DOI: https://doi.org/10.1038/s41420-024-01960-1
PMID: https://pubmed.ncbi.nlm.nih.gov/38678017
تاريخ النشر: 2024-04-27
إيقاع الساعة البيولوجية ينظم وظيفة خلايا المناعة ويشارك في تطور الأورام
الملخص
توجد الإيقاعات البيولوجية في جميع الخلايا تقريبًا وتلعب دورًا حاسمًا في تنظيم العمليات البيولوجية المختلفة. الحفاظ على إيقاع بيولوجي مستقر أمر ضروري للصحة العامة. يمكن أن يؤدي اضطراب هذا الإيقاع إلى تغيير تعبير جينات الساعة وجينات مرتبطة بالسرطان، ويؤثر على العديد من المسارات والعوامل الأيضية، مما يؤثر على وظيفة الجهاز المناعي ويساهم في حدوث الأورام وتقدمها. تهدف هذه الورقة إلى توضيح التأثيرات التنظيمية لـ BMAL1، والساعة، وغيرها من جينات الساعة على خلايا المناعة، وكشف الآلية الجزيئية لمشاركة إيقاع الساعة البيولوجية في تنظيم الورم وبيئته الدقيقة. إن فهمًا أعمق للإيقاعات البيولوجية لديه القدرة على توفير استراتيجيات جديدة لعلاج السرطان وأمراض المناعة الأخرى.
الحقائق
- تؤثر الإيقاعات البيولوجية بشكل عميق على العمليات الفسيولوجية المختلفة في الجسم.
- تؤثر الإيقاعات البيولوجية على نضوج وتوطين ووظيفة خلايا المناعة بطرق متنوعة (بما في ذلك الجينات، وجزيئات الالتصاق، والسيتوكينات، وما إلى ذلك).
- يمكن للجهاز المناعي الاستجابة من خلال التأثير على الإيقاعات البيولوجية.
- تشارك الإيقاعات البيولوجية في تنظيم دورة الخلية.
- تتغير المكونات المتعلقة بالإيقاع البيولوجي في خلايا الورم وبيئتها الدقيقة، وهناك ارتباط وبائي بين خطر الإصابة بالسرطان وتعبير جينات الإيقاع البيولوجي.
أسئلة مفتوحة
- هل يمكن استخدام اعتبارات الإيقاع البيولوجي لتحسين الإدارة السريرية، والتغذية، وتوقيت الجراحة لتحسين نتائج المرضى؟
- هل يمكن دمج الإيقاعات البيولوجية في استخدام الأدوية وتوقيت الجراحة لتحقيق نتائج أفضل؟
- هل يمكننا التنبؤ بخطر الإصابة بالسرطان من خلال الكشف عن حالة التعبير عن جينات الإيقاع البيولوجي بالاشتراك مع علامات أخرى؟
المقدمة
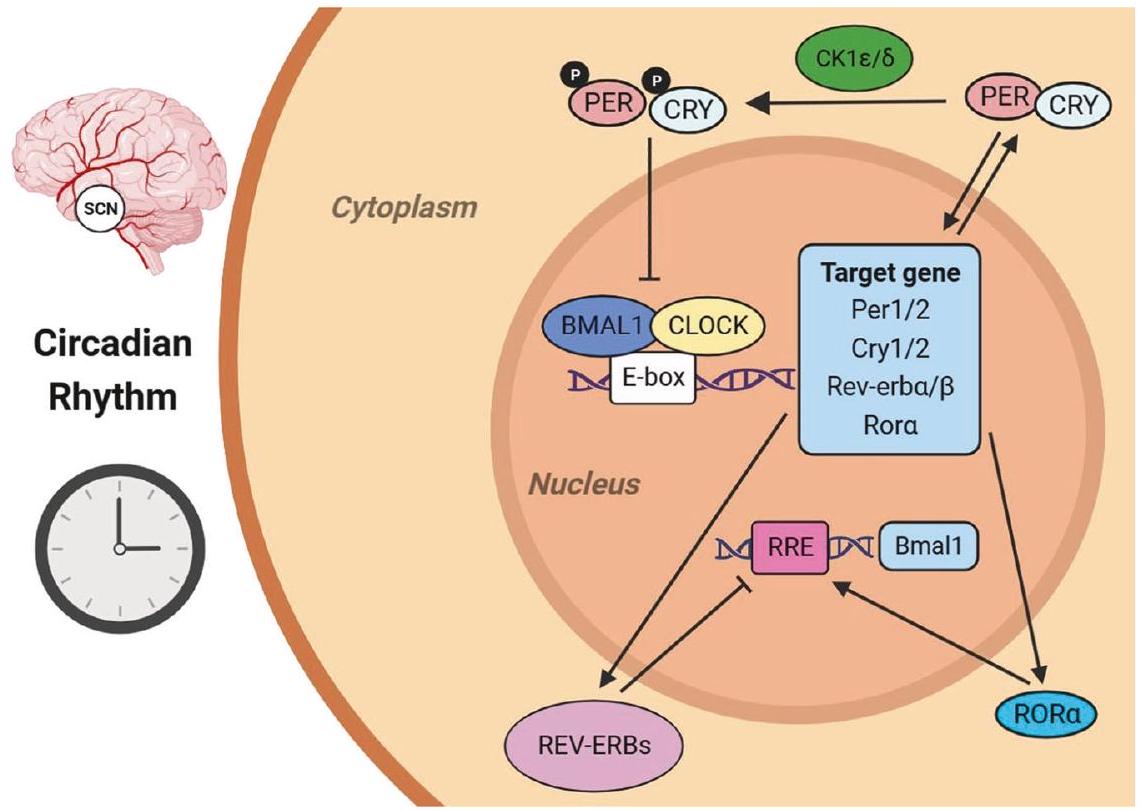
[2,3]. في جوهرها، تشكل الساعة البيولوجية تسلسلًا تنظيميًا تلقائيًا لتعبير وتراكم وتحلل منتجات جينات الساعة، مما يشكل مذبذبًا جزيئيًا مستقلًا [3]. تتحكم الساعة الجزيئية في الحيوانات في تعبير الجينات التي يتم إخراجها في جميع أنحاء الجسم، مما يتحكم في نشاط ووظيفة خلايا وأعضاء مختلفة مع مرور الوقت [4]. لذلك، يمكن أن تؤدي الطفرات في جينات الساعة إلى تغييرات في سلوكيات إيقاعية بيولوجية مختلفة [5].
إعاقة النسخ الذي يتم بوساطة CLOCK/BMAL1 [3]. قد تؤثر الطفرات الجينية التي تغير حالة الفسفرة، أو الحركة، أو تحلل بروتينات PER وCRY على سرعة الساعة [16، 17]. تتعرض بروتينات PER لعدة أحداث فسفرة متتالية لمواقع متعددة بواسطة CK1
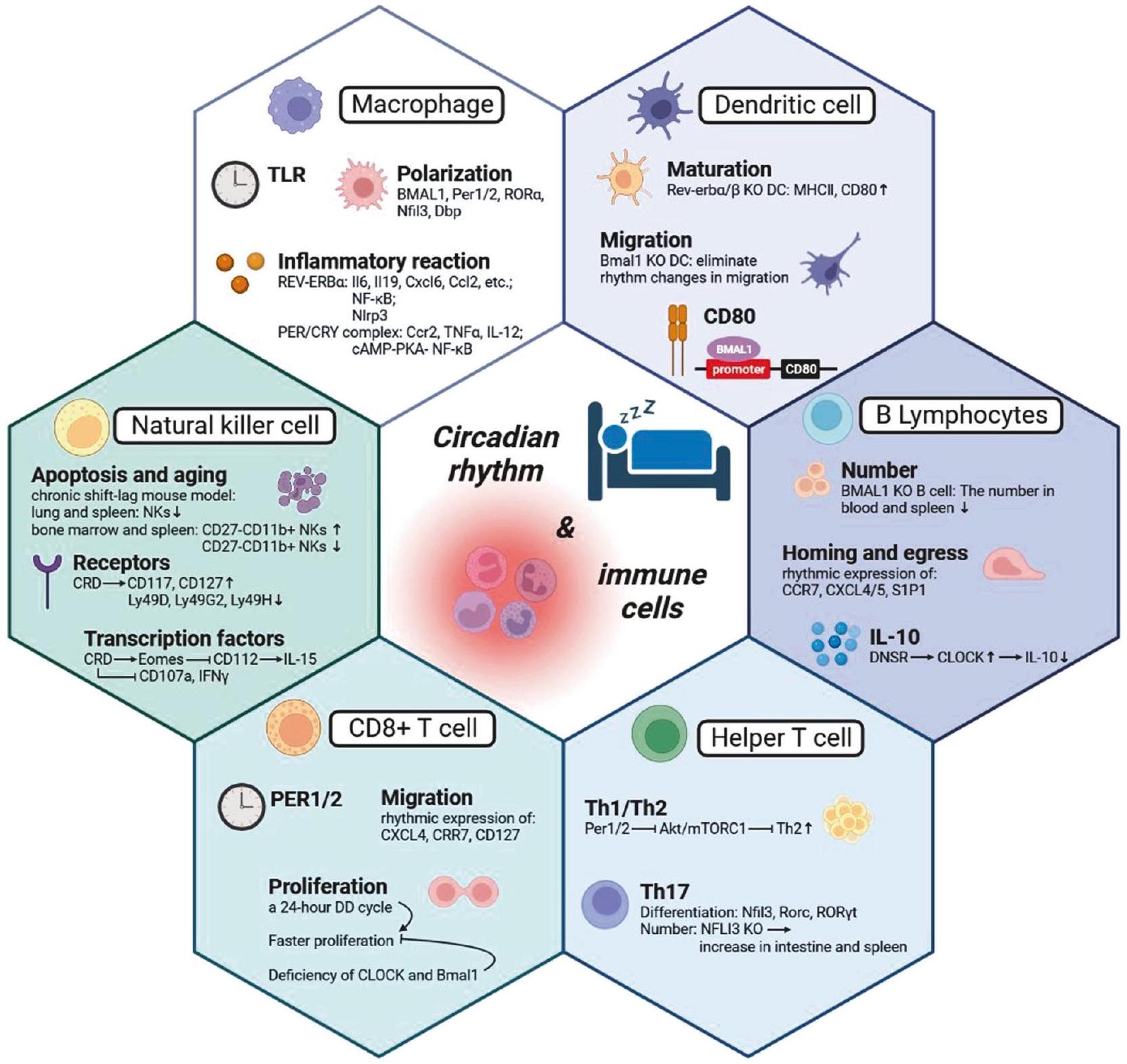
أثر الإيقاعات اليومية على خلايا المناعة: البلعميات
الخلايا الشجرية
الخلايا اللمفاوية ب
العقد استجابةً للدهون الكيميائية السفينغوزين 1-فوسفات [89].
الخلايا اللمفاوية التائية
تحت إيقاعات الساعة البيولوجية، تنظم الإنتاج الإيقاعي للكورتيزول والكيتوكول أمينات هجرة وتوطين خلايا T. كما أن تكاثر خلايا T يتأثر أيضًا بإيقاعات الساعة البيولوجية، حيث يظهر معدل تكاثر أسرع في الليل. تشير هذه النتائج البحثية إلى أن إيقاعات الساعة البيولوجية لها تأثيرات مهمة على نشاط وهجرة وتكاثر خلايا T، وأن اضطراب إيقاعات الساعة البيولوجية قد يؤثر على الاستجابات المناعية، بما في ذلك الاستجابات المناعية للأورام.
خلايا NK
دورة النوم والاستيقاظ تشارك في التأثير على وظيفة الجهاز المناعي وإصلاح تلف الحمض النووي، وتُنظم بواسطة الالتهاب والتغذية الراجعة للسيتوكينات
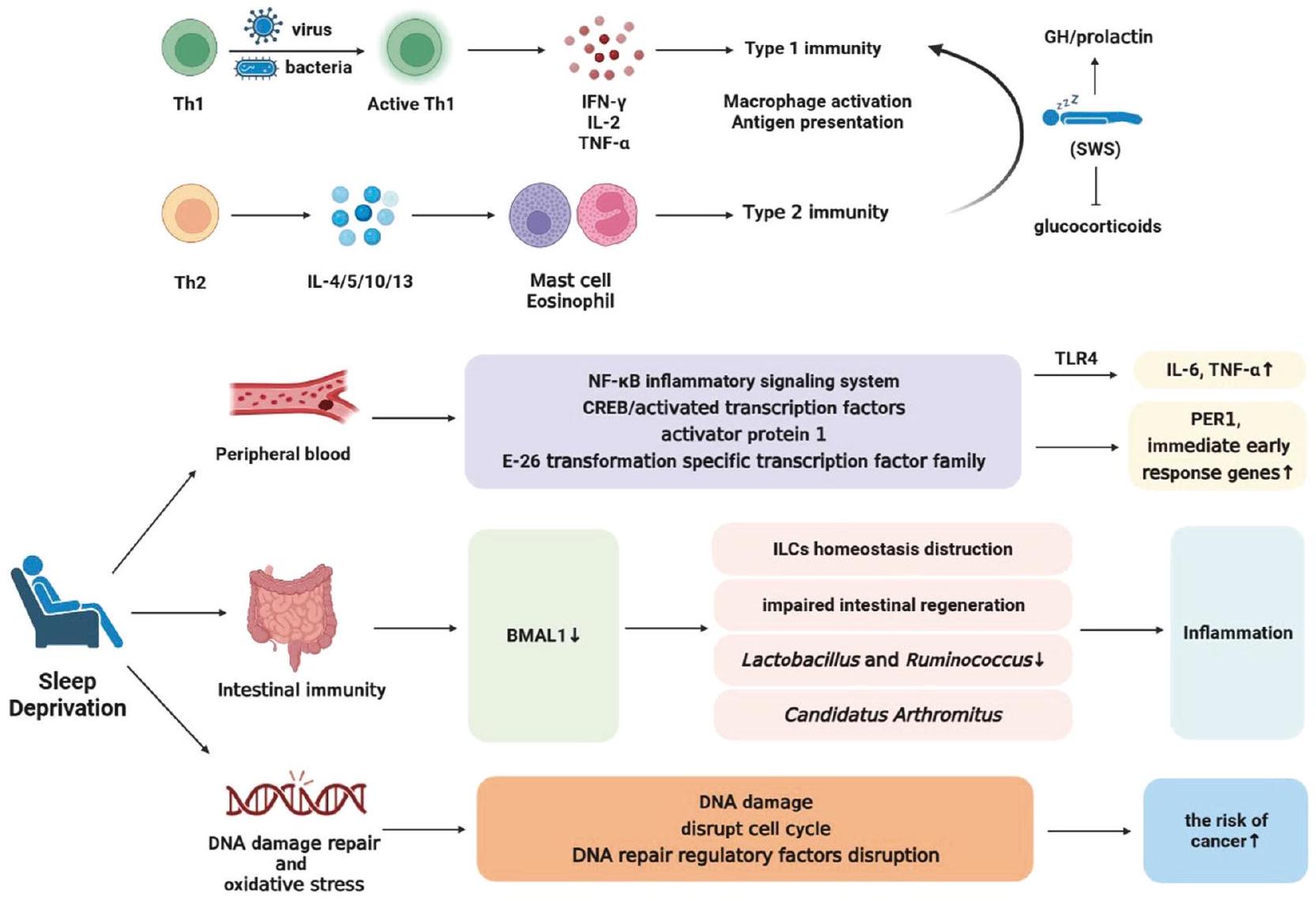
النوم يؤثر على إنتاج السيتوكينات ويشارك في تنظيم الالتهاب
في المختبر إيقاعًا بيولوجيًا، مع ذروة حوالي 03:00 خلال النوم الليلي [116، 117]. يمنع النوم، وخاصة في المراحل المبكرة التي تهيمن عليها النوم ببطء (SWS)، إفراز الجلوكوكورتيكويد ويعزز إفراز هرمون النمو (GH) والبرولاكتين [118-120]. تعمل هذه العوامل معًا على تحويل توازن Th1/Th2 نحو ميزة Th1، مما يعزز الاستجابات المناعية الخلوية ضد مسببات الأمراض داخل الخلايا [121، 122]. أكدت دراسة أخرى أن النوم الليلي يمنع إنتاج خلايا Th2 مقارنة باليقظة، بينما يبقى التأثير على خلايا Th1 غير واضح. وبالتالي، يحفز النوم تحولًا نحو هيمنة Th1 [115]. قد يساهم تثبيط إفراز الكورتيزول وزيادة مستويات الميلاتونين في الليل في هيمنة Th1 [116، 123]. ومع ذلك، يقتصر هذا التأثير على النوم المبكر، وخاصة SWS، ويعكس خلال النوم المتأخر الذي تهيمن عليه حركة العين السريعة (REM)، مع انخفاض نسبة خلايا CD4
متلازمة الجهاز الهضمي [134]، مما يبرز أهمية BMAL1 في تكاثر خلايا الأمعاء. إن الإفراط في التعبير عن CRY1 يثبط الالتهاب الوعائي الناتج عن الحرمان من النوم (SD)، ربما من خلال مسارات NF-kB وأدينوسين أحادي الفوسفات الحلقي/ بروتين كيناز A (cAMP/PKA) في الجسم [135]. يؤثر الحرمان الحاد من النوم (ASD) بشكل شائع على العمال في نوبات العمل، مما يسبب الخمول والتعب المستمر. يساهم ASD في الالتهاب من خلال التأثير على الميكروبيوم المعوي. في الأفراد المسنين الذين يعانون من الأرق، يمكن تنظيم تأثير النشاط البدني على اضطرابات النوم بواسطة المستقلبات الأساسية للميكروبيوم المعوي. يظهر جنس Monoglobus ارتباطًا سلبيًا مع النشاط البدني وزمن بدء النوم، بينما يظهر ارتباطًا إيجابيًا مع النوم.
يؤدي اضطراب إيقاع الساعة البيولوجية إلى انخفاض وفرة بكتيريا معينة (مثل Lactobacillus و Ruminococcus) جنبًا إلى جنب مع تقليل تنظيم BMAL1. قد يساهم زيادة Candidatus Arthromitus في مجموعة SD في التهاب القولون، وعجز وظيفة الحاجز المعوي، والالتهاب الجهازي. وبالتالي، يلعب إيقاع الساعة البيولوجية وتركيب الميكروبيوم أدوارًا حاسمة في الحفاظ على وظيفة الحاجز المعوي. قد يكون للنوم الكافي والتنظيم الذاتي للميكروبيوم تأثيرات وقائية ضد الأمراض ويساعد في استعادة حاجز الظهارة المعوية، مما يحسن الالتهاب.
يؤدي الحرمان من النوم إلى حدوث السرطان من خلال التأثير على إصلاح تلف الحمض النووي والإجهاد التأكسدي
تم تحديد الجينات المعنية في تنشيط نقاط تفتيش دورة الخلية وإصلاح الحمض النووي، بما في ذلك ATR وCDK4 وCDKN1C وTP53 وWEE1 وERCC6 وTP53 وRPA3 وXPA وH2AFX وPARP1 وRAD50. يشير اضطراب إيقاع الساعة البيولوجية للتعبير عن هذه الجينات إلى وجود صلة مباشرة بين آليات إيقاع الساعة البيولوجية الخلوية وخطر الإصابة بالسرطان الناتج عن العمل في النوبات الليلية [139].
يمكن أن توفر الالتهابات والسيتوكينات وخلايا المناعة تغذية راجعة لتنظيم النوم
تتوسط هذه العلاقة. ومع ذلك، لم تجد بعض الدراسات ارتباطًا بين علامات الالتهاب ووقت النوم [211-215]، مما يزيد من تباين نتائج الأبحاث.
أثر إيقاع الساعة البيولوجية على الأورام وبيئتها الدقيقة
يساهم الإيقاع اليومي في تطور الأورام من خلال التأثير على أنواع مختلفة من الخلايا
رئيسيًا.
يوجد ارتباط جزيئي بين دورة الخلية والساعة البيولوجية، وهما معًا متورطان في حدوث السرطان وتطوره.
دورة، مع دليل على تغييرات إيقاع الساعة البيولوجية في وفرة السايتوكينات وتعبير جينات الساعة في مراحل محددة من دورة الخلية. تتحكم الساعة البيولوجية في تعبير CDK والسايتوكينات مثل CDK2 وCDK4 وcyclin D1، بالإضافة إلى مثبطات دورة الخلية بما في ذلك Wee1 وp21 وp27 وp57. ينظم CLOCK-BMAL1 التعبير الإيقاعي لمثبط G2/M WEE1، بينما يتم تنظيم تعبير p21 من خلال التأثيرات المتعارضة لبروتينات ROR وREV-ERB. ينظم NONO، بالتعاون مع بروتين PER، تنظيم إيقاع الساعة البيولوجية لتعبير p16. يرتبط P53 مباشرة بمروج Per2 ويثبط النسخ الذي يتم بوساطة CLOCK-BMAL1.
يتم تنظيم الأيض والعديد من العوامل في بيئة الورم الدقيقة بواسطة الإيقاع البيولوجي
عوامل الإشارة في خلايا الورم. يؤدي حذف بروتينات الساعة Cry1 وCry2 إلى زيادة مستمرة في السيتوكينات المؤيدة للالتهابات عبر إشارات NF-кB وPKA، مما يشير إلى التنظيم المباشر لتعبير السيتوكينات بواسطة الجينات الدائرية [75]. يؤدي حذف الجين الخاص بإيقاع الساعة البيولوجية في الخلايا النخاعية إلى تعطيل إيقاع تعبير الكيموكينات وتجنيد الأنسجة لخلايا المونوسيت Ly6Chi [242]. تتأرجح جزيئات الالتصاق والكيموكينات في الخلايا البطانية في إيقاع دائري، مما يؤثر على التصاق الكريات البيضاء والانفصال الخلوي [273].
| جين الساعة | خلية | آثار | مرجع | |||
| بيمال1
|
البلاعم |
|
[71, 72] | |||
| جيم (النوع الفرعي M2) |
|
[81] | ||||
| خلية بطانية | تم القضاء على فرق الوقت في تعبير الجزيئات التي تعزز الهجرة | [83] | ||||
| ساعة
|
خلية بطانية |
|
[84] | |||
| خلايا CRC |
|
[91] | ||||
| ابكِ
|
خلية بطانية |
|
[85] | |||
| خلايا الورم |
|
[32] | ||||
| ROR
|
|
|
[89] | |||
|
|
خلايا Th17، Treg |
|
[88] | |||
| REV-ERB
|
البلاعم |
|
[75] |
الملخص
جينات الساعة يقدم طرقًا جديدة لعلاج السرطان، والعديد من الأهداف (مثل CLK8 [290]، mTOR [291]) والأدوية (مثل MLN4924 [292]) قيد التطوير مع تجارب سريرية جارية.
توفر البيانات
REFERENCES
- Scheiermann C, Gibbs J, Ince L, Loudon A. Clocking in to immunity. Nat Rev Immunol. 2018;18:423-37.
- Zhou L, Zhang Z, Nice E, Huang C, Zhang W, Tang Y. Circadian rhythms and cancers: the intrinsic links and therapeutic potentials. J Hematol Oncol. 2022;15:21.
- Patke A, Young MW, Axelrod S. Molecular mechanisms and physiological importance of circadian rhythms. Nat Rev Mol Cell Biol. 2020;21:67-84.
- Zhang R, Lahens NF, Ballance HI, Hughes ME, Hogenesch JB. A circadian gene expression atlas in mammals: implications for biology and medicine. Proc Natl Acad Sci USA. 2014;111:16219-24.
- Young MW, Kay SA. Time zones: a comparative genetics of circadian clocks. Nat Rev Genet. 2001;2:702-15.
- Fulgham CV, Dreyer AP, Nasseri A, Miller AN, Love J, Martin MM, et al. Central and peripheral clock control of circadian feeding rhythms. Biol Rhythms. 2021;36:548-66.
- Aton SJ, Herzog ED. Come together, right…now: synchronization of rhythms in a mammalian circadian clock. Neuron. 2005;48:531-4.
- Schibler U, Gotic I, Saini C, Gos P, Curie T, Emmenegger Y, et al. Clock-Talk: Interactions between Central and Peripheral Circadian Oscillators in Mammals. Cold Spring Harb Symp Quant Biol. 2015;80:223-32.
- Welsh DK, Yoo SH, Liu AC, Takahashi JS, Kay SA. Bioluminescence imaging of individual fibroblasts reveals persistent, independently phased circadian rhythms of clock gene expression. Curr Biol. 2004;14:2289-95.
- Cajochen C. Alerting effects of light. Sleep Med Rev. 2007;11:453-64.
- K Pavlova M, Latreille V. Sleep disorders. Am J Med. 2019;132:292-9.
- Ito
, Inoue . The international classification of sleep disorders, third edition. American Academy of Sleep Medicine. Includes bibliographies and index. Nihon Rinsho. 2015;73:916-23. - Sun SY, Chen GH. Treatment of circadian rhythm sleep-wake disorders. Curr Neuropharmacol. 2022;20:1022-34.
- Nagtegaal JE, Laurant MW, Kerkhof GA, Smits MG, van der Meer YG, Coenen AM. Effects of melatonin on the quality of life in patients with delayed sleep phase syndrome. J Psychosom Res. 2000;48:45-50.
- Yagita K, Tamanini F, Yasuda M, Hoeijmakers JH, van der Horst GT, Okamura H. Nucleocytoplasmic shuttling and mCRY-dependent inhibition of ubiquitylation of the mPER2 clock protein. EMBO J. 2002;21:1301-14.
- Guilding C, Scott F, Bechtold DA, Brown TM, Wegner S, Piggins HD. Suppressed cellular oscillations in after-hours mutant mice are associated with enhanced circadian phase-resetting. J Physiol. 2013;591:1063-80.
- Meng QJ, Logunova L, Maywood ES, Gallego M, Lebiecki J, Brown TM, et al. Setting clock speed in mammals: the CK1 epsilon tau mutation in mice accelerates circadian pacemakers by selectively destabilizing PERIOD proteins. Neuron. 2008;58:78-88.
- Eide EJ, Woolf MF, Kang H, Woolf P, Hurst W, Camacho F, et al. Control of mammalian circadian rhythm by CKlepsilon-regulated proteasome-mediated PER2 degradation. Mol Cell Biol. 2005;25:2795-807.
- Lowrey PL, Shimomura K, Antoch MP, Yamazaki S, Zemenides PD, Ralph MR, et al. Positional syntenic cloning and functional characterization of the mammalian circadian mutation tau. Science. 2000;288:483-92.
- Maier B, Wendt S, Vanselow JT, Wallach T, Reischl S, Oehmke S, et al. A largescale functional RNAi screen reveals a role for CK2 in the mammalian circadian clock. Genes Dev. 2009;23:708-18.
- Toh KL, Jones CR, He Y, Eide EJ, Hinz WA, Virshup DM, et al. An hPer2 phosphorylation site mutation in familial advanced sleep phase syndrome. Science. 2001;291:1040-3.
- Tsuchiya Y, Akashi M, Matsuda M, Goto K, Miyata Y, Node K, et al. Involvement of the protein kinase CK2 in the regulation of mammalian circadian rhythms. Sci Signal. 2009;2:ra26.
- Vanselow K, Vanselow JT, Westermark PO, Reischl S, Maier B, Korte T, et al. Differential effects of PER2 phosphorylation: molecular basis for the human familial advanced sleep phase syndrome (FASPS). Genes Dev. 2006;20:2660-72.
- Xu Y, Toh KL, Jones CR, Shin JY, Fu YH, Ptácek LJ. Modeling of a human circadian mutation yields insights into clock regulation by PER2. Cell. 2007;128:59-70.
- Zhou M, Kim JK, Eng GW, Forger DB, Virshup DM. A Period2 phosphoswitch regulates and temperature compensates circadian period. Mol Cell. 2015;60:77-88.
- Ohsaki K, Oishi K, Kozono Y, Nakayama K, Nakayama KI, Ishida N. The role of
P1 and P2 in circadian rhythm generation by mediating degradation of clock protein PER2. J Biochem. 2008;144:609-18. - Shirogane T, Jin J, Ang XL, Harper JW. SCFbeta-TRCP controls clock-dependent transcription via casein kinase 1-dependent degradation of the mammalian period-1 (Per1) protein. J Biol Chem. 2005;280:26863-72.
- Narasimamurthy R, Hunt SR, Lu Y, Fustin JM, Okamura H, Partch CL, et al. CK1
protein kinase primes the PER2 circadian phosphoswitch. Proc Natl Acad Sci USA. 2018;115:5986-91. - Gao P, Yoo SH, Lee KJ, Rosensweig C, Takahashi JS, Chen BP, et al. Phosphorylation of the cryptochrome 1 C-terminal tail regulates circadian period length. J Biol Chem. 2013;288:35277-86.
- Hirano A, Shi G, Jones CR, Lipzen A, Pennacchio LA, Xu Y, et al. A Cryptochrome 2 mutation yields advanced sleep phase in humans. Elife. 2016;5:e16695.
- Hirota T, Lee JW, St John PC, Sawa M, Iwaisako K, Noguchi T, et al. Identification of small molecule activators of cryptochrome. Science 2012;337:1094-7.
- Khan SK, Xu H, Ukai-Tadenuma M, Burton B, Wang Y, Ueda HR, et al. Identification of a novel cryptochrome differentiating domain required for feedback repression in circadian clock function. J Biol Chem. 2012;287:25917-26.
- Ode KL, Ukai H, Susaki EA, Narumi R, Matsumoto K, Hara J, et al. Knockout-rescue embryonic stem cell-derived mouse reveals circadian-period control by quality and quantity of CRY1. Mol Cell. 2017;65:176-90.
- Oshima T, Yamanaka I, Kumar A, Yamaguchi J, Nishiwaki-Ohkawa T, Muto K, et al. C-H activation generates period-shortening molecules that target cryptochrome in the mammalian circadian clock. Angew Chem Int Ed Engl. 2015;54:7193-7.
- Patke A, Murphy PJ, Onat OE, Krieger AC, Özçelik T, Campbell SS, et al. Mutation of the human circadian clock gene CRY1 in familial delayed sleep phase disorder. Cell. 2017;169:203-15.e13.
- Hirano A, Braas D, Fu YH, Ptáček LJ. FAD regulates CRYPTOCHROME protein stability and circadian clock in mice. Cell Rep. 2017;19:255-66.
- Busino L, Bassermann F, Maiolica A, Lee C, Nolan PM, Godinho SI, et al. SCFFbxl3 controls the oscillation of the circadian clock by directing the degradation of cryptochrome proteins. Science. 2007;316:900-4.
- Godinho SI, Maywood ES, Shaw L, Tucci V, Barnard AR, Busino L, et al. The afterhours mutant reveals a role for Fbxl3 in determining mammalian circadian period. Science. 2007;316:897-900.
- Hirano A, Yumimoto K, Tsunematsu R, Matsumoto M, Oyama M, Kozuka-Hata H, et al. FBXL21 regulates oscillation of the circadian clock through ubiquitination and stabilization of cryptochromes. Cell. 2013;152:1106-18.
- Saran AR, Kalinowska D, Oh S, Janknecht R, DiTacchio L. JMJD5 links CRY1 function and proteasomal degradation. PLoS Biol. 2018;16:e2006145.
- Siepka SM, Yoo SH, Park J, Song W, Kumar V, Hu Y, et al. Circadian mutant overtime reveals F-box protein FBXL3 regulation of cryptochrome and period gene expression. Cell. 2007;129:1011-23.
- Yoo SH, Mohawk JA, Siepka SM, Shan Y, Huh SK, Hong HK, et al. Competing E3 ubiquitin ligases govern circadian periodicity by degradation of CRY in nucleus and cytoplasm. Cell. 2013;152:1091-105.
- Zhang Y, Fang B, Emmett MJ, Damle M, Sun Z, Feng D, et al. Discrete functions of nuclear receptor Rev-erba couple metabolism to the clock. Science. 2015;348:1488-92.
- Cho H, Zhao X, Hatori M, Yu RT, Barish GD, Lam MT, et al. Regulation of circadian behaviour and metabolism by REV-ERB-a and REV-ERB-
. Nature. 2012;485:123-7. - Chung S, Lee EJ, Yun S, Choe HK, Park SB, Son HJ, et al. Impact of circadian nuclear receptor REV-ERBa on midbrain dopamine production and mood regulation. Cell. 2014;157:858-68.
- Takahashi JS. Transcriptional architecture of the mammalian circadian clock. Nat Rev Genet. 2017;18:164-79.
- Lowrey PL, Takahashi JS. Mammalian circadian biology: elucidating genomewide levels of temporal organization. Annu Rev Genomics Hum Genet. 2004;5:407-41.
- Preitner N, Damiola F, Lopez-Molina L, Zakany J, Duboule D, Albrecht U, et al. The orphan nuclear receptor REV-ERBalpha controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell. 2002;110:251-60.
- Shearman LP, Sriram S, Weaver DR, Maywood ES, Chaves I, Zheng B, et al. Interacting molecular loops in the mammalian circadian clock. Science. 2000;288:1013-9.
- Farshadi E, van der Horst G, Chaves I. Molecular links between the circadian clock and the cell cycle. J Mol Biol. 2020;432:3515-24.
- He B, Nohara K, Park N, Park YS, Guillory B, Zhao Z, et al. The small molecule nobiletin targets the molecular oscillator to enhance circadian rhythms and protect against metabolic syndrome. Cell Metab. 2016;23:610-21.
- Shinozaki A, Misawa K, Ikeda Y, Haraguchi A, Kamagata M, Tahara Y, et al. Potent effects of flavonoid nobiletin on amplitude, period, and phase of the circadian clock rhythm in PER2::LUCIFERASE mouse embryonic fibroblasts. PLoS ONE. 2017;12:e0170904.
- Onozuka H, Nakajima A, Matsuzaki K, Shin RW, Ogino K, Saigusa D, et al. Nobiletin, a citrus flavonoid, improves memory impairment and Abeta pathology in a transgenic mouse model of Alzheimer’s disease. J Pharm Exp Ther. 2008;326:739-44.
- Yabuki Y, Ohizumi Y, Yokosuka A, Mimaki Y, Fukunaga K. Nobiletin treatment improves motor and cognitive deficits seen in MPTP-induced Parkinson model mice. Neuroscience. 2014;259:126-41.
- Yi LT, Xu HL, Feng J, Zhan X, Zhou LP, Cui CC. Involvement of monoaminergic systems in the antidepressant-like effect of nobiletin. Physiol Behav. 2011;102:1-6.
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646-74.
- Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57-70.
- Vidya MK, Kumar VG, Sejian V, Bagath M, Krishnan G, Bhatta R. Toll-like receptors: significance, ligands, signaling pathways, and functions in mammals. Int Rev Immunol. 2018;37:20-36.
- Halberg F , johnson EA, brown BW, bittner JJ. Susceptibility rhythm to E. coli endotoxin and bioassay. Proc Soc Exp Biol Med. 1960;103:142-4.
- Silver AC, Buckley SM, Hughes ME, Hastings AK, Nitabach MN, Fikrig E. Daily oscillations in expression and responsiveness of Toll-like receptors in splenic immune cells. Heliyon. 2018;4:e00579.
- Kelly B, O’Neill LA. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res. 2015;25:771-84.
- Huang SC, Smith AM, Everts B, Colonna M, Pearce EL, Schilling JD, et al. Metabolic reprogramming mediated by the mTORC2-IRF4 signaling axis is essential for macrophage alternative activation. Immunity. 2016;45:817-30.
- Timmons GA, O’Siorain JR, Kennedy OD, Curtis AM, Early JO. Innate rhythms: clocks at the center of monocyte and macrophage function. Front Immunol. 2020;11:1743.
- Xu H, Li H, Woo SL, Kim SM, Shende VR, Neuendorff N, et al. Myeloid cell-specific disruption of Period1 and Period2 exacerbates diet-induced inflammation and insulin resistance. J Biol Chem. 2014;289:16374-88.
- Liu Z, Gan L, Zhang T, Ren Q, Sun C. Melatonin alleviates adipose inflammation through elevating a-ketoglutarate and diverting adipose-derived exosomes to macrophages in mice. J Pineal Res. 2018;64.
- Ding
, Lin N, Sheng X, Zhao Y, Su Y, Xu L, et al. Melatonin stabilizes ruptureprone vulnerable plaques via regulating macrophage polarization in a nuclear circadian receptor RORa-dependent manner. J Pineal Res. 2019;67:e12581. - Allen NC, Philip NH, Hui L, Zhou X, Franklin RA, Kong Y, et al. Desynchronization of the molecular clock contributes to the heterogeneity of the inflammatory response. Sci Signal. 2019;12:eaau1851.
- Pariollaud M, Gibbs JE, Hopwood TW, Brown S, Begley N, Vonslow R, et al. Circadian clock component REV-ERBa controls homeostatic regulation of pulmonary inflammation. J Clin Invest. 2018;128:2281-96.
- Gibbs JE, Blaikley J, Beesley S, Matthews L, Simpson KD, Boyce SH, et al. The nuclear receptor REV-ERBa mediates circadian regulation of innate immunity through selective regulation of inflammatory cytokines. Proc Natl Acad Sci USA. 2012;109:582-7.
- Griffin P, Dimitry JM, Sheehan PW, Lananna BV, Guo C, Robinette ML, et al. Circadian clock protein Rev-erba regulates neuroinflammation. Proc Natl Acad Sci USA. 2019;116:5102-7.
- Wang S, Lin Y, Yuan X, Li F, Guo L, Wu B. REV-ERBa integrates colon clock with experimental colitis through regulation of NF-kB/NLRP3 axis. Nat Commun. 2018;9:4246.
- Pourcet B, Zecchin M, Ferri L, Beauchamp J, Sitaula S, Billon C, et al. Nuclear receptor subfamily 1 group D member 1 regulates circadian activity of NLRP3 inflammasome to reduce the severity of fulminant hepatitis in mice. Gastroenterology. 2018;154:1449-64.e20.
- Wang T, Wang Z, Yang P, Xia L, Zhou M, Wang S, et al. PER1 prevents excessive innate immune response during endotoxin-induced liver injury through regulation of macrophage recruitment in mice. Cell Death Dis. 2016;7:e2176.
- Silver AC, Arjona A, Walker WE, Fikrig E. The circadian clock controls toll-like receptor 9-mediated innate and adaptive immunity. Immunity. 2012;36:251-61.
- Narasimamurthy R, Hatori M, Nayak SK, Liu F, Panda S, Verma IM. Circadian clock protein cryptochrome regulates the expression of proinflammatory cytokines. Proc Natl Acad Sci USA. 2012;109:12662-7.
- Yu D, Fang X, Xu Y, Xiao H, Huang T, Zhang Y, et al. Rev-erba can regulate the NF-кB/NALP3 pathway to modulate lipopolysaccharide-induced acute lung injury and inflammation. Int Immunopharmacology. 2019;73:312-20.
- McRae HM, Hargreaves DC. Old macrophages lose their (circadian) rhythm. Trends Immunol. 2022;43:265-7.
- Amir M, Campbell S, Kamenecka TM, Solt LA. Pharmacological modulation and genetic deletion of REV-ERBa and REV-ERB
regulates dendritic cell development. Biochem Biophys Res Commun. 2020;527:1000-7. - Nobis CC, Dubeau Laramée G, Kervezee L, Maurice De Sousa D, Labrecque N, Cermakian N. The circadian clock of CD8 T cells modulates their early response to vaccination and the rhythmicity of related signaling pathways. Proc Natl Acad Sci USA. 2019;116:20077-86.
- Holtkamp SJ, Ince LM, Barnoud C, Schmitt MT, Sinturel F, Pilorz V, et al. Circadian clocks guide dendritic cells into skin lymphatics. Nat Immunol. 2021;22:1375-81.
- Wang C, Barnoud C, Cenerenti M, Sun M, Caffa I, Kizil B, et al. Dendritic cells direct circadian anti-tumour immune responses. Nature. 2023;614:136-43.
- Sun Y, Yang Z, Niu Z, Peng J, Li Q, Xiong W, et al. MOP3, a component of the molecular clock, regulates the development of
cells. Immunology. 2006;119:451-60. - Hemmers S, Rudensky AY. The Cell-Intrinsic Circadian Clock Is Dispensable for Lymphocyte Differentiation and Function. Cell Rep. 2015;11:1339-49.
- Druzd D, Matveeva O, Ince L, Harrison U, He W, Schmal C, et al. Lymphocyte Circadian Clocks Control Lymph Node Trafficking and Adaptive Immune Responses. Immunity. 2017;46:120-32.
- Suzuki K, Hayano Y, Nakai A, Furuta F, Noda M. Adrenergic control of the adaptive immune response by diurnal lymphocyte recirculation through lymph nodes. J Exp Med. 2016;213:2567-74.
- Shimba A, Cui G, Tani-Ichi S, Ogawa M, Abe S, Okazaki F, et al. Glucocorticoids drive diurnal oscillations in
cell distribution and responses by inducing interleukin-7 receptor and CXCR4. Immunity. 2018;48:286-98.e6. - Förster R, Davalos-Misslitz AC, Rot A. CCR7 and its ligands: balancing immunity and tolerance. Nat Rev Immunol. 2008;8:362-71.
- Stein JV, Nombela-Arrieta C. Chemokine control of lymphocyte trafficking: a general overview. Immunology. 2005;116:1-12.
- Cyster JG, Schwab SR. Sphingosine-1-phosphate and lymphocyte egress from lymphoid organs. Annu Rev Immunol. 2012;30:69-94.
- Wang Q, Li L, Li C, Cao H, Chen Y, Zhou W, et al. Circadian protein CLOCK modulates regulatory
cell functions of nurses engaging day-night shift rotation. Cell Signal. 2022;96:110362. - Cermakian N, Labrecque N. Regulation of cytotoxic CD8 +T cells by the circadian clock. J Immunol. 2023;210:12-18.
- Besedovsky L, Born J, Lange T. Endogenous glucocorticoid receptor signaling drives rhythmic changes in human T-cell subset numbers and the expression of the chemokine receptor CXCR4. FASEB J. 2014;28:67-75.
- Dimitrov S, Benedict C, Heutling D, Westermann J, Born J, Lange T. Cortisol and epinephrine control opposing circadian rhythms in T cell subsets. Blood. 2009;113:5134-43.
- Deprés-Brummer P, Bourin P, Pages N, Metzger G, Lévi F. Persistent T lymphocyte rhythms despite suppressed circadian clock outputs in rats. Am J Physiol. 1997;273:R1891.
- Kawate T, Abo T, Hinuma S, Kumagai K. Studies of the bioperiodicity of the immune response. II. Co-variations of murine T and B cells and a role of corticosteroid. J Immunol. 1981;126:1364-7.
- Fortier EE, Rooney J, Dardente H, Hardy MP, Labrecque N, Cermakian N. Circadian variation of the response of T cells to antigen. J Immunol. 2011;187:6291-6300.
- Wu Y, Tao B, Zhang T, Fan Y, Mao R. Pan-cancer analysis reveals disrupted circadian clock associates with T cell exhaustion. Front Immunol. 2019;10:2451.
- Capelle CM, Chen A, Zeng N, Baron A, Grzyb K, Arns T, et al. Stress hormone signalling inhibits Th1 polarization in a CD4 T-cell-intrinsic manner via mTORC1 and the circadian gene PER1. Immunology. 2022;165:428-44.
- Yang G, Yang Y, Tang H, Yang K. Loss of the clock gene Per1 promotes oral squamous cell carcinoma progression via the AKT/mTOR pathway. Cancer Sci. 2020;111:1542-54.
- Juszczak M, Głabiński A. Th17 cells in the pathogenesis of multiple sclerosis. Postepy Hig Med Dosw. 2009;63:492-501.
- Yu X, Rollins D, Ruhn KA, Stubblefield JJ, Green CB, Kashiwada M, et al. TH17 cell differentiation is regulated by the circadian clock. Science. 2013;342:727-30.
- Chang JL, Qiu J. Regulation of ILC3/Th17-mediated intestinal immune response by circadian rhythm. Sichuan Da Xue Xue Bao Yi Xue Ban. 2022;53:15-19.
- Cheng FL, An YF, Xue JM, Wang YJ, Ding XW, Zhang YT, et al. Circadian rhythm disruption exacerbates Th2-like immune response in murine allergic airway inflammation. Int Forum Allergy Rhinol. 2022;12:757-70.
- Mandal A, Viswanathan C. Natural killer cells: in health and disease. Hematol Oncol Stem Cell Ther. 2015;8:47-55.
- Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S. Functions of natural killer cells. Nat Immunol. 2008;9:503-10.
- Mattiola I, Pesant M, Tentorio PF, Molgora M, Marcenaro E, Lugli E, et al. Priming of human resting NK cells by autologous M1 macrophages via the engagement of IL-1
, IFN- , and IL-15 pathways. J Immunol. 2015;195:2818-28. - Logan RW, Sarkar DK. Circadian nature of immune function. Mol Cell Endocrinol. 2012;349:82-90.
- Zeng X, Liang C, Yao J. Chronic shift-lag promotes NK cell ageing and impairs immunosurveillance in mice by decreasing the expression of CD122. J. Cell Mol Med. 2020;24:14583-95.
- Kennedy MK, Glaccum M, Brown SN, Butz EA, Viney JL, Embers M, et al. Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. J Exp Med. 2000;191:771-80.
- Chung JW, Yoon SR, Choi I. The regulation of NK cell function and development. Front Biosci. 2008;13:6432-42.
- Benca RM, Quintas J. Sleep and host defenses: a review. Sleep. 1997;20:1027-37.
- Besedovsky L, Lange T, Born J. Sleep and immune function. Pflug Arch. 2012;463:121-37.
- Lange T, Perras B, Fehm HL, Born J. Sleep enhances the human antibody response to hepatitis A vaccination. Psychosom Med. 2003;65:831-5.
- Spiegel K, Sheridan JF. Van Cauter E. Effect of sleep deprivation on response to immunization. JAMA. 2002;288:1471-2.
- Dimitrov S, Lange T, Tieken S, Fehm HL, Born J. Sleep associated regulation of T helper
helper 2 cytokine balance in humans. Brain Behav Immun. 2004;18:341-8. - Petrovsky N, Harrison LC. Diurnal rhythmicity of human cytokine production: a dynamic disequilibrium in T helper cell type
helper cell type 2 balance. J Immunol. 1997;158:5163-8. - Petrovsky N, Harrison LC. The chronobiology of human cytokine production. Int Rev Immunol. 1998;16:635-49.
- Born J, Fehm HL. Hypothalamus-pituitary-adrenal activity during human sleep: a coordinating role for the limbic hippocampal system. Exp Clin Endocrinol Diabetes. 1998;106:153-63.
- Sadamatsu M, Kato N, lida H, Takahashi S, Sakaue K, Takahashi K, et al. The 24hour rhythms in plasma growth hormone, prolactin and thyroid stimulating hormone: effect of sleep deprivation. J Neuroendocrinol. 1995;7:597-606.
- Spiegel K, Luthringer R, Follenius M, Schaltenbrand N, Macher JP, Muzet A, et al. Temporal relationship between prolactin secretion and slow-wave electroencephalic activity during sleep. Sleep. 1995;18:543-8.
- Alexander J, Satoskar AR, Russell DG. Leishmania species: models of intracellular parasitism. J Cell Sci. 1999;112:2993-3002.
- Karupiah G. Type 1 and type 2 cytokines in antiviral defense. Vet Immunol Immunopathol. 1998;63:105-9.
- Kühlwein E, Irwin M. Melatonin modulation of lymphocyte proliferation and Th1/Th2 cytokine expression. J Neuroimmunol. 2001;117:51-57.
- Lio D, Balistreri CR, Candore G, D’Anna C, Di Lorenzo G, Gervasi F, et al. In vitro treatment with interleukin-2 normalizes type-1 cytokine production by lymphocytes from elderly. Immunopharmacol Immunotoxicol. 2000;22:195-203.
- Van Cauter E, Leproult R, Plat L. Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA. 2000;284:861-8.
- Höflich C, Döcke WD, Busch A, Kern F, Volk HD. CD45RA(bright)/CD11a(bright) CD8+ T cells: effector T cells. Int Immunol. 1998;10:1837-45.
- von Andrian UH, Mackay CR. T-cell function and migration. Two sides of the same coin. N Engl J Med. 2000;343:1020-34.
- Irwin MR, Wang M, Campomayor CO, Collado-Hidalgo A, Cole S. Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation. Arch Intern Med. 2006;166:1756-62.
- Phillips B, Mannino DM. Does insomnia kill. Sleep. 2005;28:965-71.
- Vgontzas AN, Zoumakis E, Bixler EO, Lin HM, Follett H, Kales A, et al. Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. J Clin Endocrinol Metab. 2004;89:2119-26.
- Shearer WT, Reuben JM, Mullington JM, Price NJ, Lee BN, Smith EO, et al. Soluble TNF-alpha receptor 1 and IL-6 plasma levels in humans subjected to the sleep deprivation model of spaceflight. J Allergy Clin Immunol. 2001;107:165-70.
- Meier-Ewert HK, Ridker PM, Rifai N, Regan MM, Price NJ, Dinges DF, et al. Effect of sleep loss on C-reactive protein, an inflammatory marker of cardiovascular risk. J Am Coll Cardiol. 2004;43:678-83.
- Godinho-Silva C, Domingues RG, Rendas M, Raposo B, Ribeiro H, da Silva JA, et al. Light-entrained and brain-tuned circadian circuits regulate ILC3s and gut homeostasis. Nature. 2019;574:254-8.
- Stokes K, Cooke A, Chang H, Weaver DR, Breault DT, Karpowicz P. The circadian clock gene BMAL1 coordinates intestinal regeneration. Cell Mol Gastroenterol Hepatol. 2017;4:95-114.
- Qin B, Deng Y. Overexpression of circadian clock protein cryptochrome (CRY) 1 alleviates sleep deprivation-induced vascular inflammation in a mouse model. Immunol Lett. 2015;163:76-83.
- Magzal F, Shochat T, Haimov I, Tamir S, Asraf K, Tuchner-Arieli M, et al. Increased physical activity improves gut microbiota composition and reduces short-chain fatty acid concentrations in older adults with insomnia. Sci Rep. 2022;12:2265.
- Thaiss CA, Zeevi D, Levy M, Zilberman-Schapira G, Suez J, Tengeler AC, et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell. 2014;159:514-29.
- Yang DF, Huang WC, Wu CW, Huang CY, Yang Y, Tung YT. Acute sleep deprivation exacerbates systemic inflammation and psychiatry disorders through gut microbiota dysbiosis and disruption of circadian rhythms. Microbiol Res. 2023;268:127292.
- Koritala B, Porter KI, Arshad OA, Gajula RP, Mitchell HD, Arman T, et al. Night shift schedule causes circadian dysregulation of DNA repair genes and elevated DNA damage in humans. J Pineal Res. 2021;70:e12726.
- Haus EL, Smolensky MH. Shift work and cancer risk: potential mechanistic roles of circadian disruption, light at night, and sleep deprivation. Sleep Med Rev. 2013;17:273-84.
- James SM, Honn KA, Gaddameedhi S, Van Dongen H. Shift work: disrupted circadian rhythms and sleep-implications for health and well-being. Curr Sleep Med Rep. 2017;3:104-12.
- Savvidis C, Koutsilieris M. Circadian rhythm disruption in cancer biology. Mol Med. 2012;18:1249-60.
- Sahar S, Sassone-Corsi P. Metabolism and cancer: the circadian clock connection. Nat Rev Cancer. 2009;9:886-96.
- Sancar A, Lindsey-Boltz LA, Gaddameedhi S, Selby CP, Ye R, Chiou YY, et al. Circadian clock, cancer, and chemotherapy. Biochemistry. 2015;54:110-23.
- Yang Y, Liu Z, Selby CP, Sancar A. Long-term, genome-wide kinetic analysis of the effect of the circadian clock and transcription on the repair of cisplatin-DNA adducts in the mouse liver. J Biol Chem. 2019;294:11960-8.
- Gaucher J, Montellier E, Sassone-Corsi P. Molecular cogs: interplay between circadian clock and cell cycle. Trends Cell Biol. 2018;28:368-79.
- Miller BH, McDearmon EL, Panda S, Hayes KR, Zhang J, Andrews JL, et al. Circadian and CLOCK-controlled regulation of the mouse transcriptome and cell proliferation. Proc Natl Acad Sci USA. 2007;104:3342-7.
- Scheiermann C, Kunisaki Y, Frenette PS. Circadian control of the immune system. Nat Rev Immunol. 2013;13:190-8.
- Keller M, Mazuch J, Abraham U, Eom GD, Herzog ED, Volk HD, et al. A circadian clock in macrophages controls inflammatory immune responses. Proc Natl Acad Sci USA. 2009;106:21407-12.
- Masri S, Cervantes M, Sassone-Corsi P. The circadian clock and cell cycle: interconnected biological circuits. Curr Opin Cell Biol. 2013;25:730-4.
- Roenneberg T, Merrow M. The circadian clock and human health. Curr Biol. 2016;26:R432-43.
- Depner CM, Melanson EL, McHill AW, Wright KP Jr. Mistimed food intake and sleep alters 24-hour time-of-day patterns of the human plasma proteome. Proc Natl Acad Sci USA 2018;115:E5390-9.
- Kervezee L, Cuesta M, Cermakian N, Boivin DB. Simulated night shift work induces circadian misalignment of the human peripheral blood mononuclear cell transcriptome. Proc Natl Acad Sci USA. 2018;115:5540-5.
- Skene DJ, Skornyakov E, Chowdhury NR, Gajula RP, Middleton B, Satterfield BC, et al. Separation of circadian- and behavior-driven metabolite rhythms in humans provides a window on peripheral oscillators and metabolism. Proc Natl Acad Sci USA. 2018;115:7825-30.
- Resuehr D, Wu G, Johnson RL Jr, Young ME, Hogenesch JB, Gamble KL. Shift work disrupts circadian regulation of the transcriptome in hospital nurses. J Biol Rhythms. 2019;34:167-77.
- Humans, IMotloCHt, Work iNS. Night shift work. 2020. Lyon (FR). other.
- Papagiannakopoulos T, Bauer MR, Davidson SM, Heimann M, Subbaraj L, Bhutkar A, et al. Circadian rhythm disruption promotes lung tumorigenesis. Cell Metab. 2016;24:324-31.
- Lee Y, Lahens NF, Zhang S, Bedont J, Field JM, Sehgal A. G1/S cell cycle regulators mediate effects of circadian dysregulation on tumor growth and provide targets for timed anticancer treatment. PLoS Biol. 2019;17:e3000228.
- Van Dycke KC, Rodenburg W, van Oostrom CT, van Kerkhof LW, Pennings JL, Roenneberg T, et al. Chronically alternating light cycles increase breast cancer risk in mice. Curr Biol. 2015;25:1932-7.
- Aiello I, Fedele M, Román F, Marpegan L, Caldart C, Chiesa JJ, et al. Circadian disruption promotes tumor-immune microenvironment remodeling favoring tumor cell proliferation. Sci Adv. 2020;6:eaaz4530.
- Bentivoglio M, Grassi-Zucconi G. The pioneering experimental studies on sleep deprivation. Sleep. 1997;20:570-6.
- Rechtschaffen A, Gilliland MA, Bergmann BM, Winter JB. Physiological correlates of prolonged sleep deprivation in rats. Science. 1983;221:182-4.
- Shaw PJ, Tononi G, Greenspan RJ, Robinson DF. Stress response genes protect against lethal effects of sleep deprivation in Drosophila. Nature. 2002;417:287-91.
- Stephenson R, Chu KM, Lee J. Prolonged deprivation of sleep-like rest raises metabolic rate in the Pacific beetle cockroach, Diploptera punctata (Eschscholtz). J Exp Biol. 2007;210:2540-7.
- Donlea JM. Roles for sleep in memory: insights from the fly. Curr Opin Neurobiol. 2019;54:120-6.
- Killgore WD. Effects of sleep deprivation on cognition. Prog Brain Res. 2010;185:105-29.
- Krause AJ, Simon EB, Mander BA, Greer SM, Saletin JM, Goldstein-Piekarski AN, et al. The sleep-deprived human brain. Nat Rev Neurosci. 2017;18:404-18.
- Reimund E. The free radical flux theory of sleep. Med Hypotheses. 1994;43:231-3.
- Alzoubi KH, Khabour OF, Rashid BA, Damaj IM, Salah HA. The neuroprotective effect of vitamin E on chronic sleep deprivation-induced memory impairment: the role of oxidative stress. Behav Brain Res. 2012;226:205-10.
- Hill VM, O’Connor RM, Sissoko GB, Irobunda IS, Leong S, Canman JC, et al. A bidirectional relationship between sleep and oxidative stress in Drosophila. PLoS Biol. 2018;16:e2005206.
- Kanazawa L, Vecchia DD, Wendler EM, Hocayen P, Dos Reis Lívero FA, Stipp MC, et al. Quercetin reduces manic-like behavior and brain oxidative stress induced by paradoxical sleep deprivation in mice. Free Radic Biol Med. 2016;99:79-86.
- Süer C, Dolu N, Artis AS, Sahin L, Yilmaz A, Cetin A. The effects of long-term sleep deprivation on the long-term potentiation in the dentate gyrus and brain oxidation status in rats. Neurosci Res. 2011;70:71-77.
- Villafuerte G, Miguel-Puga A, Rodríguez EM, Machado S, Manjarrez E, AriasCarrión O. Sleep deprivation and oxidative stress in animal models: a systematic review. Oxid Med Cell Longev. 2015;2015:234952.
- Vaccaro A, Kaplan Dor Y, Nambara K, Pollina EA, Lin C, Greenberg ME, et al. Sleep loss can cause death through accumulation of reactive oxygen species in the gut. Cell. 2020;181:1307-28.e15.
- Balaban RS, Nemoto S, Finkel T. Mitochondria, oxidants, and aging. Cell. 2005;120:483-95.
- Shimizu Y, Hendershot LM. Oxidative folding: cellular strategies for dealing with the resultant equimolar production of reactive oxygen species. Antioxid Redox Signal. 2009;11:2317-31.
- Cao SS, Kaufman RJ. Endoplasmic reticulum stress and oxidative stress in cell fate decision and human disease. Antioxid Redox Signal. 2014;21:396-413.
- Santos CX, Tanaka LY, Wosniak J, Laurindo FR. Mechanisms and implications of reactive oxygen species generation during the unfolded protein response: roles of endoplasmic reticulum oxidoreductases, mitochondrial electron transport, and NADPH oxidase. Antioxid Redox Signal. 2009;11:2409-27.
- Zeeshan HM, Lee GH, Kim HR, Chae HJ. Endoplasmic Reticulum Stress and Associated ROS. Int J Mol Sci. 2016;17:327.
- Cirelli C, Faraguna U, Tononi G. Changes in brain gene expression after longterm sleep deprivation. J Neurochem. 2006;98:1632-45.
- Eiland MM, Ramanathan L, Gulyani S, Gilliland M, Bergmann BM, Rechtschaffen A, et al. Increases in amino-cupric-silver staining of the supraoptic nucleus after sleep deprivation. Brain Res. 2002;945:1-8.
- Gopalakrishnan A, Ji LL, Cirelli C. Sleep deprivation and cellular responses to oxidative stress. Sleep. 2004;27:27-35.
- Hipólide DC, D’Almeida V, Raymond R, Tufik S, Nobrega JN. Sleep deprivation does not affect indices of necrosis or apoptosis in rat brain. Int J Neurosci. 2002;112:155-66.
- Brown MK, Strus E, Naidoo N. Reduced sleep during social isolation leads to cellular stress and induction of the unfolded protein response. Sleep. 2017;40:zsx095.
- Nikonova EV, Naidoo N, Zhang L, Romer M, Cater JR, Scharf MT, et al. Changes in components of energy regulation in mouse cortex with increases in wakefulness. Sleep. 2010;33:889-900.
- Jones S, Pfister-Genskow M, Benca RM, Cirelli C. Molecular correlates of sleep and wakefulness in the brain of the white-crowned sparrow. J Neurochem. 2008;105:46-62.
- Cirelli C, LaVaute TM, Tononi G. Sleep and wakefulness modulate gene expression in Drosophila. J Neurochem. 2005;94:1411-9.
- Cirelli C. Cellular consequences of sleep deprivation in the brain. Sleep Med Rev. 2006;10:307-21.
- Ali T, Choe J, Awab A, Wagener TL, Orr WC. Sleep, immunity and inflammation in gastrointestinal disorders. World J Gastroenterol. 2013;19:9231-9.
- Khanijow V, Prakash P, Emsellem HA, Borum ML, Doman DB. Sleep dysfunction and gastrointestinal diseases. Gastroenterol Hepatol. 2015;11:817-25.
- Parekh PJ, Oldfield EC 4th, Johnson DA. Wake-up call to clinicians: the impact of sleep dysfunction on gastrointestinal health and disease. J Clin Gastroenterol. 2018;52:194-203.
- Bhattacharyya A, Chattopadhyay R, Mitra S, Crowe SE. Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol Rev. 2014;94:329-54.
- Campbell EL, Colgan SP. Control and dysregulation of redox signalling in the gastrointestinal tract. Nat Rev Gastroenterol Hepatol. 2019;16:106-20.
- Pérez S, Taléns-Visconti R, Rius-Pérez S, Finamor I, Sastre J. Redox signaling in the gastrointestinal tract. Free Radic Biol Med. 2017;104:75-103.
- Aviello G, Knaus UG. ROS in gastrointestinal inflammation: Rescue Or Sabotage. Br J Pharm. 2017;174:1704-18.
- Lasry A, Zinger A, Ben-Neriah Y. Inflammatory networks underlying colorectal cancer. Nat Immunol. 2016;17:230-40.
- Chen GY, Nuñez G. Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol. 2010;10:826-37.
- Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11:98-107.
- Libby P, Lichtman AH, Hansson GK. Immune effector mechanisms implicated in atherosclerosis: from mice to humans. Immunity. 2013;38:1092-104.
- Bruunsgaard H, Ladelund S, Pedersen AN, Schroll M, Jørgensen T, Pedersen BK. Predicting death from tumour necrosis factor-alpha and interleukin-6 in 80-yearold people. Clin Exp Immunol. 2003;132:24-31.
- Volpato S, Guralnik JM, Ferrucci L, Balfour J, Chaves P, Fried LP, et al. Cardiovascular disease, interleukin-6, and risk of mortality in older women: the women’s health and aging study. Circulation. 2001;103:947-53.
- Smagula SF, Stone KL, Redline S, Ancoli-Israel S, Barrett-Connor E, Lane NE, et al. Actigraphy- and polysomnography-measured sleep disturbances, inflammation, and mortality among older men. Psychosom Med. 2016;78:686-96.
- Ridker PM. From C-reactive protein to interleukin-6 to interleukin-1: moving upstream to identify novel targets for atheroprotection. Circ Res. 2016;118:145-56.
- Grandner MA, Alfonso-Miller P, Fernandez-Mendoza J, Shetty S, Shenoy S, Combs D. Sleep: important considerations for the prevention of cardiovascular disease. Curr Opin Cardiol. 2016;31:551-65.
- Ferrie JE, Kivimäki M, Akbaraly TN, Singh-Manoux A, Miller MA, Gimeno D, et al. Associations between change in sleep duration and inflammation: findings on C-reactive protein and interleukin 6 in the Whitehall II Study. Am J Epidemiol. 2013;178:956-61.
- Carroll JE, Irwin MR, Stein Merkin S, Seeman TE. Sleep and multisystem biological risk: a population-based study. PLoS ONE. 2015;10:e0118467.
- Clark AJ, Dich N, Lange T, Jennum P, Hansen AM, Lund R, et al. Impaired sleep and allostatic load: cross-sectional results from the Danish Copenhagen Aging and Midlife Biobank. Sleep Med. 2014;15:1571-8.
- Dowd JB, Goldman N, Weinstein M. Sleep duration, sleep quality, and biomarkers of inflammation in a Taiwanese population. Ann Epidemiol. 2011;21:799-806.
- Patel SR, Zhu X, Storfer-Isser A, Mehra R, Jenny NS, Tracy R, et al. Sleep duration and biomarkers of inflammation. Sleep. 2009;32:200-4.
- Prather AA, Vogelzangs N, Penninx BW. Sleep duration, insomnia, and markers of systemic inflammation: results from the Netherlands Study of Depression and Anxiety (NESDA). J Psychiatr Res. 2015;60:95-102.
- Jackowska M, Steptoe A. Sleep and future cardiovascular risk: prospective anaIysis from the English Longitudinal Study of Ageing. Sleep Med. 2015;16:768-74.
- McDade TW, Hawkley LC, Cacioppo JT. Psychosocial and behavioral predictors of inflammation in middle-aged and older adults: the Chicago health, aging, and social relations study. Psychosom Med. 2006;68:376-81.
- Prather AA, Marsland AL, Hall M, Neumann SA, Muldoon MF, Manuck SB. Normative variation in self-reported sleep quality and sleep debt is associated with stimulated pro-inflammatory cytokine production. Biol Psychol. 2009;82:12-17.
- Suarez EC. Self-reported symptoms of sleep disturbance and inflammation, coagulation, insulin resistance and psychosocial distress: evidence for gender disparity. Brain Behav Immun. 2008;22:960-8.
- Taheri S, Austin D, Lin L, Nieto FJ, Young T, Mignot E. Correlates of serum C-reactive protein (CRP)-no association with sleep duration or sleep disordered breathing. Sleep. 2007;30:991-6.
- Prather AA, Epel ES, Cohen BE, Neylan TC, Whooley MA. Gender differences in the prospective associations of self-reported sleep quality with biomarkers of systemic inflammation and coagulation: findings from the Heart and Soul Study. J Psychiatr Res. 2013;47:1228-35.
- Obayashi K, Saeki K, Kurumatani N. Gender differences in the association between objective sleep quality and leukocyte count: The HEIJO-KYO cohort. Physiol Behav. 2016;164:19-24.
- Blair LM, Porter K, Leblebicioglu B, Christian LM. Poor sleep quality and associated inflammation predict preterm birth: heightened risk among African Americans. Sleep. 2015;38:1259-67.
- Okun ML, Luther JF, Wisniewski SR, Wisner KL. Disturbed sleep and inflammatory cytokines in depressed and nondepressed pregnant women: an exploratory analysis of pregnancy outcomes. Psychosom Med. 2013;75:670-81.
- Tsujimura T, Matsuo Y, Keyaki T, Sakurada K, Imanishi J. Correlations of sleep disturbance with the immune system in type 2 diabetes mellitus. Diabetes Res Clin Pr. 2009;85:286-92.
- Besedovsky L, Lange T, Haack M. The sleep-immune crosstalk in health and disease. Physiol Rev. 2019;99:1325-80.
- Opp MR. Cytokines and sleep. Sleep Med Rev. 2005;9:355-64.
- Lou X, Wang H, Tu Y, Tan W, Jiang C, Sun J, et al. Alterations of sleep quality and circadian rhythm genes expression in elderly thyroid nodule patients and risks associated with thyroid malignancy. Sci Rep. 2021;11:13682.
- Zhou L, Luo Z, Li Z, Huang Q. Circadian clock is associated with tumor microenvironment in kidney renal clear cell carcinoma. Aging. 2020;12:14620-32.
- Ramos CA, Ouyang C, Qi Y, Chung Y, Cheng CT, LaBarge MA, et al. A noncanonical function of BMAL1 metabolically limits obesity-promoted triplenegative breast cancer. iScience. 2020;23:100839.
- Chen P, Hsu WH, Chang A, Tan Z, Lan Z, Zhou A, et al. Circadian regulator CLOCK recruits immune-suppressive microglia into the GBM tumor microenvironment. Cancer Discov. 2020;10:371-81.
- He L, Fan Y, Zhang Y, Tu T, Zhang Q, Yuan F, et al. Single-cell transcriptomic analysis reveals circadian rhythm disruption associated with poor prognosis and drug-resistance in lung adenocarcinoma. J Pineal Res. 2022;73:e12803.
- Chun SK, Fortin BM, Fellows RC, Habowski AN, Verlande A, Song WA, et al. Disruption of the circadian clock drives Apc loss of heterozygosity to accelerate colorectal cancer. Sci Adv. 2022;8:eabo2389.
- Hunt T, Sassone-Corsi P. Riding tandem: circadian clocks and the cell cycle. Cell. 2007;129:461-4.
- Satyanarayana A, Kaldis P. Mammalian cell-cycle regulation: several Cdks, numerous cyclins and diverse compensatory mechanisms. Oncogene. 2009;28:2925-39.
- Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19:1423-37.
- Xuan W, Khan F, James CD, Heimberger AB, Lesniak MS, Chen P. Circadian regulation of cancer cell and tumor microenvironment crosstalk. Trends Cell Biol. 2021;31:940-50.
- Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39:1-10.
- Raskov H, Orhan A, Christensen JP, Gögenur I. Cytotoxic CD8(+) T cells in cancer and cancer immunotherapy. Br J Cancer. 2021;124:359-67.
- Marvel D, Gabrilovich Dl. Myeloid-derived suppressor cells in the tumor microenvironment: expect the unexpected. J Clin Invest. 2015;125:3356-64.
- Roberts NT, MacDonald CR, Mohammadpour H, Antoch MP, Repasky EA. Circadian rhythm disruption increases tumor growth rate and accumulation of myeloid-derived suppressor cells. Adv Biol. 2022;6:e2200031.
- Méndez-Ferrer S, Lucas D, Battista M, Frenette PS. Haematopoietic stem cell release is regulated by circadian oscillations. Nature. 2008;452:442-7.
- Logan RW, Arjona A, Sarkar DK. Role of sympathetic nervous system in the entrainment of circadian natural-killer cell function. Brain Behav Immun. 2011;25:101-9.
- Pathria P, Louis TL, Varner JA. Targeting tumor-associated macrophages in cancer. Trends Immunol. 2019;40:310-27.
- Alexander RK, Liou YH, Knudsen NH, Starost KA, Xu C, Hyde AL, et al. Bmal1 integrates mitochondrial metabolism and macrophage activation. Elife. 2020;9:e54090.
- Early JO, Menon D, Wyse CA, Cervantes-Silva MP, Zaslona Z, Carroll RG, et al. Circadian clock protein BMAL1 regulates IL-1
in macrophages via NRF2. Proc Natl Acad Sci USA. 2018;115:E8460-8. - Nguyen KD, Fentress SJ, Qiu Y, Yun K, Cox JS, Chawla A. Circadian gene Bmal1 regulates diurnal oscillations of Ly6C(hi) inflammatory monocytes. Science. 2013;341:1483-8.
- Huang C , Zhang C , Cao Y, Li J, Bi F. Major roles of the circadian clock in cancer. Cancer Biol Med. 2023;20:1-24.
- Sato S, Sakurai T, Ogasawara J, Takahashi M, Izawa T, Imaizumi K, et al. A circadian clock gene, Rev-erba, modulates the inflammatory function of macrophages through the negative regulation of Ccl 2 expression. J Immunol. 2014;192:407-17.
- Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004;25:677-86.
- Verreck FA, de Boer T, Langenberg DM, Hoeve MA, Kramer M, Vaisberg E, et al. Human IL-23-producing type 1 macrophages promote but IL-10-producing type 2 macrophages subvert immunity to (myco)bacteria. Proc Natl Acad Sci USA. 2004;101:4560-5.
- Qin C, Zhou LQ, Ma XT, Hu ZW, Yang S, Chen M, et al. Dual functions of microglia in ischemic stroke. Neurosci Bull. 2019;35:921-33.
- Li X, Guan J, Jiang Z, Cheng S, Hou W, Yao J, et al. Microglial exosome miR-72393p promotes glioma progression by regulating circadian genes. Neurosci Bull. 2021;37:497-510.
- Newman AM, Alizadeh AA. High-throughput genomic profiling of tumorinfiltrating leukocytes. Curr Opin Immunol. 2016;41:77-84.
- He W, Holtkamp S, Hergenhan SM, Kraus K, de Juan A, Weber J, et al. Circadian expression of migratory factors establishes lineage-specific signatures that guide the homing of leukocyte subsets to tissues. Immunity. 2018;49:1175-90.e7.
- Gao Y, Meng D, Sun N, Zhu Z, Zhao R, Lu C, et al. Clock upregulates intercellular adhesion molecule-1 expression and promotes mononuclear cells adhesion to endothelial cells. Biochem Biophys Res Commun. 2014;443:586-91.
- Hadadi E, Taylor W, Li XM, Aslan Y, Villote M, Rivière J, et al. Chronic circadian disruption modulates breast cancer stemness and immune microenvironment to drive metastasis in mice. Nat Commun. 2020;11:3193.
- Hu X, Liu X, Moisan J, Wang Y, Lesch CA, Spooner C, et al. Synthetic RORY agonists regulate multiple pathways to enhance antitumor immunity. Oncoimmunology. 2016;5:e1254854.
- Lee IK, Song H, Kim H, Kim IS, Tran NL, Kim SH, et al. RORa regulates cholesterol metabolism of CD8+ T cells for anticancer immunity. Cancers. 2020;12:1733.
- Kaplon J, van Dam L, Peeper D. Two-way communication between the metabolic and cell cycle machineries: the molecular basis. Cell Cycle. 2015;14:2022-32.
- de Winter L, Schepers LW, Cuaresma M, Barbosa MJ, Martens DE, Wijffels RH. Circadian rhythms in the cell cycle and biomass composition of Neochloris oleoabundans under nitrogen limitation. J Biotechnol. 2014;187:25-33.
- Krishnaiah SY, Wu G, Altman BJ, Growe J, Rhoades SD, Coldren F, et al. Clock regulation of metabolites reveals coupling between transcription and metabolism. Cell Metab. 2017;25:961-74.e4.
- Janich P, Pascual G, Merlos-Suárez A, Batlle E, Ripperger J, Albrecht U, et al. The circadian molecular clock creates epidermal stem cell heterogeneity. Nature. 2011;480:209-14.
- Geyfman M, Kumar V, Liu Q, Ruiz R, Gordon W, Espitia F, et al. Brain and muscle Arnt-like protein-1 (BMAL1) controls circadian cell proliferation and susceptibility to UVB-induced DNA damage in the epidermis. Proc Natl Acad Sci USA. 2012;109:11758-63.
- Plikus MV, Vollmers C, de la Cruz D, Chaix A, Ramos R, Panda S, et al. Local circadian clock gates cell cycle progression of transient amplifying cells during regenerative hair cycling. Proc Natl Acad Sci USA. 2013;110:E2106-15.
- Karpowicz P, Zhang Y, Hogenesch JB, Emery P, Perrimon N. The circadian clock gates the intestinal stem cell regenerative state. Cell Rep. 2013;3:996-1004.
- Weger M, Diotel N, Dorsemans AC, Dickmeis T, Weger BD. Stem cells and the circadian clock. Dev Biol. 2017;431:111-23.
- Kowalska E, Ripperger JA, Hoegger DC, Bruegger P, Buch T, Birchler T, et al. NONO couples the circadian clock to the cell cycle. Proc Natl Acad Sci USA. 2013;110:1592-9.
- Bjarnason GA, Jordan RC, Wood PA, Li Q, Lincoln DW, Sothern RB, et al. Circadian expression of clock genes in human oral mucosa and skin: association with specific cell-cycle phases. Am J Pathol. 2001;158:1793-801.
- Bjarnason GA, Jordan RC, Sothern RB. Circadian variation in the expression of cell-cycle proteins in human oral epithelium. Am J Pathol. 1999;154:613-22.
- Soták M, Sumová A, Pácha J. Cross-talk between the circadian clock and the cell cycle in cancer. Ann Med. 2014;46:221-32.
- Matsuo T, Yamaguchi S, Mitsui S, Emi A, Shimoda F, Okamura H. Control mechanism of the circadian clock for timing of cell division in vivo. Science. 2003;302:255-9.
- Gréchez-Cassiau A, Rayet B, Guillaumond F, Teboul M, Delaunay F. The circadian clock component BMAL1 is a critical regulator of p21WAF1/CIP1 expression and hepatocyte proliferation. J Biol Chem. 2008;283:4535-42.
- Miki T, Matsumoto T, Zhao Z, Lee CC. p53 regulates Period2 expression and the circadian clock. Nat Commun. 2013;4:2444.
- Bee L, Marini S, Pontarin G, Ferraro P, Costa R, Albrecht U, et al. Nucleotide excision repair efficiency in quiescent human fibroblasts is modulated by circadian clock. Nucleic Acids Res. 2015;43:2126-37.
- Oklejewicz M, Destici E, Tamanini F, Hut RA, Janssens R, van der Horst GT. Phase resetting of the mammalian circadian clock by DNA damage. Curr Biol. 2008;18:286-91.
- Masri S, Kinouchi K, Sassone-Corsi P. Circadian clocks, epigenetics, and cancer. Curr Opin Oncol. 2015;27:50-56.
- Scheiermann C, Kunisaki Y, Lucas D, Chow A, Jang JE, Zhang D, et al. Adrenergic nerves govern circadian leukocyte recruitment to tissues. Immunity. 2012;37:290-301.
- Wang Y, Sun N, Lu C, Bei Y, Qian R, Hua L. Upregulation of circadian gene ‘hClock’ contribution to metastasis of colorectal cancer. Int J Oncol. 2017;50:2191-9.
- Hu Z, Brooks SA, Dormoy V, Hsu CW, Hsu HY, Lin LT, et al. Assessing the carcinogenic potential of low-dose exposures to chemical mixtures in the environment: focus on the cancer hallmark of tumor angiogenesis. Carcinogenesis. 2015;36:S184-202.
- Jiang X, Wang J, Deng X, Xiong F, Zhang S, Gong Z, et al. The role of microenvironment in tumor angiogenesis. J Exp Clin Cancer Res. 2020;39:204.
- Koyanagi S, Kuramoto Y, Nakagawa H, Aramaki H, Ohdo S, Soeda S, et al. A molecular mechanism regulating circadian expression of vascular endothelial growth factor in tumor cells. Cancer Res. 2003;63:7277-83.
- Zhou J, Li X, Zhang M, Gong J, Li Q, Shan B, et al. The aberrant expression of rhythm genes affects the genome instability and regulates the cancer immunity in pan-cancer. Cancer Med. 2020;9:1818-29.
- Shi SQ, Ansari TS, McGuinness OP, Wasserman DH, Johnson CH. Circadian disruption leads to insulin resistance and obesity. Curr Biol. 2013;23:372-81.
- Kettner NM, Mayo SA, Hua J, Lee C, Moore DD, Fu L. Circadian dysfunction induces leptin resistance in mice. Cell Metab. 2015;22:448-59.
- Gabrilovich DI, Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol. 2009;9:162-74.
- Michaeli J, Shaul ME, Mishalian I, Hovav AH, Levy L, Zolotriov L, et al. Tumorassociated neutrophils induce apoptosis of non-activated CD8 T-cells in a TNFa and NO-dependent mechanism, promoting a tumor-supportive environment. Oncoimmunology. 2017;6:e1356965.
- Feng S, Cheng X, Zhang L, Lu X, Chaudhary S, Teng R, et al. Myeloid-derived suppressor cells inhibit T cell activation through nitrating LCK in mouse cancers. Proc Natl Acad Sci USA. 2018;115:10094-9.
- Peranzoni E, Lemoine J, Vimeux L, Feuillet V, Barrin S, Kantari-Mimoun C, et al. Macrophages impede CD8 T cells from reaching tumor cells and limit the efficacy of anti-PD-1 treatment. Proc Natl Acad Sci USA. 2018;115:E4041-50.
- Zhao Y, Liu M, Chan XY, Tan SY, Subramaniam S, Fan Y, et al. Uncovering the mystery of opposite circadian rhythms between mouse and human leukocytes in humanized mice. Blood. 2017;130:1995-2005.
- Müller A, Homey B, Soto H, Ge N, Catron D, Buchanan ME, et al. Involvement of chemokine receptors in breast cancer metastasis. Nature. 2001;410:50-6.
- Helbig G, Christopherson KW 2nd, Bhat-Nakshatri P, Kumar S, Kishimoto H, Miller KD, et al. NF-kappaB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J Biol Chem. 2003;278:21631-8.
- Nagarsheth N, Wicha MS, Zou W. Chemokines in the cancer microenvironment and their relevance in cancer immunotherapy. Nat Rev Immunol. 2017;17:559-72.
- Cermakian N, Stegeman SK, Tekade K, Labrecque N. Circadian rhythms in adaptive immunity and vaccination. Semin Immunopathol. 2022; 44:193-207.
- Doruk YU, Yarparvar D, Akyel YK, Gul S, Taskin AC, Yilmaz F, et al. A CLOCKbinding small molecule disrupts the interaction between CLOCK and BMAL1 and enhances circadian rhythm amplitude. J Biol Chem. 2020;295:3518-31.
- Ramanathan C, Kathale ND, Liu D, Lee C, Freeman DA, Hogenesch JB, et al. mTOR signaling regulates central and peripheral circadian clock function. PLoS Genet. 2018;14:e1007369.
- Zhang S, Zhang J, Deng Z, Liu H, Mao W, Jiang F, et al. Circadian clock components RORa and Bmal1 mediate the anti-proliferative effect of MLN4924 in osteosarcoma cells. Oncotarget. 2016;7:66087-99.
- Zhang H, Liu Y, Liu D, Zeng Q, Li L, Zhou Q, et al. Time of day influences immune response to an inactivated vaccine against SARS-CoV-2. Cell Res. 2021;31:1215-7.
- Bass J. Circadian topology of metabolism. Nature. 2012;491:348-56.
- Masri S, Sassone-Corsi P. The emerging link between cancer, metabolism, and circadian rhythms. Nat Med. 2018;24:1795-803.
مساهمات المؤلفين
التمويل
المصالح المتنافسة
معلومات إضافية
معلومات إعادة الطبع والإذن متاحة علىhttp://www.nature.com/إعادة الطبع

© المؤلف(ون) 2024
قسم المناعة، كلية العلوم الطبية الأساسية، جامعة القوات الجوية الطبية، شيان، الصين. كلية علوم الحياة، جامعة شمال غرب، شيان، الصين. ساهم هؤلاء المؤلفون بالتساوي: يوان زينغ، زيتشان قوه. البريد الإلكتروني: chenlh@fmmu.edu.cn
DOI: https://doi.org/10.1038/s41420-024-01960-1
PMID: https://pubmed.ncbi.nlm.nih.gov/38678017
Publication Date: 2024-04-27
Circadian rhythm regulates the function of immune cells and participates in the development of tumors
Abstract
Circadian rhythms are present in almost all cells and play a crucial role in regulating various biological processes. Maintaining a stable circadian rhythm is essential for overall health. Disruption of this rhythm can alter the expression of clock genes and cancerrelated genes, and affect many metabolic pathways and factors, thereby affecting the function of the immune system and contributing to the occurrence and progression of tumors. This paper aims to elucidate the regulatory effects of BMAL1, clock and other clock genes on immune cells, and reveal the molecular mechanism of circadian rhythm’s involvement in tumor and its microenvironment regulation. A deeper understanding of circadian rhythms has the potential to provide new strategies for the treatment of cancer and other immune-related diseases.
FACTS
- Circadian rhythms profoundly affect various physiological processes in the body.
- Circadian rhythms influence the maturation, localization, and function of immune cells in a variety of ways (including genes, adhesion molecules, cytokines, etc.).
- The immune system can respond by influencing circadian rhythms.
- Circadian rhythms are involved in cell cycle regulation.
- The components related to circadian rhythm in tumor cells and their microenvironment are altered, and there is an epidemiological link between cancer risk and circadian rhythm gene expression.
OPEN QUESTIONS
- Can circadian rhythm considerations be used to optimize clinical administration, nutrition, and surgical timing to optimize patient outcomes?
- Can circadian rhythms be integrated into drug use and surgical timing for better outcomes?
- Can we predict cancer risk by detecting the expression status of circadian rhythm genes in combination with other markers?
INTRODUCTION
[2,3]. In essence, the circadian clock constitutes an automatic regulatory sequence for the expression, accumulation and degradation of clock gene products, forming an autonomous molecular oscillator [3]. The molecular clock in the animals controls the expression of genes that are output throughout the body, thereby controlling the activity and function of different cells and organs over time [4]. Therefore, mutations in the clock gene can lead to changes in various biological rhythmic behaviors [5].
blocking CLOCK/BMAL1-mediated transcription [3]. Gene mutations that change the phosphorylation state, mobility, or degradation of PER and CRY proteins may affect the clock speed [16, 17]. PER proteins are subjected to successive phosphorylation events of multiple residues by CK1

EFFECT OF CIRCADIAN RHYTHMS ON IMMUNE CELLS Macrophages
Dendritic cells
B lymphocytes
nodes in response to the chemotactic lipid sphingosine 1-phosphate [89].
T lymphocytes
under circadian rhythms, and the rhythmic production of cortisol and catechol amines regulates the migration and localization of T cells. The proliferation of T cells is also influenced by circadian rhythms, showing a faster rate of proliferation at night. These research findings suggest that circadian rhythms have important effects on the activity, migration, and proliferation of T cells, and disruption of circadian rhythms may impact immune responses, including tumor immune responses.
NK cells
THE SLEEP-WAKE CYCLE IS INVOLVED IN INFLUENCING IMMUNE SYSTEM FUNCTION AND DNA DAMAGE REPAIR, AND IS REGULATED BY INFLAMMATION AND CYTOKINE FEEDBACK
Sleep affects the production of cytokines and participates in the regulation of inflammation
production in vitro exhibit a circadian rhythm, with a peak at around 03:00 during nighttime sleep [116, 117]. Sleep, particularly in the early stages dominated by slow-wave sleep (SWS), inhibits glucocorticoid release and promotes growth hormone (GH) and prolactin release [118-120]. These factors shift the Th1/Th2 balance towards Th1 advantage, promoting cell-mediated immune responses against intracellular pathogens [121, 122]. Another study confirmed that nocturnal sleep inhibits Th2 cell production compared to wakefulness, while the effect on Th1 cells remains unclear. Thus, sleep induces a shift towards Th1 dominance [115]. Inhibition of cortisol release and increased melatonin levels at night may contribute to Th1 dominance [116, 123]. However, this effect is limited to early sleep, mainly SWS, and reversed during late sleep dominated by rapid eye movement (REM) sleep, with lower CD4

gastrointestinal syndrome [134], highlighting the importance of BMAL1 in intestinal cell proliferation. Overexpression of CRY1 inhibits vascular inflammation induced by sleep deprivation (SD), possibly through the NF-kB and cyclic adenosine monophosphate/ protein kinase A (cAMP/PKA) pathways in vivo [135]. Acute sleep deprivation (ASD) commonly affects shift workers, causing lethargy and persistent fatigue. ASD contributes to inflammation by influencing the intestinal microbiota. In elderly individuals with insomnia, the impact of physical activity on sleep disorders can be regulated by the gut microbiota’s primary metabolites. The genus Monoglobus shows a negative correlation with physical activity and sleep latency, while exhibiting a positive correlation with sleep
efficiency [136]. Disruption of circadian rhythm leads to decreased abundance of specific bacteria (e.g., Lactobacillus and Ruminococcus) along with downregulation of BMAL1 [137]. Increased Candidatus Arthromitus in the SD group may contribute to colitis, intestinal barrier dysfunction, and systemic inflammation [138]. Thus, circadian rhythm and microbiota composition play crucial roles in maintaining intestinal barrier function. Adequate sleep and self-regulation of the microbiota might have protective effects against disease and help restore the intestinal epithelial barrier, thereby improving inflammation [138].
Sleep deprivation mediates the occurrence of cancer via affecting DNA damage repair and oxidative stress
genes involved in cell cycle checkpoint activation and DNA repair were identified, including ATR, CDK4, CDKN1C, TP53, WEE1, ERCC6, TP53, RPA3, XPA, H2AFX, PARP1, and RAD50. Disruption of the circadian rhythm of these genes’ expression suggests a direct link between cellular circadian rhythm mechanisms and the carcinogenic risk of night shift work [139].
Inflammation, cytokines, and immune cells can provide feedback to regulate sleep

mediate this relationship. However, some studies have not found an association between inflammatory markers and sleep time [211-215], increasing the heterogeneity of research results.
EFFECT OF CIRCADIAN RHYTHM ON TUMORS AND THEIR MICROENVIRONMENT
Circadian rhythm participates in the development of tumors by affecting various kinds of cells
major role.
There is molecular coupling between cell cycle and biological clock, and they are jointly involved in the occurrence and development of cancer
cycle, with evidence of circadian rhythm changes in cyclin abundance and clock gene expression at specific cell cycle stages [264, 265]. The circadian clock controls the expression of CDK and cyclins such as CDK2, CDK4, and cyclin D1, as well as cell cycle inhibitors including Wee1, p21, p27, and p57 [266]. CLOCK-BMAL1 regulates the circadian expression of the G2/M inhibitor WEE1 [267], while the expression of p21 is regulated by antagonistic effects of ROR and REV-ERB proteins [268]. NONO, in association with PER protein, regulates the circadian rhythm regulation of p16 expression [263]. P53 directly binds to the Per2 promoter and inhibits CLOCK-BMAL1-mediated transcription [269].
Metabolism and multiple factors in tumor microenvironment are regulated by biological rhythm
signal factors in tumor cells. Knockout of clock proteins Cry1 and Cry2 leads to a constitutive increase in pro-inflammatory cytokines via NF-кB and PKA signals, indicating direct regulation of cytokine expression by circadian genes [75]. Deletion of myeloid-specific circadian rhythm gene disrupts chemokine expression rhythm and Ly6Chi monocyte tissue recruitment [242]. Adhesion molecules and chemokines in endothelial cells oscillate in a circadian rhythm, affecting leukocyte adhesion and exosmosis [273].
| Clock gene | Cell | Effects | Ref. | |||
| BMAL1
|
Macrophage |
|
[71, 72] | |||
| GAM (M2 subtype) |
|
[81] | ||||
| Endothelial cell | Time difference in the expression of migration-promoting molecules was eliminated | [83] | ||||
| CLOCK
|
Endothelial cell |
|
[84] | |||
| CRC cells |
|
[91] | ||||
| CRY
|
Endothelial cell |
|
[85] | |||
| Tumor cell |
|
[32] | ||||
| ROR
|
|
|
[89] | |||
|
|
Th17 cell, Treg |
|
[88] | |||
| REV-ERB
|
Macrophage |
|
[75] |
SUMMARY
clock genes offers new avenues for cancer treatment, and various targets (e.g., CLK8 [290], mTOR [291]) and drugs (e.g., MLN4924 [292]) are under development with ongoing clinical trials.
DATA AVAILABILITY
REFERENCES
- Scheiermann C, Gibbs J, Ince L, Loudon A. Clocking in to immunity. Nat Rev Immunol. 2018;18:423-37.
- Zhou L, Zhang Z, Nice E, Huang C, Zhang W, Tang Y. Circadian rhythms and cancers: the intrinsic links and therapeutic potentials. J Hematol Oncol. 2022;15:21.
- Patke A, Young MW, Axelrod S. Molecular mechanisms and physiological importance of circadian rhythms. Nat Rev Mol Cell Biol. 2020;21:67-84.
- Zhang R, Lahens NF, Ballance HI, Hughes ME, Hogenesch JB. A circadian gene expression atlas in mammals: implications for biology and medicine. Proc Natl Acad Sci USA. 2014;111:16219-24.
- Young MW, Kay SA. Time zones: a comparative genetics of circadian clocks. Nat Rev Genet. 2001;2:702-15.
- Fulgham CV, Dreyer AP, Nasseri A, Miller AN, Love J, Martin MM, et al. Central and peripheral clock control of circadian feeding rhythms. Biol Rhythms. 2021;36:548-66.
- Aton SJ, Herzog ED. Come together, right…now: synchronization of rhythms in a mammalian circadian clock. Neuron. 2005;48:531-4.
- Schibler U, Gotic I, Saini C, Gos P, Curie T, Emmenegger Y, et al. Clock-Talk: Interactions between Central and Peripheral Circadian Oscillators in Mammals. Cold Spring Harb Symp Quant Biol. 2015;80:223-32.
- Welsh DK, Yoo SH, Liu AC, Takahashi JS, Kay SA. Bioluminescence imaging of individual fibroblasts reveals persistent, independently phased circadian rhythms of clock gene expression. Curr Biol. 2004;14:2289-95.
- Cajochen C. Alerting effects of light. Sleep Med Rev. 2007;11:453-64.
- K Pavlova M, Latreille V. Sleep disorders. Am J Med. 2019;132:292-9.
- Ito
, Inoue . The international classification of sleep disorders, third edition. American Academy of Sleep Medicine. Includes bibliographies and index. Nihon Rinsho. 2015;73:916-23. - Sun SY, Chen GH. Treatment of circadian rhythm sleep-wake disorders. Curr Neuropharmacol. 2022;20:1022-34.
- Nagtegaal JE, Laurant MW, Kerkhof GA, Smits MG, van der Meer YG, Coenen AM. Effects of melatonin on the quality of life in patients with delayed sleep phase syndrome. J Psychosom Res. 2000;48:45-50.
- Yagita K, Tamanini F, Yasuda M, Hoeijmakers JH, van der Horst GT, Okamura H. Nucleocytoplasmic shuttling and mCRY-dependent inhibition of ubiquitylation of the mPER2 clock protein. EMBO J. 2002;21:1301-14.
- Guilding C, Scott F, Bechtold DA, Brown TM, Wegner S, Piggins HD. Suppressed cellular oscillations in after-hours mutant mice are associated with enhanced circadian phase-resetting. J Physiol. 2013;591:1063-80.
- Meng QJ, Logunova L, Maywood ES, Gallego M, Lebiecki J, Brown TM, et al. Setting clock speed in mammals: the CK1 epsilon tau mutation in mice accelerates circadian pacemakers by selectively destabilizing PERIOD proteins. Neuron. 2008;58:78-88.
- Eide EJ, Woolf MF, Kang H, Woolf P, Hurst W, Camacho F, et al. Control of mammalian circadian rhythm by CKlepsilon-regulated proteasome-mediated PER2 degradation. Mol Cell Biol. 2005;25:2795-807.
- Lowrey PL, Shimomura K, Antoch MP, Yamazaki S, Zemenides PD, Ralph MR, et al. Positional syntenic cloning and functional characterization of the mammalian circadian mutation tau. Science. 2000;288:483-92.
- Maier B, Wendt S, Vanselow JT, Wallach T, Reischl S, Oehmke S, et al. A largescale functional RNAi screen reveals a role for CK2 in the mammalian circadian clock. Genes Dev. 2009;23:708-18.
- Toh KL, Jones CR, He Y, Eide EJ, Hinz WA, Virshup DM, et al. An hPer2 phosphorylation site mutation in familial advanced sleep phase syndrome. Science. 2001;291:1040-3.
- Tsuchiya Y, Akashi M, Matsuda M, Goto K, Miyata Y, Node K, et al. Involvement of the protein kinase CK2 in the regulation of mammalian circadian rhythms. Sci Signal. 2009;2:ra26.
- Vanselow K, Vanselow JT, Westermark PO, Reischl S, Maier B, Korte T, et al. Differential effects of PER2 phosphorylation: molecular basis for the human familial advanced sleep phase syndrome (FASPS). Genes Dev. 2006;20:2660-72.
- Xu Y, Toh KL, Jones CR, Shin JY, Fu YH, Ptácek LJ. Modeling of a human circadian mutation yields insights into clock regulation by PER2. Cell. 2007;128:59-70.
- Zhou M, Kim JK, Eng GW, Forger DB, Virshup DM. A Period2 phosphoswitch regulates and temperature compensates circadian period. Mol Cell. 2015;60:77-88.
- Ohsaki K, Oishi K, Kozono Y, Nakayama K, Nakayama KI, Ishida N. The role of
P1 and P2 in circadian rhythm generation by mediating degradation of clock protein PER2. J Biochem. 2008;144:609-18. - Shirogane T, Jin J, Ang XL, Harper JW. SCFbeta-TRCP controls clock-dependent transcription via casein kinase 1-dependent degradation of the mammalian period-1 (Per1) protein. J Biol Chem. 2005;280:26863-72.
- Narasimamurthy R, Hunt SR, Lu Y, Fustin JM, Okamura H, Partch CL, et al. CK1
protein kinase primes the PER2 circadian phosphoswitch. Proc Natl Acad Sci USA. 2018;115:5986-91. - Gao P, Yoo SH, Lee KJ, Rosensweig C, Takahashi JS, Chen BP, et al. Phosphorylation of the cryptochrome 1 C-terminal tail regulates circadian period length. J Biol Chem. 2013;288:35277-86.
- Hirano A, Shi G, Jones CR, Lipzen A, Pennacchio LA, Xu Y, et al. A Cryptochrome 2 mutation yields advanced sleep phase in humans. Elife. 2016;5:e16695.
- Hirota T, Lee JW, St John PC, Sawa M, Iwaisako K, Noguchi T, et al. Identification of small molecule activators of cryptochrome. Science 2012;337:1094-7.
- Khan SK, Xu H, Ukai-Tadenuma M, Burton B, Wang Y, Ueda HR, et al. Identification of a novel cryptochrome differentiating domain required for feedback repression in circadian clock function. J Biol Chem. 2012;287:25917-26.
- Ode KL, Ukai H, Susaki EA, Narumi R, Matsumoto K, Hara J, et al. Knockout-rescue embryonic stem cell-derived mouse reveals circadian-period control by quality and quantity of CRY1. Mol Cell. 2017;65:176-90.
- Oshima T, Yamanaka I, Kumar A, Yamaguchi J, Nishiwaki-Ohkawa T, Muto K, et al. C-H activation generates period-shortening molecules that target cryptochrome in the mammalian circadian clock. Angew Chem Int Ed Engl. 2015;54:7193-7.
- Patke A, Murphy PJ, Onat OE, Krieger AC, Özçelik T, Campbell SS, et al. Mutation of the human circadian clock gene CRY1 in familial delayed sleep phase disorder. Cell. 2017;169:203-15.e13.
- Hirano A, Braas D, Fu YH, Ptáček LJ. FAD regulates CRYPTOCHROME protein stability and circadian clock in mice. Cell Rep. 2017;19:255-66.
- Busino L, Bassermann F, Maiolica A, Lee C, Nolan PM, Godinho SI, et al. SCFFbxl3 controls the oscillation of the circadian clock by directing the degradation of cryptochrome proteins. Science. 2007;316:900-4.
- Godinho SI, Maywood ES, Shaw L, Tucci V, Barnard AR, Busino L, et al. The afterhours mutant reveals a role for Fbxl3 in determining mammalian circadian period. Science. 2007;316:897-900.
- Hirano A, Yumimoto K, Tsunematsu R, Matsumoto M, Oyama M, Kozuka-Hata H, et al. FBXL21 regulates oscillation of the circadian clock through ubiquitination and stabilization of cryptochromes. Cell. 2013;152:1106-18.
- Saran AR, Kalinowska D, Oh S, Janknecht R, DiTacchio L. JMJD5 links CRY1 function and proteasomal degradation. PLoS Biol. 2018;16:e2006145.
- Siepka SM, Yoo SH, Park J, Song W, Kumar V, Hu Y, et al. Circadian mutant overtime reveals F-box protein FBXL3 regulation of cryptochrome and period gene expression. Cell. 2007;129:1011-23.
- Yoo SH, Mohawk JA, Siepka SM, Shan Y, Huh SK, Hong HK, et al. Competing E3 ubiquitin ligases govern circadian periodicity by degradation of CRY in nucleus and cytoplasm. Cell. 2013;152:1091-105.
- Zhang Y, Fang B, Emmett MJ, Damle M, Sun Z, Feng D, et al. Discrete functions of nuclear receptor Rev-erba couple metabolism to the clock. Science. 2015;348:1488-92.
- Cho H, Zhao X, Hatori M, Yu RT, Barish GD, Lam MT, et al. Regulation of circadian behaviour and metabolism by REV-ERB-a and REV-ERB-
. Nature. 2012;485:123-7. - Chung S, Lee EJ, Yun S, Choe HK, Park SB, Son HJ, et al. Impact of circadian nuclear receptor REV-ERBa on midbrain dopamine production and mood regulation. Cell. 2014;157:858-68.
- Takahashi JS. Transcriptional architecture of the mammalian circadian clock. Nat Rev Genet. 2017;18:164-79.
- Lowrey PL, Takahashi JS. Mammalian circadian biology: elucidating genomewide levels of temporal organization. Annu Rev Genomics Hum Genet. 2004;5:407-41.
- Preitner N, Damiola F, Lopez-Molina L, Zakany J, Duboule D, Albrecht U, et al. The orphan nuclear receptor REV-ERBalpha controls circadian transcription within the positive limb of the mammalian circadian oscillator. Cell. 2002;110:251-60.
- Shearman LP, Sriram S, Weaver DR, Maywood ES, Chaves I, Zheng B, et al. Interacting molecular loops in the mammalian circadian clock. Science. 2000;288:1013-9.
- Farshadi E, van der Horst G, Chaves I. Molecular links between the circadian clock and the cell cycle. J Mol Biol. 2020;432:3515-24.
- He B, Nohara K, Park N, Park YS, Guillory B, Zhao Z, et al. The small molecule nobiletin targets the molecular oscillator to enhance circadian rhythms and protect against metabolic syndrome. Cell Metab. 2016;23:610-21.
- Shinozaki A, Misawa K, Ikeda Y, Haraguchi A, Kamagata M, Tahara Y, et al. Potent effects of flavonoid nobiletin on amplitude, period, and phase of the circadian clock rhythm in PER2::LUCIFERASE mouse embryonic fibroblasts. PLoS ONE. 2017;12:e0170904.
- Onozuka H, Nakajima A, Matsuzaki K, Shin RW, Ogino K, Saigusa D, et al. Nobiletin, a citrus flavonoid, improves memory impairment and Abeta pathology in a transgenic mouse model of Alzheimer’s disease. J Pharm Exp Ther. 2008;326:739-44.
- Yabuki Y, Ohizumi Y, Yokosuka A, Mimaki Y, Fukunaga K. Nobiletin treatment improves motor and cognitive deficits seen in MPTP-induced Parkinson model mice. Neuroscience. 2014;259:126-41.
- Yi LT, Xu HL, Feng J, Zhan X, Zhou LP, Cui CC. Involvement of monoaminergic systems in the antidepressant-like effect of nobiletin. Physiol Behav. 2011;102:1-6.
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646-74.
- Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57-70.
- Vidya MK, Kumar VG, Sejian V, Bagath M, Krishnan G, Bhatta R. Toll-like receptors: significance, ligands, signaling pathways, and functions in mammals. Int Rev Immunol. 2018;37:20-36.
- Halberg F , johnson EA, brown BW, bittner JJ. Susceptibility rhythm to E. coli endotoxin and bioassay. Proc Soc Exp Biol Med. 1960;103:142-4.
- Silver AC, Buckley SM, Hughes ME, Hastings AK, Nitabach MN, Fikrig E. Daily oscillations in expression and responsiveness of Toll-like receptors in splenic immune cells. Heliyon. 2018;4:e00579.
- Kelly B, O’Neill LA. Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res. 2015;25:771-84.
- Huang SC, Smith AM, Everts B, Colonna M, Pearce EL, Schilling JD, et al. Metabolic reprogramming mediated by the mTORC2-IRF4 signaling axis is essential for macrophage alternative activation. Immunity. 2016;45:817-30.
- Timmons GA, O’Siorain JR, Kennedy OD, Curtis AM, Early JO. Innate rhythms: clocks at the center of monocyte and macrophage function. Front Immunol. 2020;11:1743.
- Xu H, Li H, Woo SL, Kim SM, Shende VR, Neuendorff N, et al. Myeloid cell-specific disruption of Period1 and Period2 exacerbates diet-induced inflammation and insulin resistance. J Biol Chem. 2014;289:16374-88.
- Liu Z, Gan L, Zhang T, Ren Q, Sun C. Melatonin alleviates adipose inflammation through elevating a-ketoglutarate and diverting adipose-derived exosomes to macrophages in mice. J Pineal Res. 2018;64.
- Ding
, Lin N, Sheng X, Zhao Y, Su Y, Xu L, et al. Melatonin stabilizes ruptureprone vulnerable plaques via regulating macrophage polarization in a nuclear circadian receptor RORa-dependent manner. J Pineal Res. 2019;67:e12581. - Allen NC, Philip NH, Hui L, Zhou X, Franklin RA, Kong Y, et al. Desynchronization of the molecular clock contributes to the heterogeneity of the inflammatory response. Sci Signal. 2019;12:eaau1851.
- Pariollaud M, Gibbs JE, Hopwood TW, Brown S, Begley N, Vonslow R, et al. Circadian clock component REV-ERBa controls homeostatic regulation of pulmonary inflammation. J Clin Invest. 2018;128:2281-96.
- Gibbs JE, Blaikley J, Beesley S, Matthews L, Simpson KD, Boyce SH, et al. The nuclear receptor REV-ERBa mediates circadian regulation of innate immunity through selective regulation of inflammatory cytokines. Proc Natl Acad Sci USA. 2012;109:582-7.
- Griffin P, Dimitry JM, Sheehan PW, Lananna BV, Guo C, Robinette ML, et al. Circadian clock protein Rev-erba regulates neuroinflammation. Proc Natl Acad Sci USA. 2019;116:5102-7.
- Wang S, Lin Y, Yuan X, Li F, Guo L, Wu B. REV-ERBa integrates colon clock with experimental colitis through regulation of NF-kB/NLRP3 axis. Nat Commun. 2018;9:4246.
- Pourcet B, Zecchin M, Ferri L, Beauchamp J, Sitaula S, Billon C, et al. Nuclear receptor subfamily 1 group D member 1 regulates circadian activity of NLRP3 inflammasome to reduce the severity of fulminant hepatitis in mice. Gastroenterology. 2018;154:1449-64.e20.
- Wang T, Wang Z, Yang P, Xia L, Zhou M, Wang S, et al. PER1 prevents excessive innate immune response during endotoxin-induced liver injury through regulation of macrophage recruitment in mice. Cell Death Dis. 2016;7:e2176.
- Silver AC, Arjona A, Walker WE, Fikrig E. The circadian clock controls toll-like receptor 9-mediated innate and adaptive immunity. Immunity. 2012;36:251-61.
- Narasimamurthy R, Hatori M, Nayak SK, Liu F, Panda S, Verma IM. Circadian clock protein cryptochrome regulates the expression of proinflammatory cytokines. Proc Natl Acad Sci USA. 2012;109:12662-7.
- Yu D, Fang X, Xu Y, Xiao H, Huang T, Zhang Y, et al. Rev-erba can regulate the NF-кB/NALP3 pathway to modulate lipopolysaccharide-induced acute lung injury and inflammation. Int Immunopharmacology. 2019;73:312-20.
- McRae HM, Hargreaves DC. Old macrophages lose their (circadian) rhythm. Trends Immunol. 2022;43:265-7.
- Amir M, Campbell S, Kamenecka TM, Solt LA. Pharmacological modulation and genetic deletion of REV-ERBa and REV-ERB
regulates dendritic cell development. Biochem Biophys Res Commun. 2020;527:1000-7. - Nobis CC, Dubeau Laramée G, Kervezee L, Maurice De Sousa D, Labrecque N, Cermakian N. The circadian clock of CD8 T cells modulates their early response to vaccination and the rhythmicity of related signaling pathways. Proc Natl Acad Sci USA. 2019;116:20077-86.
- Holtkamp SJ, Ince LM, Barnoud C, Schmitt MT, Sinturel F, Pilorz V, et al. Circadian clocks guide dendritic cells into skin lymphatics. Nat Immunol. 2021;22:1375-81.
- Wang C, Barnoud C, Cenerenti M, Sun M, Caffa I, Kizil B, et al. Dendritic cells direct circadian anti-tumour immune responses. Nature. 2023;614:136-43.
- Sun Y, Yang Z, Niu Z, Peng J, Li Q, Xiong W, et al. MOP3, a component of the molecular clock, regulates the development of
cells. Immunology. 2006;119:451-60. - Hemmers S, Rudensky AY. The Cell-Intrinsic Circadian Clock Is Dispensable for Lymphocyte Differentiation and Function. Cell Rep. 2015;11:1339-49.
- Druzd D, Matveeva O, Ince L, Harrison U, He W, Schmal C, et al. Lymphocyte Circadian Clocks Control Lymph Node Trafficking and Adaptive Immune Responses. Immunity. 2017;46:120-32.
- Suzuki K, Hayano Y, Nakai A, Furuta F, Noda M. Adrenergic control of the adaptive immune response by diurnal lymphocyte recirculation through lymph nodes. J Exp Med. 2016;213:2567-74.
- Shimba A, Cui G, Tani-Ichi S, Ogawa M, Abe S, Okazaki F, et al. Glucocorticoids drive diurnal oscillations in
cell distribution and responses by inducing interleukin-7 receptor and CXCR4. Immunity. 2018;48:286-98.e6. - Förster R, Davalos-Misslitz AC, Rot A. CCR7 and its ligands: balancing immunity and tolerance. Nat Rev Immunol. 2008;8:362-71.
- Stein JV, Nombela-Arrieta C. Chemokine control of lymphocyte trafficking: a general overview. Immunology. 2005;116:1-12.
- Cyster JG, Schwab SR. Sphingosine-1-phosphate and lymphocyte egress from lymphoid organs. Annu Rev Immunol. 2012;30:69-94.
- Wang Q, Li L, Li C, Cao H, Chen Y, Zhou W, et al. Circadian protein CLOCK modulates regulatory
cell functions of nurses engaging day-night shift rotation. Cell Signal. 2022;96:110362. - Cermakian N, Labrecque N. Regulation of cytotoxic CD8 +T cells by the circadian clock. J Immunol. 2023;210:12-18.
- Besedovsky L, Born J, Lange T. Endogenous glucocorticoid receptor signaling drives rhythmic changes in human T-cell subset numbers and the expression of the chemokine receptor CXCR4. FASEB J. 2014;28:67-75.
- Dimitrov S, Benedict C, Heutling D, Westermann J, Born J, Lange T. Cortisol and epinephrine control opposing circadian rhythms in T cell subsets. Blood. 2009;113:5134-43.
- Deprés-Brummer P, Bourin P, Pages N, Metzger G, Lévi F. Persistent T lymphocyte rhythms despite suppressed circadian clock outputs in rats. Am J Physiol. 1997;273:R1891.
- Kawate T, Abo T, Hinuma S, Kumagai K. Studies of the bioperiodicity of the immune response. II. Co-variations of murine T and B cells and a role of corticosteroid. J Immunol. 1981;126:1364-7.
- Fortier EE, Rooney J, Dardente H, Hardy MP, Labrecque N, Cermakian N. Circadian variation of the response of T cells to antigen. J Immunol. 2011;187:6291-6300.
- Wu Y, Tao B, Zhang T, Fan Y, Mao R. Pan-cancer analysis reveals disrupted circadian clock associates with T cell exhaustion. Front Immunol. 2019;10:2451.
- Capelle CM, Chen A, Zeng N, Baron A, Grzyb K, Arns T, et al. Stress hormone signalling inhibits Th1 polarization in a CD4 T-cell-intrinsic manner via mTORC1 and the circadian gene PER1. Immunology. 2022;165:428-44.
- Yang G, Yang Y, Tang H, Yang K. Loss of the clock gene Per1 promotes oral squamous cell carcinoma progression via the AKT/mTOR pathway. Cancer Sci. 2020;111:1542-54.
- Juszczak M, Głabiński A. Th17 cells in the pathogenesis of multiple sclerosis. Postepy Hig Med Dosw. 2009;63:492-501.
- Yu X, Rollins D, Ruhn KA, Stubblefield JJ, Green CB, Kashiwada M, et al. TH17 cell differentiation is regulated by the circadian clock. Science. 2013;342:727-30.
- Chang JL, Qiu J. Regulation of ILC3/Th17-mediated intestinal immune response by circadian rhythm. Sichuan Da Xue Xue Bao Yi Xue Ban. 2022;53:15-19.
- Cheng FL, An YF, Xue JM, Wang YJ, Ding XW, Zhang YT, et al. Circadian rhythm disruption exacerbates Th2-like immune response in murine allergic airway inflammation. Int Forum Allergy Rhinol. 2022;12:757-70.
- Mandal A, Viswanathan C. Natural killer cells: in health and disease. Hematol Oncol Stem Cell Ther. 2015;8:47-55.
- Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S. Functions of natural killer cells. Nat Immunol. 2008;9:503-10.
- Mattiola I, Pesant M, Tentorio PF, Molgora M, Marcenaro E, Lugli E, et al. Priming of human resting NK cells by autologous M1 macrophages via the engagement of IL-1
, IFN- , and IL-15 pathways. J Immunol. 2015;195:2818-28. - Logan RW, Sarkar DK. Circadian nature of immune function. Mol Cell Endocrinol. 2012;349:82-90.
- Zeng X, Liang C, Yao J. Chronic shift-lag promotes NK cell ageing and impairs immunosurveillance in mice by decreasing the expression of CD122. J. Cell Mol Med. 2020;24:14583-95.
- Kennedy MK, Glaccum M, Brown SN, Butz EA, Viney JL, Embers M, et al. Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. J Exp Med. 2000;191:771-80.
- Chung JW, Yoon SR, Choi I. The regulation of NK cell function and development. Front Biosci. 2008;13:6432-42.
- Benca RM, Quintas J. Sleep and host defenses: a review. Sleep. 1997;20:1027-37.
- Besedovsky L, Lange T, Born J. Sleep and immune function. Pflug Arch. 2012;463:121-37.
- Lange T, Perras B, Fehm HL, Born J. Sleep enhances the human antibody response to hepatitis A vaccination. Psychosom Med. 2003;65:831-5.
- Spiegel K, Sheridan JF. Van Cauter E. Effect of sleep deprivation on response to immunization. JAMA. 2002;288:1471-2.
- Dimitrov S, Lange T, Tieken S, Fehm HL, Born J. Sleep associated regulation of T helper
helper 2 cytokine balance in humans. Brain Behav Immun. 2004;18:341-8. - Petrovsky N, Harrison LC. Diurnal rhythmicity of human cytokine production: a dynamic disequilibrium in T helper cell type
helper cell type 2 balance. J Immunol. 1997;158:5163-8. - Petrovsky N, Harrison LC. The chronobiology of human cytokine production. Int Rev Immunol. 1998;16:635-49.
- Born J, Fehm HL. Hypothalamus-pituitary-adrenal activity during human sleep: a coordinating role for the limbic hippocampal system. Exp Clin Endocrinol Diabetes. 1998;106:153-63.
- Sadamatsu M, Kato N, lida H, Takahashi S, Sakaue K, Takahashi K, et al. The 24hour rhythms in plasma growth hormone, prolactin and thyroid stimulating hormone: effect of sleep deprivation. J Neuroendocrinol. 1995;7:597-606.
- Spiegel K, Luthringer R, Follenius M, Schaltenbrand N, Macher JP, Muzet A, et al. Temporal relationship between prolactin secretion and slow-wave electroencephalic activity during sleep. Sleep. 1995;18:543-8.
- Alexander J, Satoskar AR, Russell DG. Leishmania species: models of intracellular parasitism. J Cell Sci. 1999;112:2993-3002.
- Karupiah G. Type 1 and type 2 cytokines in antiviral defense. Vet Immunol Immunopathol. 1998;63:105-9.
- Kühlwein E, Irwin M. Melatonin modulation of lymphocyte proliferation and Th1/Th2 cytokine expression. J Neuroimmunol. 2001;117:51-57.
- Lio D, Balistreri CR, Candore G, D’Anna C, Di Lorenzo G, Gervasi F, et al. In vitro treatment with interleukin-2 normalizes type-1 cytokine production by lymphocytes from elderly. Immunopharmacol Immunotoxicol. 2000;22:195-203.
- Van Cauter E, Leproult R, Plat L. Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA. 2000;284:861-8.
- Höflich C, Döcke WD, Busch A, Kern F, Volk HD. CD45RA(bright)/CD11a(bright) CD8+ T cells: effector T cells. Int Immunol. 1998;10:1837-45.
- von Andrian UH, Mackay CR. T-cell function and migration. Two sides of the same coin. N Engl J Med. 2000;343:1020-34.
- Irwin MR, Wang M, Campomayor CO, Collado-Hidalgo A, Cole S. Sleep deprivation and activation of morning levels of cellular and genomic markers of inflammation. Arch Intern Med. 2006;166:1756-62.
- Phillips B, Mannino DM. Does insomnia kill. Sleep. 2005;28:965-71.
- Vgontzas AN, Zoumakis E, Bixler EO, Lin HM, Follett H, Kales A, et al. Adverse effects of modest sleep restriction on sleepiness, performance, and inflammatory cytokines. J Clin Endocrinol Metab. 2004;89:2119-26.
- Shearer WT, Reuben JM, Mullington JM, Price NJ, Lee BN, Smith EO, et al. Soluble TNF-alpha receptor 1 and IL-6 plasma levels in humans subjected to the sleep deprivation model of spaceflight. J Allergy Clin Immunol. 2001;107:165-70.
- Meier-Ewert HK, Ridker PM, Rifai N, Regan MM, Price NJ, Dinges DF, et al. Effect of sleep loss on C-reactive protein, an inflammatory marker of cardiovascular risk. J Am Coll Cardiol. 2004;43:678-83.
- Godinho-Silva C, Domingues RG, Rendas M, Raposo B, Ribeiro H, da Silva JA, et al. Light-entrained and brain-tuned circadian circuits regulate ILC3s and gut homeostasis. Nature. 2019;574:254-8.
- Stokes K, Cooke A, Chang H, Weaver DR, Breault DT, Karpowicz P. The circadian clock gene BMAL1 coordinates intestinal regeneration. Cell Mol Gastroenterol Hepatol. 2017;4:95-114.
- Qin B, Deng Y. Overexpression of circadian clock protein cryptochrome (CRY) 1 alleviates sleep deprivation-induced vascular inflammation in a mouse model. Immunol Lett. 2015;163:76-83.
- Magzal F, Shochat T, Haimov I, Tamir S, Asraf K, Tuchner-Arieli M, et al. Increased physical activity improves gut microbiota composition and reduces short-chain fatty acid concentrations in older adults with insomnia. Sci Rep. 2022;12:2265.
- Thaiss CA, Zeevi D, Levy M, Zilberman-Schapira G, Suez J, Tengeler AC, et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell. 2014;159:514-29.
- Yang DF, Huang WC, Wu CW, Huang CY, Yang Y, Tung YT. Acute sleep deprivation exacerbates systemic inflammation and psychiatry disorders through gut microbiota dysbiosis and disruption of circadian rhythms. Microbiol Res. 2023;268:127292.
- Koritala B, Porter KI, Arshad OA, Gajula RP, Mitchell HD, Arman T, et al. Night shift schedule causes circadian dysregulation of DNA repair genes and elevated DNA damage in humans. J Pineal Res. 2021;70:e12726.
- Haus EL, Smolensky MH. Shift work and cancer risk: potential mechanistic roles of circadian disruption, light at night, and sleep deprivation. Sleep Med Rev. 2013;17:273-84.
- James SM, Honn KA, Gaddameedhi S, Van Dongen H. Shift work: disrupted circadian rhythms and sleep-implications for health and well-being. Curr Sleep Med Rep. 2017;3:104-12.
- Savvidis C, Koutsilieris M. Circadian rhythm disruption in cancer biology. Mol Med. 2012;18:1249-60.
- Sahar S, Sassone-Corsi P. Metabolism and cancer: the circadian clock connection. Nat Rev Cancer. 2009;9:886-96.
- Sancar A, Lindsey-Boltz LA, Gaddameedhi S, Selby CP, Ye R, Chiou YY, et al. Circadian clock, cancer, and chemotherapy. Biochemistry. 2015;54:110-23.
- Yang Y, Liu Z, Selby CP, Sancar A. Long-term, genome-wide kinetic analysis of the effect of the circadian clock and transcription on the repair of cisplatin-DNA adducts in the mouse liver. J Biol Chem. 2019;294:11960-8.
- Gaucher J, Montellier E, Sassone-Corsi P. Molecular cogs: interplay between circadian clock and cell cycle. Trends Cell Biol. 2018;28:368-79.
- Miller BH, McDearmon EL, Panda S, Hayes KR, Zhang J, Andrews JL, et al. Circadian and CLOCK-controlled regulation of the mouse transcriptome and cell proliferation. Proc Natl Acad Sci USA. 2007;104:3342-7.
- Scheiermann C, Kunisaki Y, Frenette PS. Circadian control of the immune system. Nat Rev Immunol. 2013;13:190-8.
- Keller M, Mazuch J, Abraham U, Eom GD, Herzog ED, Volk HD, et al. A circadian clock in macrophages controls inflammatory immune responses. Proc Natl Acad Sci USA. 2009;106:21407-12.
- Masri S, Cervantes M, Sassone-Corsi P. The circadian clock and cell cycle: interconnected biological circuits. Curr Opin Cell Biol. 2013;25:730-4.
- Roenneberg T, Merrow M. The circadian clock and human health. Curr Biol. 2016;26:R432-43.
- Depner CM, Melanson EL, McHill AW, Wright KP Jr. Mistimed food intake and sleep alters 24-hour time-of-day patterns of the human plasma proteome. Proc Natl Acad Sci USA 2018;115:E5390-9.
- Kervezee L, Cuesta M, Cermakian N, Boivin DB. Simulated night shift work induces circadian misalignment of the human peripheral blood mononuclear cell transcriptome. Proc Natl Acad Sci USA. 2018;115:5540-5.
- Skene DJ, Skornyakov E, Chowdhury NR, Gajula RP, Middleton B, Satterfield BC, et al. Separation of circadian- and behavior-driven metabolite rhythms in humans provides a window on peripheral oscillators and metabolism. Proc Natl Acad Sci USA. 2018;115:7825-30.
- Resuehr D, Wu G, Johnson RL Jr, Young ME, Hogenesch JB, Gamble KL. Shift work disrupts circadian regulation of the transcriptome in hospital nurses. J Biol Rhythms. 2019;34:167-77.
- Humans, IMotloCHt, Work iNS. Night shift work. 2020. Lyon (FR). other.
- Papagiannakopoulos T, Bauer MR, Davidson SM, Heimann M, Subbaraj L, Bhutkar A, et al. Circadian rhythm disruption promotes lung tumorigenesis. Cell Metab. 2016;24:324-31.
- Lee Y, Lahens NF, Zhang S, Bedont J, Field JM, Sehgal A. G1/S cell cycle regulators mediate effects of circadian dysregulation on tumor growth and provide targets for timed anticancer treatment. PLoS Biol. 2019;17:e3000228.
- Van Dycke KC, Rodenburg W, van Oostrom CT, van Kerkhof LW, Pennings JL, Roenneberg T, et al. Chronically alternating light cycles increase breast cancer risk in mice. Curr Biol. 2015;25:1932-7.
- Aiello I, Fedele M, Román F, Marpegan L, Caldart C, Chiesa JJ, et al. Circadian disruption promotes tumor-immune microenvironment remodeling favoring tumor cell proliferation. Sci Adv. 2020;6:eaaz4530.
- Bentivoglio M, Grassi-Zucconi G. The pioneering experimental studies on sleep deprivation. Sleep. 1997;20:570-6.
- Rechtschaffen A, Gilliland MA, Bergmann BM, Winter JB. Physiological correlates of prolonged sleep deprivation in rats. Science. 1983;221:182-4.
- Shaw PJ, Tononi G, Greenspan RJ, Robinson DF. Stress response genes protect against lethal effects of sleep deprivation in Drosophila. Nature. 2002;417:287-91.
- Stephenson R, Chu KM, Lee J. Prolonged deprivation of sleep-like rest raises metabolic rate in the Pacific beetle cockroach, Diploptera punctata (Eschscholtz). J Exp Biol. 2007;210:2540-7.
- Donlea JM. Roles for sleep in memory: insights from the fly. Curr Opin Neurobiol. 2019;54:120-6.
- Killgore WD. Effects of sleep deprivation on cognition. Prog Brain Res. 2010;185:105-29.
- Krause AJ, Simon EB, Mander BA, Greer SM, Saletin JM, Goldstein-Piekarski AN, et al. The sleep-deprived human brain. Nat Rev Neurosci. 2017;18:404-18.
- Reimund E. The free radical flux theory of sleep. Med Hypotheses. 1994;43:231-3.
- Alzoubi KH, Khabour OF, Rashid BA, Damaj IM, Salah HA. The neuroprotective effect of vitamin E on chronic sleep deprivation-induced memory impairment: the role of oxidative stress. Behav Brain Res. 2012;226:205-10.
- Hill VM, O’Connor RM, Sissoko GB, Irobunda IS, Leong S, Canman JC, et al. A bidirectional relationship between sleep and oxidative stress in Drosophila. PLoS Biol. 2018;16:e2005206.
- Kanazawa L, Vecchia DD, Wendler EM, Hocayen P, Dos Reis Lívero FA, Stipp MC, et al. Quercetin reduces manic-like behavior and brain oxidative stress induced by paradoxical sleep deprivation in mice. Free Radic Biol Med. 2016;99:79-86.
- Süer C, Dolu N, Artis AS, Sahin L, Yilmaz A, Cetin A. The effects of long-term sleep deprivation on the long-term potentiation in the dentate gyrus and brain oxidation status in rats. Neurosci Res. 2011;70:71-77.
- Villafuerte G, Miguel-Puga A, Rodríguez EM, Machado S, Manjarrez E, AriasCarrión O. Sleep deprivation and oxidative stress in animal models: a systematic review. Oxid Med Cell Longev. 2015;2015:234952.
- Vaccaro A, Kaplan Dor Y, Nambara K, Pollina EA, Lin C, Greenberg ME, et al. Sleep loss can cause death through accumulation of reactive oxygen species in the gut. Cell. 2020;181:1307-28.e15.
- Balaban RS, Nemoto S, Finkel T. Mitochondria, oxidants, and aging. Cell. 2005;120:483-95.
- Shimizu Y, Hendershot LM. Oxidative folding: cellular strategies for dealing with the resultant equimolar production of reactive oxygen species. Antioxid Redox Signal. 2009;11:2317-31.
- Cao SS, Kaufman RJ. Endoplasmic reticulum stress and oxidative stress in cell fate decision and human disease. Antioxid Redox Signal. 2014;21:396-413.
- Santos CX, Tanaka LY, Wosniak J, Laurindo FR. Mechanisms and implications of reactive oxygen species generation during the unfolded protein response: roles of endoplasmic reticulum oxidoreductases, mitochondrial electron transport, and NADPH oxidase. Antioxid Redox Signal. 2009;11:2409-27.
- Zeeshan HM, Lee GH, Kim HR, Chae HJ. Endoplasmic Reticulum Stress and Associated ROS. Int J Mol Sci. 2016;17:327.
- Cirelli C, Faraguna U, Tononi G. Changes in brain gene expression after longterm sleep deprivation. J Neurochem. 2006;98:1632-45.
- Eiland MM, Ramanathan L, Gulyani S, Gilliland M, Bergmann BM, Rechtschaffen A, et al. Increases in amino-cupric-silver staining of the supraoptic nucleus after sleep deprivation. Brain Res. 2002;945:1-8.
- Gopalakrishnan A, Ji LL, Cirelli C. Sleep deprivation and cellular responses to oxidative stress. Sleep. 2004;27:27-35.
- Hipólide DC, D’Almeida V, Raymond R, Tufik S, Nobrega JN. Sleep deprivation does not affect indices of necrosis or apoptosis in rat brain. Int J Neurosci. 2002;112:155-66.
- Brown MK, Strus E, Naidoo N. Reduced sleep during social isolation leads to cellular stress and induction of the unfolded protein response. Sleep. 2017;40:zsx095.
- Nikonova EV, Naidoo N, Zhang L, Romer M, Cater JR, Scharf MT, et al. Changes in components of energy regulation in mouse cortex with increases in wakefulness. Sleep. 2010;33:889-900.
- Jones S, Pfister-Genskow M, Benca RM, Cirelli C. Molecular correlates of sleep and wakefulness in the brain of the white-crowned sparrow. J Neurochem. 2008;105:46-62.
- Cirelli C, LaVaute TM, Tononi G. Sleep and wakefulness modulate gene expression in Drosophila. J Neurochem. 2005;94:1411-9.
- Cirelli C. Cellular consequences of sleep deprivation in the brain. Sleep Med Rev. 2006;10:307-21.
- Ali T, Choe J, Awab A, Wagener TL, Orr WC. Sleep, immunity and inflammation in gastrointestinal disorders. World J Gastroenterol. 2013;19:9231-9.
- Khanijow V, Prakash P, Emsellem HA, Borum ML, Doman DB. Sleep dysfunction and gastrointestinal diseases. Gastroenterol Hepatol. 2015;11:817-25.
- Parekh PJ, Oldfield EC 4th, Johnson DA. Wake-up call to clinicians: the impact of sleep dysfunction on gastrointestinal health and disease. J Clin Gastroenterol. 2018;52:194-203.
- Bhattacharyya A, Chattopadhyay R, Mitra S, Crowe SE. Oxidative stress: an essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol Rev. 2014;94:329-54.
- Campbell EL, Colgan SP. Control and dysregulation of redox signalling in the gastrointestinal tract. Nat Rev Gastroenterol Hepatol. 2019;16:106-20.
- Pérez S, Taléns-Visconti R, Rius-Pérez S, Finamor I, Sastre J. Redox signaling in the gastrointestinal tract. Free Radic Biol Med. 2017;104:75-103.
- Aviello G, Knaus UG. ROS in gastrointestinal inflammation: Rescue Or Sabotage. Br J Pharm. 2017;174:1704-18.
- Lasry A, Zinger A, Ben-Neriah Y. Inflammatory networks underlying colorectal cancer. Nat Immunol. 2016;17:230-40.
- Chen GY, Nuñez G. Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol. 2010;10:826-37.
- Donath MY, Shoelson SE. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 2011;11:98-107.
- Libby P, Lichtman AH, Hansson GK. Immune effector mechanisms implicated in atherosclerosis: from mice to humans. Immunity. 2013;38:1092-104.
- Bruunsgaard H, Ladelund S, Pedersen AN, Schroll M, Jørgensen T, Pedersen BK. Predicting death from tumour necrosis factor-alpha and interleukin-6 in 80-yearold people. Clin Exp Immunol. 2003;132:24-31.
- Volpato S, Guralnik JM, Ferrucci L, Balfour J, Chaves P, Fried LP, et al. Cardiovascular disease, interleukin-6, and risk of mortality in older women: the women’s health and aging study. Circulation. 2001;103:947-53.
- Smagula SF, Stone KL, Redline S, Ancoli-Israel S, Barrett-Connor E, Lane NE, et al. Actigraphy- and polysomnography-measured sleep disturbances, inflammation, and mortality among older men. Psychosom Med. 2016;78:686-96.
- Ridker PM. From C-reactive protein to interleukin-6 to interleukin-1: moving upstream to identify novel targets for atheroprotection. Circ Res. 2016;118:145-56.
- Grandner MA, Alfonso-Miller P, Fernandez-Mendoza J, Shetty S, Shenoy S, Combs D. Sleep: important considerations for the prevention of cardiovascular disease. Curr Opin Cardiol. 2016;31:551-65.
- Ferrie JE, Kivimäki M, Akbaraly TN, Singh-Manoux A, Miller MA, Gimeno D, et al. Associations between change in sleep duration and inflammation: findings on C-reactive protein and interleukin 6 in the Whitehall II Study. Am J Epidemiol. 2013;178:956-61.
- Carroll JE, Irwin MR, Stein Merkin S, Seeman TE. Sleep and multisystem biological risk: a population-based study. PLoS ONE. 2015;10:e0118467.
- Clark AJ, Dich N, Lange T, Jennum P, Hansen AM, Lund R, et al. Impaired sleep and allostatic load: cross-sectional results from the Danish Copenhagen Aging and Midlife Biobank. Sleep Med. 2014;15:1571-8.
- Dowd JB, Goldman N, Weinstein M. Sleep duration, sleep quality, and biomarkers of inflammation in a Taiwanese population. Ann Epidemiol. 2011;21:799-806.
- Patel SR, Zhu X, Storfer-Isser A, Mehra R, Jenny NS, Tracy R, et al. Sleep duration and biomarkers of inflammation. Sleep. 2009;32:200-4.
- Prather AA, Vogelzangs N, Penninx BW. Sleep duration, insomnia, and markers of systemic inflammation: results from the Netherlands Study of Depression and Anxiety (NESDA). J Psychiatr Res. 2015;60:95-102.
- Jackowska M, Steptoe A. Sleep and future cardiovascular risk: prospective anaIysis from the English Longitudinal Study of Ageing. Sleep Med. 2015;16:768-74.
- McDade TW, Hawkley LC, Cacioppo JT. Psychosocial and behavioral predictors of inflammation in middle-aged and older adults: the Chicago health, aging, and social relations study. Psychosom Med. 2006;68:376-81.
- Prather AA, Marsland AL, Hall M, Neumann SA, Muldoon MF, Manuck SB. Normative variation in self-reported sleep quality and sleep debt is associated with stimulated pro-inflammatory cytokine production. Biol Psychol. 2009;82:12-17.
- Suarez EC. Self-reported symptoms of sleep disturbance and inflammation, coagulation, insulin resistance and psychosocial distress: evidence for gender disparity. Brain Behav Immun. 2008;22:960-8.
- Taheri S, Austin D, Lin L, Nieto FJ, Young T, Mignot E. Correlates of serum C-reactive protein (CRP)-no association with sleep duration or sleep disordered breathing. Sleep. 2007;30:991-6.
- Prather AA, Epel ES, Cohen BE, Neylan TC, Whooley MA. Gender differences in the prospective associations of self-reported sleep quality with biomarkers of systemic inflammation and coagulation: findings from the Heart and Soul Study. J Psychiatr Res. 2013;47:1228-35.
- Obayashi K, Saeki K, Kurumatani N. Gender differences in the association between objective sleep quality and leukocyte count: The HEIJO-KYO cohort. Physiol Behav. 2016;164:19-24.
- Blair LM, Porter K, Leblebicioglu B, Christian LM. Poor sleep quality and associated inflammation predict preterm birth: heightened risk among African Americans. Sleep. 2015;38:1259-67.
- Okun ML, Luther JF, Wisniewski SR, Wisner KL. Disturbed sleep and inflammatory cytokines in depressed and nondepressed pregnant women: an exploratory analysis of pregnancy outcomes. Psychosom Med. 2013;75:670-81.
- Tsujimura T, Matsuo Y, Keyaki T, Sakurada K, Imanishi J. Correlations of sleep disturbance with the immune system in type 2 diabetes mellitus. Diabetes Res Clin Pr. 2009;85:286-92.
- Besedovsky L, Lange T, Haack M. The sleep-immune crosstalk in health and disease. Physiol Rev. 2019;99:1325-80.
- Opp MR. Cytokines and sleep. Sleep Med Rev. 2005;9:355-64.
- Lou X, Wang H, Tu Y, Tan W, Jiang C, Sun J, et al. Alterations of sleep quality and circadian rhythm genes expression in elderly thyroid nodule patients and risks associated with thyroid malignancy. Sci Rep. 2021;11:13682.
- Zhou L, Luo Z, Li Z, Huang Q. Circadian clock is associated with tumor microenvironment in kidney renal clear cell carcinoma. Aging. 2020;12:14620-32.
- Ramos CA, Ouyang C, Qi Y, Chung Y, Cheng CT, LaBarge MA, et al. A noncanonical function of BMAL1 metabolically limits obesity-promoted triplenegative breast cancer. iScience. 2020;23:100839.
- Chen P, Hsu WH, Chang A, Tan Z, Lan Z, Zhou A, et al. Circadian regulator CLOCK recruits immune-suppressive microglia into the GBM tumor microenvironment. Cancer Discov. 2020;10:371-81.
- He L, Fan Y, Zhang Y, Tu T, Zhang Q, Yuan F, et al. Single-cell transcriptomic analysis reveals circadian rhythm disruption associated with poor prognosis and drug-resistance in lung adenocarcinoma. J Pineal Res. 2022;73:e12803.
- Chun SK, Fortin BM, Fellows RC, Habowski AN, Verlande A, Song WA, et al. Disruption of the circadian clock drives Apc loss of heterozygosity to accelerate colorectal cancer. Sci Adv. 2022;8:eabo2389.
- Hunt T, Sassone-Corsi P. Riding tandem: circadian clocks and the cell cycle. Cell. 2007;129:461-4.
- Satyanarayana A, Kaldis P. Mammalian cell-cycle regulation: several Cdks, numerous cyclins and diverse compensatory mechanisms. Oncogene. 2009;28:2925-39.
- Quail DF, Joyce JA. Microenvironmental regulation of tumor progression and metastasis. Nat Med. 2013;19:1423-37.
- Xuan W, Khan F, James CD, Heimberger AB, Lesniak MS, Chen P. Circadian regulation of cancer cell and tumor microenvironment crosstalk. Trends Cell Biol. 2021;31:940-50.
- Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39:1-10.
- Raskov H, Orhan A, Christensen JP, Gögenur I. Cytotoxic CD8(+) T cells in cancer and cancer immunotherapy. Br J Cancer. 2021;124:359-67.
- Marvel D, Gabrilovich Dl. Myeloid-derived suppressor cells in the tumor microenvironment: expect the unexpected. J Clin Invest. 2015;125:3356-64.
- Roberts NT, MacDonald CR, Mohammadpour H, Antoch MP, Repasky EA. Circadian rhythm disruption increases tumor growth rate and accumulation of myeloid-derived suppressor cells. Adv Biol. 2022;6:e2200031.
- Méndez-Ferrer S, Lucas D, Battista M, Frenette PS. Haematopoietic stem cell release is regulated by circadian oscillations. Nature. 2008;452:442-7.
- Logan RW, Arjona A, Sarkar DK. Role of sympathetic nervous system in the entrainment of circadian natural-killer cell function. Brain Behav Immun. 2011;25:101-9.
- Pathria P, Louis TL, Varner JA. Targeting tumor-associated macrophages in cancer. Trends Immunol. 2019;40:310-27.
- Alexander RK, Liou YH, Knudsen NH, Starost KA, Xu C, Hyde AL, et al. Bmal1 integrates mitochondrial metabolism and macrophage activation. Elife. 2020;9:e54090.
- Early JO, Menon D, Wyse CA, Cervantes-Silva MP, Zaslona Z, Carroll RG, et al. Circadian clock protein BMAL1 regulates IL-1
in macrophages via NRF2. Proc Natl Acad Sci USA. 2018;115:E8460-8. - Nguyen KD, Fentress SJ, Qiu Y, Yun K, Cox JS, Chawla A. Circadian gene Bmal1 regulates diurnal oscillations of Ly6C(hi) inflammatory monocytes. Science. 2013;341:1483-8.
- Huang C , Zhang C , Cao Y, Li J, Bi F. Major roles of the circadian clock in cancer. Cancer Biol Med. 2023;20:1-24.
- Sato S, Sakurai T, Ogasawara J, Takahashi M, Izawa T, Imaizumi K, et al. A circadian clock gene, Rev-erba, modulates the inflammatory function of macrophages through the negative regulation of Ccl 2 expression. J Immunol. 2014;192:407-17.
- Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 2004;25:677-86.
- Verreck FA, de Boer T, Langenberg DM, Hoeve MA, Kramer M, Vaisberg E, et al. Human IL-23-producing type 1 macrophages promote but IL-10-producing type 2 macrophages subvert immunity to (myco)bacteria. Proc Natl Acad Sci USA. 2004;101:4560-5.
- Qin C, Zhou LQ, Ma XT, Hu ZW, Yang S, Chen M, et al. Dual functions of microglia in ischemic stroke. Neurosci Bull. 2019;35:921-33.
- Li X, Guan J, Jiang Z, Cheng S, Hou W, Yao J, et al. Microglial exosome miR-72393p promotes glioma progression by regulating circadian genes. Neurosci Bull. 2021;37:497-510.
- Newman AM, Alizadeh AA. High-throughput genomic profiling of tumorinfiltrating leukocytes. Curr Opin Immunol. 2016;41:77-84.
- He W, Holtkamp S, Hergenhan SM, Kraus K, de Juan A, Weber J, et al. Circadian expression of migratory factors establishes lineage-specific signatures that guide the homing of leukocyte subsets to tissues. Immunity. 2018;49:1175-90.e7.
- Gao Y, Meng D, Sun N, Zhu Z, Zhao R, Lu C, et al. Clock upregulates intercellular adhesion molecule-1 expression and promotes mononuclear cells adhesion to endothelial cells. Biochem Biophys Res Commun. 2014;443:586-91.
- Hadadi E, Taylor W, Li XM, Aslan Y, Villote M, Rivière J, et al. Chronic circadian disruption modulates breast cancer stemness and immune microenvironment to drive metastasis in mice. Nat Commun. 2020;11:3193.
- Hu X, Liu X, Moisan J, Wang Y, Lesch CA, Spooner C, et al. Synthetic RORY agonists regulate multiple pathways to enhance antitumor immunity. Oncoimmunology. 2016;5:e1254854.
- Lee IK, Song H, Kim H, Kim IS, Tran NL, Kim SH, et al. RORa regulates cholesterol metabolism of CD8+ T cells for anticancer immunity. Cancers. 2020;12:1733.
- Kaplon J, van Dam L, Peeper D. Two-way communication between the metabolic and cell cycle machineries: the molecular basis. Cell Cycle. 2015;14:2022-32.
- de Winter L, Schepers LW, Cuaresma M, Barbosa MJ, Martens DE, Wijffels RH. Circadian rhythms in the cell cycle and biomass composition of Neochloris oleoabundans under nitrogen limitation. J Biotechnol. 2014;187:25-33.
- Krishnaiah SY, Wu G, Altman BJ, Growe J, Rhoades SD, Coldren F, et al. Clock regulation of metabolites reveals coupling between transcription and metabolism. Cell Metab. 2017;25:961-74.e4.
- Janich P, Pascual G, Merlos-Suárez A, Batlle E, Ripperger J, Albrecht U, et al. The circadian molecular clock creates epidermal stem cell heterogeneity. Nature. 2011;480:209-14.
- Geyfman M, Kumar V, Liu Q, Ruiz R, Gordon W, Espitia F, et al. Brain and muscle Arnt-like protein-1 (BMAL1) controls circadian cell proliferation and susceptibility to UVB-induced DNA damage in the epidermis. Proc Natl Acad Sci USA. 2012;109:11758-63.
- Plikus MV, Vollmers C, de la Cruz D, Chaix A, Ramos R, Panda S, et al. Local circadian clock gates cell cycle progression of transient amplifying cells during regenerative hair cycling. Proc Natl Acad Sci USA. 2013;110:E2106-15.
- Karpowicz P, Zhang Y, Hogenesch JB, Emery P, Perrimon N. The circadian clock gates the intestinal stem cell regenerative state. Cell Rep. 2013;3:996-1004.
- Weger M, Diotel N, Dorsemans AC, Dickmeis T, Weger BD. Stem cells and the circadian clock. Dev Biol. 2017;431:111-23.
- Kowalska E, Ripperger JA, Hoegger DC, Bruegger P, Buch T, Birchler T, et al. NONO couples the circadian clock to the cell cycle. Proc Natl Acad Sci USA. 2013;110:1592-9.
- Bjarnason GA, Jordan RC, Wood PA, Li Q, Lincoln DW, Sothern RB, et al. Circadian expression of clock genes in human oral mucosa and skin: association with specific cell-cycle phases. Am J Pathol. 2001;158:1793-801.
- Bjarnason GA, Jordan RC, Sothern RB. Circadian variation in the expression of cell-cycle proteins in human oral epithelium. Am J Pathol. 1999;154:613-22.
- Soták M, Sumová A, Pácha J. Cross-talk between the circadian clock and the cell cycle in cancer. Ann Med. 2014;46:221-32.
- Matsuo T, Yamaguchi S, Mitsui S, Emi A, Shimoda F, Okamura H. Control mechanism of the circadian clock for timing of cell division in vivo. Science. 2003;302:255-9.
- Gréchez-Cassiau A, Rayet B, Guillaumond F, Teboul M, Delaunay F. The circadian clock component BMAL1 is a critical regulator of p21WAF1/CIP1 expression and hepatocyte proliferation. J Biol Chem. 2008;283:4535-42.
- Miki T, Matsumoto T, Zhao Z, Lee CC. p53 regulates Period2 expression and the circadian clock. Nat Commun. 2013;4:2444.
- Bee L, Marini S, Pontarin G, Ferraro P, Costa R, Albrecht U, et al. Nucleotide excision repair efficiency in quiescent human fibroblasts is modulated by circadian clock. Nucleic Acids Res. 2015;43:2126-37.
- Oklejewicz M, Destici E, Tamanini F, Hut RA, Janssens R, van der Horst GT. Phase resetting of the mammalian circadian clock by DNA damage. Curr Biol. 2008;18:286-91.
- Masri S, Kinouchi K, Sassone-Corsi P. Circadian clocks, epigenetics, and cancer. Curr Opin Oncol. 2015;27:50-56.
- Scheiermann C, Kunisaki Y, Lucas D, Chow A, Jang JE, Zhang D, et al. Adrenergic nerves govern circadian leukocyte recruitment to tissues. Immunity. 2012;37:290-301.
- Wang Y, Sun N, Lu C, Bei Y, Qian R, Hua L. Upregulation of circadian gene ‘hClock’ contribution to metastasis of colorectal cancer. Int J Oncol. 2017;50:2191-9.
- Hu Z, Brooks SA, Dormoy V, Hsu CW, Hsu HY, Lin LT, et al. Assessing the carcinogenic potential of low-dose exposures to chemical mixtures in the environment: focus on the cancer hallmark of tumor angiogenesis. Carcinogenesis. 2015;36:S184-202.
- Jiang X, Wang J, Deng X, Xiong F, Zhang S, Gong Z, et al. The role of microenvironment in tumor angiogenesis. J Exp Clin Cancer Res. 2020;39:204.
- Koyanagi S, Kuramoto Y, Nakagawa H, Aramaki H, Ohdo S, Soeda S, et al. A molecular mechanism regulating circadian expression of vascular endothelial growth factor in tumor cells. Cancer Res. 2003;63:7277-83.
- Zhou J, Li X, Zhang M, Gong J, Li Q, Shan B, et al. The aberrant expression of rhythm genes affects the genome instability and regulates the cancer immunity in pan-cancer. Cancer Med. 2020;9:1818-29.
- Shi SQ, Ansari TS, McGuinness OP, Wasserman DH, Johnson CH. Circadian disruption leads to insulin resistance and obesity. Curr Biol. 2013;23:372-81.
- Kettner NM, Mayo SA, Hua J, Lee C, Moore DD, Fu L. Circadian dysfunction induces leptin resistance in mice. Cell Metab. 2015;22:448-59.
- Gabrilovich DI, Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol. 2009;9:162-74.
- Michaeli J, Shaul ME, Mishalian I, Hovav AH, Levy L, Zolotriov L, et al. Tumorassociated neutrophils induce apoptosis of non-activated CD8 T-cells in a TNFa and NO-dependent mechanism, promoting a tumor-supportive environment. Oncoimmunology. 2017;6:e1356965.
- Feng S, Cheng X, Zhang L, Lu X, Chaudhary S, Teng R, et al. Myeloid-derived suppressor cells inhibit T cell activation through nitrating LCK in mouse cancers. Proc Natl Acad Sci USA. 2018;115:10094-9.
- Peranzoni E, Lemoine J, Vimeux L, Feuillet V, Barrin S, Kantari-Mimoun C, et al. Macrophages impede CD8 T cells from reaching tumor cells and limit the efficacy of anti-PD-1 treatment. Proc Natl Acad Sci USA. 2018;115:E4041-50.
- Zhao Y, Liu M, Chan XY, Tan SY, Subramaniam S, Fan Y, et al. Uncovering the mystery of opposite circadian rhythms between mouse and human leukocytes in humanized mice. Blood. 2017;130:1995-2005.
- Müller A, Homey B, Soto H, Ge N, Catron D, Buchanan ME, et al. Involvement of chemokine receptors in breast cancer metastasis. Nature. 2001;410:50-6.
- Helbig G, Christopherson KW 2nd, Bhat-Nakshatri P, Kumar S, Kishimoto H, Miller KD, et al. NF-kappaB promotes breast cancer cell migration and metastasis by inducing the expression of the chemokine receptor CXCR4. J Biol Chem. 2003;278:21631-8.
- Nagarsheth N, Wicha MS, Zou W. Chemokines in the cancer microenvironment and their relevance in cancer immunotherapy. Nat Rev Immunol. 2017;17:559-72.
- Cermakian N, Stegeman SK, Tekade K, Labrecque N. Circadian rhythms in adaptive immunity and vaccination. Semin Immunopathol. 2022; 44:193-207.
- Doruk YU, Yarparvar D, Akyel YK, Gul S, Taskin AC, Yilmaz F, et al. A CLOCKbinding small molecule disrupts the interaction between CLOCK and BMAL1 and enhances circadian rhythm amplitude. J Biol Chem. 2020;295:3518-31.
- Ramanathan C, Kathale ND, Liu D, Lee C, Freeman DA, Hogenesch JB, et al. mTOR signaling regulates central and peripheral circadian clock function. PLoS Genet. 2018;14:e1007369.
- Zhang S, Zhang J, Deng Z, Liu H, Mao W, Jiang F, et al. Circadian clock components RORa and Bmal1 mediate the anti-proliferative effect of MLN4924 in osteosarcoma cells. Oncotarget. 2016;7:66087-99.
- Zhang H, Liu Y, Liu D, Zeng Q, Li L, Zhou Q, et al. Time of day influences immune response to an inactivated vaccine against SARS-CoV-2. Cell Res. 2021;31:1215-7.
- Bass J. Circadian topology of metabolism. Nature. 2012;491:348-56.
- Masri S, Sassone-Corsi P. The emerging link between cancer, metabolism, and circadian rhythms. Nat Med. 2018;24:1795-803.
AUTHOR CONTRIBUTIONS
FUNDING
COMPETING INTERESTS
ADDITIONAL INFORMATION
Reprints and permission information is available at http://www.nature.com/ reprints

© The Author(s) 2024
Department of Immunology, School of Basic Medical Sciences, Air Force Medical University, Xi’an, China. Faculty of Life Sciences, Northwest University, Xi’an, China. These authors contributed equally: Yuen Zeng, Zichan Guo. email: chenlh@fmmu.edu.cn
