Omega-3 fatty acids: a comprehensive scientific review of their sources, functions and health benefits
Abstract
Background In recent years, public awareness of healthy diets has significantly increased, leading to a rise in the consumption of nutritional supplements. Among these, omega-3 fatty acids have become particularly popular.
Background
(1) An increase in energy intake in comparison to energy expenditure.
(2) An increased intake of dietary saturated fat, omega-6 fatty acids and trans fatty acids than that of omega-3 fatty acids.
(3) Diet with less content of complex carbohydrates and fiber.
(4) Diet with more proportions of cereal grains than of fruits and vegetables.
(5) Reduced intake of protein, antioxidants and calcium.
The initial evidence of the health benefits of omega-3 fatty acids, namely eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), came from Greenland Eskimos, who consumed a diet heavy in fish and had low rates of multiple sclerosis, asthma, type I diabetes mellitus and coronary heart disease. Numerous studies have shown that omega- 3 fats are necessary for the body to synthesize cell membranes in the body and their impact on the function of cell receptors in these membranes. They function as the basic component of hormones that regulate clotting, inflammation, and the contraction and relaxation of arterial walls. Moreover, they can attach to cell receptors that regulate genetic activity. These benefits include the prevention of heart disease and stroke, the management of lupus, eczema, rheumatoid arthritis and the function of omega- 3 fats as preventive agents against cancer and other illnesses [5-7].
High polyunsaturated fatty acids (PUFAs) include alpha-linolenic acid (ALA), docosahexaenoic acid (DHA), eicosapentaenoic acid (EPA) and

Main text
Omega-3 fatty acids types
Sources of omega-3

Synthesis of omega-3
The hormonal balance, end-product feedback inhibition and nutritional state influence the activities of the
delta-6 and delta-5 desaturases. To convert EPA to DHA (22:6n-3), two carbons must be added to EPA to form DHA (
Therapeutic health benefits of omega-3


Mechanism of omega-3 fatty acids
Effects of omega-3 supplements on health outcomes
| Sr. no | Disease | Mechanism of action | Receptors involved | Benefits offered | References |
| 1 | Cardiovascular disease | Altering the mitochondrial membrane phospholipid composition | G protein-coupled receptors (GPRs), peroxisome proliferator-activated receptors (PPARs) | Reduces mitochondrial oxygen consumption, lowers blood pressure, improves blood vessel function | [30-34] |
| 2 | Ophthalmological disease | Altering the characteristics of the lipid phase, thickness, fluidity and permeability. Rhodopsin regeneration and phototransduction | GPRs and PPARs directly incorporated into retinal membranes | By regulating ocular surface inflammation and enhancing tear-lipid profiles, DHA mitigates the symptoms and indicators of DES | [29, 35-37] |
| 3 | Hyperalgesia | Omega-3 s suppress the expression of proinflammatory enzymes and cytokines. Ion channel modulation mechanism | PGE2 receptor, TRPV1 and TRPM8 ion channels | Reduced inflammation and decreases abnormal sensitivity to pain | [38-40] |
| 4 | Neurological disease | Reduces inflammatory cytokines | TNF-a, Interleukin-1 and interleukin-6 | Modulation of central
|
[39-41] |
| 5 | Anti-inflammatory | DHA inhibited leucocyte-endothelial cell adhesive contacts, adhesion molecule expression and the generation of classical inflammatory cytokines | PGE2 receptor, TNF-a, Interleukin and COX-2 | Decreases inflammation and pain in joints and reduces swelling | [16] |
| 6 | Intervertebral disc degeneration | Omega 3 will reduce the inflammatory cytokines and interact with receptors on disc cells. Synthesis, cartilage breakdown and apoptosis | PGE2 receptor, TNF-a, Interleukin | Reduces inflammation and degeneration also promoting disc health and tissue repair | [42,43] |
| 7 | Wound healing | Modulating inflammation. Enhancing angiogenesis. Promoting collagen synthesis and organization | Inflammatory cytokines like IL-6 and TNF-a. IL-10. Proliferation of keratinocytes. G-proteincoupled receptors (GPCRs) | Promoting a faster, more efficient healing process. Improved blood supply accelerates healing | [37,44] |
| 8 | Metabolic syndrome | Lowering triglycerides. Improving insulin sensitivity. Reducing inflammation. Modulating blood pressure | Peroxisome proliferator-activated receptors (PPARs). GPR120 and GPR40 | Reduces triglyceride levels in the blood. Increases cells’ response toward insulin, leading to better blood sugar control | [45] |
| 9 | Neurodevelopment | Membrane building blocks Neurostimulation. Neuroinflammation modulation. Gene expression regulation | G protein-coupled receptors (GPCRs). Retinoic acid receptors (RARs). Peroxisome proliferatoractivated receptors (PPARs) | DHA intake ensures proper neuronal structure and function, facilitating signal transmission and information processing | [46,47] |
Stability problem
Oxidation of omega-3 long-chain PUFA occurs in three
phases
Propagation phase
- Oxygen attack The lipid radical reacts with oxygen to form a peroxyl radical (LOO).
- Chain reaction The peroxyl radical abstracts a hydrogen atom from another lipid molecule, generating another lipid radical and propagating the chain reaction.
- Hydroperoxide formation The abstracted hydrogen atom combines with oxygen to form a hydroperoxide (LOOH), the primary oxidation product.
Termination phase
- Radical scavenging Two radicals can combine, terminating the chain reaction. Alternatively, antioxidants can scavenge radicals, preventing further propagation.
Secondary reactions
Approaches to enhance the stability of omega-3 oil Encapsulation
Stabilization of foods containing omega-3 LC-PUFAs through modified atmospheric packaging
Applications of omega-3 oil in formulation and development Bioavailability challenges

Advanced delivery systems
Self-nanoemulsifying drug delivery systems (SNEDDS) to enhance DHA bioavailability were investigated by Alhakamy Na et al. to overcome the limit imposed due to its poor solubility. The optimum droplet size of 111.5 nm , and zeta potential – 23.53 mV enhanced absorption and stability of the formulation. SNEDDS administration resulted in a 3.32 -fold increase in DHA plasma levels in comparison to that of the aqueous dispersion, highlighting the efficacy of this delivery approach [53].
Microencapsulation techniques can prevent oxidation of omega-3 fatty acids, increase their shelf life and mask unpleasant flavors. It also provides an effective method for stabilizing and improving the administration of bioactive ingredients. Spray drying and complex coacervation processes are the most popular commercial approaches for microencapsulation. A study by Natalia Castejon et al. examined the effects of the microencapsulation process on the composition of fatty acids and reported that microencapsulated omega-3 lipids with particle sizes ranging from 1.5 to
FDA approval for safe use of omega 3 oils
Over the last 20 years, the FDA and other international regulatory bodies have assessed some DHA or DHA-rich oil sources for the possible inclusion of DHA in foods intended for human consumption. GRN 137 is one of the pertinent US GRAS notifications (FDA, 2008). Every GRAS notification included details and data from clinical studies to substantiate the suggested DHA constituents’ safety for use in food intended for human consumption.
The FDA has approved the use of certain qualified health claims on the ability of foods and dietary supplements containing the omega-3 fatty acids, EPA and DHA, to lower blood pressure and lower the risk of hypertension and coronary heart disease. The ruling was issued in response to a petition that was brought in 2014 by the Global Organization for EPA and DHA Omega-3 (GOED) [59].
To lower the incidence of cardiovascular events in some patients with or at high risk for cardiovascular disease (CVD), the FDA authorized a new use for icosapent ethyl (Vascepa), a medication that is a highly refined version of eicosapentaenoic acid (EPA), an omega-3 fatty acid found in fish. The EPA/DHA brand-name Epanova from AstraZeneca and two generic versions, which all target hypertriglyceridemia (triglyceride levels of
owned by Lindsay Goldberg LLC and Aker BioMarine ASA [60].
Omega-3 interactions with medications
Omega-3 deficiency
Half-life
Omega-3 intakes
acids (approximately 40 mg for children and teenagers and around 90 mg for adults) [7].
Conclusion
Abbreviations
| EPA | Eicosapentaenoic acid |
| DHA | Docosahexaenoic acid |
| ALA | Alpha-linolenic acid |
| PUFAs | High polyunsaturated fatty acids |
| OM3FAs | Omega-3 fatty acids |
| LPL | Lipo-protein-lipase |
| PPARs | Peroxisome proliferator-activated receptors |
| CAT 1 | Carnitine acetyl transferase 1 |
| GPRs | G Protein-coupled receptors |
Acknowledgements
Author contributions
Funding
Availability of data and materials
Declarations
Ethics approval and consent to participate
Competing interests
Published online: 29 July 2024
References
- Theinel MH, Nucci MP, Alves AH, Dias OFM, Mamani JB, Garrigós MM, Oliveira FA, Rego GNA, Valle NME, Cianciarullo G, Gamarra LF (2023) The effects of omega-3 polyunsaturated fatty acids on breast cancer as a preventive measure or as an adjunct to conventional treatments. Nutrients 15:1310. https://doi.org/10.3390/nu15061310
- Sharifi E, Yazdani Z, Najafi M, Hosseini-khah Z, Jafarpour A, Rafiei A (2022) The combined effect of fish oil containing omega-3 fatty acids and Lactobacillus plantarum on colorectal cancer. Food Sci Nutr 10:4411-4418. https://doi.org/10.1002/fsn3.3037
- Liang P, Henning SM, Guan J, Grogan T, Elashoff D, Cohen P, Aronson WJ (2020) Effect of dietary omega-3 fatty acids on castrate-resistant prostate cancer and tumor-associated macrophages. Prostate Cancer Prostatic Dis 23:127-135. https://doi.org/10.1038/s41391-019-0168-8
- Simopoulos AP (2008) The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp Biol Med 233:674-688. https://doi.org/10.3181/0711-MR-311
- Ratanabanangkoon P, Guzman H, Almarsson O, Berkovitz D, Tokarcyzk S, Straughn AB, Chen H (2008) A high-throughput approach towards a novel formulation of fenofibrate in omega-3 oil. Eur J Pharm Sci 33:351-360. https://doi.org/10.1016/j.ejps.2008.01.005
- Elagizi A, Lavie CJ, O’Keefe E, Marshall K, O’Keefe JH, Milani RV (2021) An update on omega-3 polyunsaturated fatty acids and cardiovascular health. Nutrients 13:204. https://doi.org/10.3390/nu13010204
- The Nutrition Source. Omega-3 fatty acids: an essential contribution [Internet]. 2023 [cited 2024 Jun 27]. Available from: https://www.hsph. harvard.edu/nutritionsource/what-should-you-eat/fats-and-cholesterol/ types-of-fat/omega-3-fats/
- Djuricic I, Calder PC (2021) Beneficial outcomes of omega-6 and omega-3 polyunsaturated fatty acids on human health: an update for 2021. Nutrients 13:2421. https://doi.org/10.3390/nu13072421
- Wikipedia contributors. Omega-3 fatty acid [Internet]. Wikipedia. 2024 [cited 2024 Jun 27]. Available from: https://en.wikipedia.org/wiki/Omega3_fatty_acid
- Tur JA, Bibiloni MM, Sureda A, Pons A (2012) Dietary sources of omega 3 fatty acids: public health risks and benefits. Br J Nutr 107:S23-S52. https:// doi.org/10.1017/S0007114512001456
- Office of Dietary Supplements. Omega-3 fatty acids [Internet]. [Cited 2024 Jun 27]. Available from: https://ods.od.nih.gov/factsheets/Omega 3FattyAcids-HealthProfessional/
- Miller MR, Nichols PD, Carter CG (2008) Omega 3 Oil sources for use in aquaculture-alternatives to the unsustainable harvest of wild fish. Nutr Res Rev 21:85-96. https://doi.org/10.1017/S0954422408102414
- Davidson MH, Kling D, Maki KC (2011) Novel developments in omega-3 fatty acid-based strategies. Curr Opin Lipidol 22:437-444. https://doi.org/ 10.1097/MOL.0b013e32834bd642
- Ms FH. 12 foods that are very high in omega- 3 [Internet]. Healthline (2023) Available from: https://www.healthline.com/nutrition/12-omega-3-rich-foods#1.-Mackerel-(4,580-mg-per-serving)
- Cunningham E (2012) Are krill oil supplements a better source of
fatty acids than fish oil supplements. J Acad Nutr Diet 112:344. https://doi.org/ 10.1016/j.jand.2011.12.016 - Calder PC (2013) Omega-3 polyunsaturated fatty acids and inflammatory processes: nutrition or pharmacology. Br J Clin Pharmacol 75:645-662. https://doi.org/10.1111/j.1365-2125.2012.04374.x
- Backes J, Anzalone D, Hilleman D, Catini J (2016) The clinical relevance of omega-3 fatty acids in the management of hypertriglyceridemia. Lipids Health Dis 15:118. https://doi.org/10.1186/s12944-016-0286-4
18 Noreen EE, Sass MJ, Crowe ML, Pabon VA, Brandauer J, Averill LK (2010) Effects of supplemental fish oil on resting metabolic rate, body composition, and salivary cortisol in healthy adults. J Int Soc Sports Nutr. https:// doi.org/10.1186/1550-2783-7-31 - Bays HE, Tighe AP, Sadovsky R, Davidson MH (2008) Prescription omega-3 fatty acids and their lipid effects: physiologic mechanisms of action and clinical implications. Expert Rev Cardiovasc Ther 6:391-409. https://doi. org/10.1586/14779072.6.3.391
- Logan SL, Spriet LL (2015) Omega-3 fatty acid supplementation for 12 weeks increases resting and exercise metabolic rate in healthy community-dwelling older females. PLOS ONE 10:e0144828. https://doi. org/10.1371/journal.pone. 0144828
- Seo T, Blaner WS, Deckelbaum RJ (2005) Omega-3 fatty acids: molecular approaches to optimal biological outcomes. Curr Opin Lipidol 16:11-18. https://doi.org/10.1097/00041433-200502000-00004
- Kota B, Huang T, Roufogalis B (2005) An overview on biological mechanisms of PPARs. Pharmacol Res 51:85-94. https://doi.org/10.1016/j.phrs. 2004.07.012
- Nabavi SF, Bilotto S, Russo GL, Orhan IE, Habtemariam S, Daglia M, Devi KP, Loizzo MR, Tundis R, Nabavi SM (2015) Omega-3 polyunsaturated fatty acids and cancer: lessons learned from clinical trials. Cancer Metastasis Rev 34:359-380. https://doi.org/10.1007/s10555-015-9572-2
- Ishihara T, Yoshida M, Arita M (2019) Omega-3 fatty acid-derived mediators that control inflammation and tissue homeostasis. Int Immunol 31:559-567. https://doi.org/10.1093/intimm/dxz001
- Svensson RU et al (2016) Inhibition of acetyl-CoA carboxylase suppresses fatty acid synthesis and tumour growth of non-small-cell lung cancer in preclinical models. Nat Med 22:1108-1119. https://doi.org/10.1038/nm. 4181
- Chew EY, Clemons TE, Agrón E, Launer LJ, Grodstein F, Bernstein PS (2015) Effect of omega-3 fatty acids, lutein/zeaxanthin, or other nutrient supplementation on cognitive function. JAMA 314:791. https://doi.org/10.1001/ jama.2015.9677
- Lim W, Gammack J, Van Niekerk J, Dangour A (2005) Omega 3 fatty acid for the prevention of dementia. In: Lim W-S (ed) Cochrane database syst rev. John Wiley & Sons Ltd, Chichester, UK. https://doi.org/10.1002/14651 858.CD005379
- Tully AM, Roche HM, Doyle R, Fallon C, Bruce I, Lawlor B, Coakley D, Gibney MJ (2003) Low serum cholesteryl ester-docosahexaenoic acid levels in Alzheimer’s disease: a case-control study. Br J Nutr 89:483-489. https:// doi.org/10.1079/BJN2002804
- SanGiovanni JP, Chew EY (2005) The role of omega-3 long-chain polyunsaturated fatty acids in health and disease of the retina. Prog Retin Eye Res 24:87-138. https://doi.org/10.1016/j.preteyeres.2004.06.002
- Sakamoto A, Saotome M, Iguchi K, Maekawa Y (2019) Marine-derived omega-3 polyunsaturated fatty acids and heart failure: current understanding for basic to clinical relevance. Int J Mol Sci 20:4025. https://doi. org/10.3390/ijms20164025
- Nodari S, Metra M, Milesi G, Manerba A, Cesana BM, Gheorghiade M, Dei Cas
(2009) The role of n-3 PUFAs in preventing the arrhythmic risk in patients with idiopathic dilated cardiomyopathy. Cardiovasc Drugs Ther 23:5-15. https://doi.org/10.1007/s10557-008-6142-7
32 London B, Albert C, Anderson ME et al (2007) Omega-3 fatty acids and cardiac arrhythmias: prior studies and recommendations for future research. Circulation. https://doi.org/10.1161/CIRCULATIONAHA. 107. 712984 - Albert CM (1998) Fish consumption and risk of sudden cardiac death. JAMA 279:23. https://doi.org/10.1001/jama.279.1.23
- AbuMweis S, Jew S, Tayyem R, Agraib L (2018) Eicosapentaenoic acid and docosahexaenoic acid containing supplements modulate risk factors for cardiovascular disease: a meta-analysis of randomised placebo-control human clinical trials. J Hum Nutr Diet 31:67-84. https://doi.org/10.1111/ jhn. 12493
- Zhang AC, Singh S, Craig JP, Downie LE (2020) Omega-3 fatty acids and eye health: opinions and self-reported practice behaviors of optometrists in Australia and New Zealand. Nutrients 12:1179. https://doi.org/10.3390/ nu12041179
- Bhargava R, Pandey K, Ranjan S, Mehta B, Malik A (2023) Omega-3 fatty acids supplements for dry eye-Are they effective or ineffective. Indian J Ophthalmol 71:1619-1625. https://doi.org/10.4103/IJO.IJO_2789_22
- McDaniel JC, Rausch J, Tan A (2020) Impact of omega-3 fatty acid oral therapy on healing of chronic venous leg ulcers in older adults: Study protocol for a randomized controlled single-center trial. Trials 21:93. https://doi.org/10.1186/s13063-019-3970-7
39. Silva RV, Oliveira JT, Santos BLR, Dias FC, Martinez AMB, Lima CKF, Miranda ALP (2017) Long-chain omega-3 fatty acids supplementation accelerates nerve regeneration and prevents neuropathic pain behavior in mice. Front Pharmacol. https://doi.org/10.3389/fphar.2017.00723
40. Unda SR, Villegas EA, Toledo ME, AsisOnell G, Laino CH (2020) Beneficial effects of fish oil enriched in omega-3 fatty acids on the development and maintenance of neuropathic pain. J Pharm Pharmacol 72:437-447. https://doi.org/10.1111/jphp. 13213
41. Galan Arriero I, Serrano-Munoz D, Gómez-Soriano J, Goicoechea C, Taylor J, Velasco A (2017) The role of omega-3 and omega-9 fatty acids for the treatment of neuropathic pain after neurotrauma. Biochim et Biophys Acta (BBA) – Biomembranes 1859:1629-1635. https://doi.org/10.1016/j. bbamem.2017.05.003
42. NaPier Z, Kanim LEA, Arabi Y, Salehi K, Sears B, Perry M et al (2019) Omega-3 fatty acid supplementation reduces intervertebral disc degeneration. Med Sci Monit 25:9531-9537. https://doi.org/10.12659/MSM. 918649
43. Chong EW-T (2008) Dietary
44. Ishak WMW, Katas H, Yuen NP, Abdullah MA, Zulfakar MH (2019) Topical application of omega-3-, omega-6-, and omega-9-rich oil emulsions for cutaneous wound healing in rats. Drug Deliv Transl Res 9:418-433. https://doi.org/10.1007/s13346-018-0522-8
45. Garcia Lopez S, Villanueva Arriaga RE, Nájera Medina O, Rodríguez López CP et al (2016) one month of omega-3 fatty acid supplementation improves lipid profiles, glucose levels and blood pressure in overweight schoolchildren with metabolic syndrome. J Pediatr Endocrinol Metab. https://doi.org/10.1515/jpem-2015-0324
46 Bernardi JR, Escobar RS, Ferreira CF, Silveira PP (2012) Fetal and neonatal levels of omega-3: effects on neurodevelopment, nutrition, and growth. Sci World J. https://doi.org/10.1100/2012/202473
47. Nevins JEH, Donovan SM, Snetselaar L, Dewey KG, Novotny R (2021) Omega-3 fatty acid dietary supplements consumed during pregnancy and lactation and child neurodevelopment: a systematic review. J Nutr 151:3483-3494. https://doi.org/10.1093/jn/nxab238
48 Shen SC, Gong C, Jin KQ, Zhou L, Xiao Y, Ma L (2022) Omega-3 fatty acid supplementation and coronary heart disease risks: a meta-analysis of randomized controlled clinical trials. Front Nutr. https://doi.org/10.3389/ fnut.2022.809311
49 Dong Y, Zhu H, Chen L, Huang Y, Christen W, Cook NR, Copeland T, Mora S, Buring JE, Lee IM, Costenbader KH, Manson JAE (2022) Effects of vitamin D3 and marine omega-3 fatty acids supplementation on biomarkers of systemic inflammation: 4-year findings from the VITAL randomized trial. Nutrients. https://doi.org/10.3390/nu14245307
50. Arab-Tehrany E, Jacquot M, Gaiani C, Imran M, Desobry S, Linder M (2012) Beneficial effects and oxidative stability of omega-3 long-chain polyunsaturated fatty acids. Trends Food Sci Technol 25:24-33. https://doi.org/ 10.1016/j.tifs.2011.12.002
51. Jensen HA, Sørensen A-DM, Hansen LT, Bøknæs N, Mejlholm O, Jacobsen C (2023) Effect of artificial light on the lipid oxidation of whole, cooked Northern shrimp (Pandalus borealis) during frozen storage. Food Chemistry Advances 3:100369. https://doi.org/10.1016/j.focha.2023.100369
52. Bremmell KE, Briskey D, Meola TR, Mallard A, Prestidge CA, Rao A (2020) A self-emulsifying omega-3 ethyl ester formulation (AquaCelle) significantly improves eicosapentaenoic and docosahexaenoic acid bioavailability in healthy adults. Eur J Nutr 59:2729-2737. https://doi.org/10.1007/ s00394-019-02118-x
53. Alhakamy NA, Aldawsari HM, Hosny KM, Ahmad J, Akhter S, Kammoun AK, Alghaith AF, Asfour HZ, Al-Rabia MW, Md S (2020) Formulation design and pharmacokinetic evaluation of docosahexaenoic acid containing self-nanoemulsifying drug delivery system for oral administration. Nanomater Nanotechnol 10:184798042095098. https://doi.org/10.1177/18479 80420950988
54. Castejon N, Luna P, Senorans FJ (2021) Microencapsulation by spray drying of omega-3 lipids extracted from oilseeds and microalgae: effect on polyunsaturated fatty acid composition. LWT 148:111789. https://doi.org/ 10.1016/j.lwt.2021.111789
55. Hamed S, Elshafei K, El-Sayed H, Abo-Elwafa G, Afifi S, Zahran H (2020) Formulation of multi-functional omega-3 oil rich microcapsules by spray drying methodology. Egypt J Chem. https://doi.org/10.21608/ejchem. 2020.43946.2891
56. Jagtap AA, Badhe YS, Hegde MV, Zanwar AA (2021) Development and characterization of stabilized omega-3 fatty acid and micronutrient emulsion formulation for food fortification. J Food Sci Technol 58:996-1004. https://doi.org/10.1007/s13197-020-04614-z
57. Sreedhar R, Kumar VS, Bhaskaran Pillai AK, Mangalathillam S (2019) Omega-3 fatty acid based nanolipid formulation of atorvastatin for treating hyperlipidemia. Adv Pharm Bull 9:271-280. https://doi.org/10.15171/ apb.2019.031
58. Oklahoma State University. Foods, health and omega-3 oils [Internet] (2017) Available from: https://extension.okstate.edu/fact-sheets/foods-health-and-omega-3-oils.html
59. GRAS Notice 000732: Docosahexaenoic acid oil produced in Schizochytrium sp. | FDA [Internet]. Available from: https://www.fda.gov/ media/112824
60. Harvard Health (2020) FDA approves fish oil-based drug for heart attack and stroke prevention [Internet]. Harvard Health. Available from: https:// www.health.harvard.edu/heart-health/fda-approves-fish-oil-based-drug-for-heart-attack-and-stroke-prevention
61. Grebow J (2020) FDA approves three more omega- 3 drugs. Is this good or bad news for supplements? Nutr Outlook [Internet]. 2020 Nov 15. Available from: https://www.nutritionaloutlook.com/view/fda-approves-three-more-omega-3-drugs-good-or-bad-news-supplements
62. Wachira JK, Larson MK, Harris WS (2014) n-3 Fatty acids affect haemostasis but do not increase the risk of bleeding: clinical observations and mechanistic insights. Br J Nutr 111:1652-1662. https://doi.org/10.1017/ S000711451300425X
Publisher’s Note
- *Correspondence:
Rajashree S. Masareddy
rajashreemasareddy@klepharm.edu
1 Department of Pharmaceutics, KLE College of Pharmacy, KAHER
University, KLE Academy of Higher Education and Research, Belagavi, Karnataka 590001, India
