DOI: https://doi.org/10.2147/jmdh.s419714
PMID: https://pubmed.ncbi.nlm.nih.gov/38840704
تاريخ النشر: 2024-05-01
ترجمة، التكيف عبر الثقافات، والتحقق من أدوات القياس: دليل عملي للباحثين المبتدئين
الملخص
التحقق عبر الثقافات من أدوات القياس المبلغ عنها ذاتيًا للبحث هو عملية طويلة ومعقدة، تتضمن مخاطر محددة من التحيز التي قد تؤثر على عملية البحث والنتائج. علاوة على ذلك، يتطلب من الباحثين أن يكون لديهم مجموعة واسعة من المعرفة التقنية حول جوانب الترجمة والتكيف والاختبار المسبق، وأغراضها وخياراتها، حول الخصائص السيكومترية المختلفة، والأدلة المطلوبة لتقييمها ومعرفة معالجة البيانات الكمية وتحليلها باستخدام البرمجيات الإحصائية. كان الهدف من هذه المقالة: 1) تحديد جميع الإرشادات والتوصيات للترجمة، التكيف عبر الثقافات، والتحقق ضمن علوم الرعاية الصحية؛ 2) وصف المناهج المنهجية التي تم وضعها في هذه الإرشادات لإجراء الترجمة، التكيف، والتحقق عبر الثقافات؛ و 3) تقديم دليل عملي يتضمن خيارات منهجية متنوعة للباحثين المبتدئين المشاركين في ترجمة، تكيف، والتحقق من أدوات القياس. تم الحصول على اثنين وأربعين إرشادًا حول الترجمة، التكيف، أو التحقق عبر الثقافات من أدوات القياس من “CINAHL مع النص الكامل” (عبر EBSCO) و”MEDLINE مع النص الكامل”. تم إجراء تحليل محتوى لتحديد أوجه التشابه والاختلاف في المناهج المنهجية الموصى بها. بناءً على هذه التشابهات والاختلافات، اقترحنا دليلًا من ثمانية خطوات يتضمن: أ) الترجمة الأمامية؛ 2) تجميع الترجمات؛ 3) الترجمة العكسية؛ 4) التوافق؛ 5) الاختبار المسبق؛ 6) الاختبار الميداني؛ 7) التحقق السيكومتري، و 8) تحليل الخصائص السيكومترية. إنه دليل عملي لأنه يوفر معلومات شاملة وواسعة حول المناهج المنهجية المتاحة للباحثين. هذه هي أول مراجعة أدبية منهجية تُجرى في علوم الرعاية الصحية بشأن المناهج المنهجية الموصى بها من قبل الإرشادات الموجودة.
المقدمة
موثوقة،
المفاهيم والمصطلحات المحددة
أنواع المعادلة
معادلة هيردمان وآخرون
أنماط التحيزات
النهج المنهجية
مبررات البحث والأهداف
المواد والأساليب
تحديد الإرشادات الموجودة
تحليل محتوى الإرشادات الموجودة
اقتراح لإرشادات عملية
إرشادات عملية مستندة إلى منظور شامل،
النتائج
المعلومات العامة
للأداة وكمواد تكميلية في نشر مقال علمي يصف عملية التكييف عبر الثقافات لأداة القياس.
الترجمة عبر الثقافات
المفاهيم مع عناصر غير متضمنة في الترجمات السابقة ولكن مع معرفة واسعة بالمفاهيم المراد قياسها.
التكيف عبر الثقافات
من المشاركين في الاختبار المسبق،
التحقق عبر الثقافات
المؤلفين يشيرون بشكل عام إلى تقييمها.
النقاش
توثيق العملية
كتابي على الأهمية المحتملة للأدوات للمشاركين من “الثقافة المستهدفة”، فضلاً عن المعادلة التشغيلية للأداة. قد يكون توثيق القرارات تجريبيًا ولكن معظمها سيكون نظريًا بطبيعته.
مراحل العملية
الترجمة الأمامية
ترجمة أمامية تركيبية
الترجمة العكسية
من الوثيقة الأصلية. بغض النظر عما إذا كانت هناك اختلافات طفيفة بين النسختين، فإن الجانب الرئيسي الذي يحتاج إلى التحليل في الخطوة التالية هو ما إذا كان هناك تغيير في المعنى بين العناصر في الترجمة العكسية والعناصر في “الأداة الأصلية”.
التناغم
الاختبار المسبق
العينة الملائمة.
اختبار الميدان
التحقق النفسي
معامل الارتباط بين العنصر والإجمالي.
تحليل الخصائص النفسية
تداعيات للممارسة
تسمح لهؤلاء الباحثين بالتركيز على المعرفة والمهارات حول الأساليب المنهجية النوعية والكمية التي يحتاجون إلى اكتسابها وتطويرها ليكونوا قادرين على إجراء دراسة تحقق. أخيرًا، تتيح للباحثين المبتدئين تطوير مهارات القيادة الجماعية في عملية البحث. يشمل ذلك ليس فقط تنسيق أنشطة فريق المترجمين والخبراء الذين يتكون منهم فريق البحث ولكن أيضًا توقع النتائج البحثية التي يمكن أن تتولد من عملية الترجمة والتكيف والتحقق عبر الثقافات. تشمل أمثلة هذه النتائج مقالة مراجعة أدبية حول بناء الأداة، ومقالة تحليل مفهوم، ومقالة منهجية توضح عملية الترجمة والتكيف عبر الثقافات، وأخرى تفصل جودة الخصائص النفسية للأداة.
استنتاج
شكر وتقدير
إفصاح
References
- May C, Finch T, Rapley T. Normalization process theory. In: Nielsen P, Birken SA, editors. Handbook of Implementation Science. Edward Elgar Publishing; 2020:157.
- Portney LG. Designing surveys and questionnaires. In: Foundations of Clinical Research: Applications to Evidence-Based Practice. 4th ed. F.A. Davis Company; 2020:141-150.
- Chow S-C, Liu J-P. Basic statistic concepts. In: Design and Analysis of Clinical Trials. 2nd ed. Wiley-Interscience; 2004:47-54.
- Sidani S, Braden CJ. Examination of interventions’ acceptance. In: Nursing and Health Interventions: Design, Evaluation, and Implementation. 2nd ed. Wiley-Blackwell; 2021:218-225.
- Efstathiou G. Translation, adaptation and validation process of research intruments. In: Suohnen R, Stolt M, Papatravou E, editors. Individualized Care: Theory, Measurement, Research and Practice. Springer; 2019.
- Cha E-S, Kim KH, Erlen JA. Translation of scales in cross-cultural research: issues and techniques. J Adv Nurs. 2007;58(4):386-395. doi:10.1111/j.1365-2648.2007.04242.x
- Sutherland S. Outcomes research. In: Gray JR, Grove SK, editors. The Practice of Nursing Research: Appraisal, Synthesis, and Generation of Evidence. 9th ed. Elsevier; 2020:365.
- Miller LA, Lovler RL. How do we assess the psychometric quality of a test? In: Foundation of Psychological Testing: A Practical Approach. 6th ed. Sage Publications; 2020:326.
- Kristjansson EA, Desrochers A, Zumbo B. Translating and adapting measurement instruments for cross-linguistic and cross-cultural research: a guide for practitioners. Can J Nurs Res. 2003;35(2):127-142.
- Alagappan T. The cross-cultural adaptation process of a patient-reported outcome measure. J Sci Soc. 2023;50(1):13. doi:10.4103/jss. jss_136_21
- Sul SIR, Lucas PRMB. Translation and validation of the anticipated turnover scale for the Portuguese cultural context. Nurs Open. 2020;7 (5):1475-1481. doi:10.1002/nop2.521
- Almeida S, Nascimento A, Lucas PB, Jesus É, Araújo B. RN4CAST study in Portugal: validation of the Portuguese version of the Practice Environment Scale of the Nursing Work Index. Aquichan. 2020;20(3):1-10. doi:10.5294/aqui.2020.20.3.8
- Lucas P, Jesus E, Almeida S, Araújo B. Validation of the psychometric properties of the Practice Environment Scale of Nursing Work Index in primary health care in Portugal. Int J Environ Res Public Health. 2021;18(12):6422. doi:10.3390/ijerph18126422
- Anunciada S, Benito P, Gaspar F, Lucas P. Validation of psychometric properties of the Nursing Work Index: revised scale in Portugal. Int J Environ Res Public Health. 2022;19(9):4933. doi:10.3390/ijerph 19094933
- Carvalho M, Gaspar F, Potra T, Lucas P. Translation, adaptation, and validation of the Self-efficacy scale for clinical nurse leaders for the Portuguese culture. Int J Environ Res Public Health. 2022;19(14):8590. doi:10.3390/ijerph19148590
- Sousa E, Lin C-F, Gaspar F, Lucas P. Translation and validation of the indicators of quality nursing work environments in the Portuguese cultural context. Int J Environ Res Public Health. 2022;19(19):12313. doi:10.3390/ijerph191912313
- Cabrita C, Lucas P, Teixeira G, Gaspar F. Translation and validation of the Individual Workload Perception Scale: revised for Portuguese nurses. Healthcare. 2022;10(12):2476. doi:10.3390/healthcare10122476
- Gomes P, Ribeiro S, Silva M, et al. Cross-cultural validation of the Portuguese version of the Quality of Oncology Nursing Care Scale. Cancers (Basel). 2024;16(5):859. doi:10.3390/cancers16050859
- Cunha F, Pinto MR, Riesch S, Lucas P, Almeida S, Vieira M. Translation, adaptation, and validation of the Portuguese version of the Exercise of Self-Care Agency Scale. Healthcare. 2024;12(2):159. doi:10.3390/healthcare12020159
- Waltz CF, Strickland OL, Lenz ER. Other mesurement issues. In: Measurement in Nursing and Health Research. 4th ed. Springer Publishing; 2010:446-448.
- Beaton D, Bombardier C, Guillemin F, Ferraz MB. Guidelines for the process of cross-cultural adaptation of self-report measures. Spine (Phila Pa 1976). 2000;25(24):3186-3191. doi:10.1097/00007632-200012150-00014
- Hedrih V. Test translation. In: Adapting Psychological Tests and Measurement Instruments for Cross-Cultural Research: An Introduction. Routledge; 2020:48-98.
- Nord C. Defining translation functions: the translation brief as a guideline for the trainee translator. Ilha Do Desterro. 1997;33:41-55.
- Montalt V, González-Davies M. Understanding medical communication. In: Medical Translation Step by Step: Learning by Drafting. Routledge; 2006:47.
- Epstein J, Santo RM, Guillemin F. A review of guidelines for cross-cultural adaptation of questionnaires could not bring out a consensus.
Clin Epidemiol. 2015;68(4):435-441. doi:10.1016/j.jclinepi.2014.11.021 - Herdman M, Fox-Rushby J, Badia X. A model of equivalence in the cultural adaptation of HRQoL instruments: the universalist approach. Qual Life Res. 1998;7(4):323-335. doi:10.1023/a:1024985930536
- Peña ED. Lost in translation: methodological considerations in cross-cultural research. Child Dev. 2007;78(4):1255-1264. doi:10.1111/j.14678624.2007.01064.x
- Alonso J, Black C, Norregaard J-C, et al. Cross-cultural differences in the reporting of global functional capacity. Med Care. 1998;36 (6):868-878. doi:10.1097/00005650-199806000-00010
- Muniz J, Hambleton RK, Xing D. Small samples studies to detect flaws in item translations. Int J Test. 2001;1(2):115-135. doi:10.1207/ S15327574IJT0102_2
- Waltz CF, Strickland OL, Lenz ER. Other measurement issues. In: Measurement in Nursing and Health Research. 4th ed. Springer Publishing; 2010:449-452.
- Riccio CA, Yoon H, McCormick AS. Neuropsychological test selection with clients who are Asian. In: Davis JM, D’Amato RC, editors. Neuropsychology of Asians and Asian-Americans: Practical and Theoretical Considerations. Spinger Publishing; 2014:153.
- Smith TW, et al. Developing and evaluating cross national survey instruments. In: Presser S, Rothgeb JM, Couper MP, editors. Methods for Testing and Evaluating Survey Questionnaires. Wiley Interscience; 2004. 439-442.
- van de Vijver F, Portinga YH. Conceptual and methodological issues in adapting tests. In: Hembletom RK, Merenda PF, Spilberg CD, editors. Adapting Educational and Psychological Tests for Cross-Cultural Assessment. Lawrence Erlbaum Associates; 2005:41-47.
- Austin PC, Brunner LJ. Type I error inflation in the presence of a ceiling effect. Am Stat. 2003;57(2):97-104. doi:10.1198/0003130031450
- Holbrook A. Acquiescence response bias. In: Lavrakas PJ, editor. Encyclopedia of Survey Research Methods. Vol. I. Sage; 2008:3.
- van de Vijver FJR, Poortinga YH. Towards an Integrated Analysis of Bias in Cross-Cultural Assessment. Eur J Psychol Assess. 1997; 13 (1):29-37. doi:10.1027/1015-5759.13.1.29
- Bartram D. Increasing validity with forced-choice criterion measurement formats. Int J Sel Assess. 2007;15(3):263-272. doi:10.1111/j.14682389.2007.00386.x
- Wivagg J. Forced choice. In: Lavrakas PJ, editor. Encyclopedia of Survey Research Methods. Sage; 2008:289-290.
- Maitland A. Attitudes measurement. In: Lavrakas PJ, editor. Encyclopedia of Survey Research Methods. Sage; 2008:37-38.
- Hilton A, Skrutkowski M. Translating instruments into other languages: development and testing processes. Cancer Nurs. 2002;25(1):1-7. doi:10.1097/00002820-200202000-00001
- Lino de CR, Brüggemann M, Souza M de L de OM, Barbosa de S, Santos Dos EKA. Adaptação transcultural de instrumentos de pesquisa conduzida pela enfermagem do Brasil: uma revisão integrativa. Texto Context – Enferm. 2018;26(4). doi:10.1590/0104-07072017001730017
- Guillemin F, Bombardier C, Beaton D. Cross-cultural adaptation of health-related quality of life measures: literature review and proposed guidelines. J Clin Epidemiol. 1993;46(12):1417-1432. doi:10.1016/0895-4356(93)90142-N
- Machado da R, Fernandes AD de BF S, Oliveira de ALCB, Soares LS, Gouveia de O MT, Silva da GRF. Métodos de adaptação transcultural de instrumentos na área da enfermagem. Rev Gaúcha Enferm. 2018;39. doi:10.1590/1983-1447.2018.2017-0164
- Acquadro C, Conway K, Hareendran A, Aaronson N. Literature review of methods to translate Health-Related Quality of Life Questionnaires for use in multinational clinical trials. Value Heal. 2008;11(3):509-521. doi:10.1111/j.1524-4733.2007.00292.x
- Farina N, Jacobs R, Sani TP, et al. Description of the cross-cultural process adopted in the STRiDE (STrengthening Responses to dementia in DEveloping countries) program: a methodological overview. Alzheimer’s Dement Diagnosis, Assess Dis Monit. 2022;14(1). doi:10.1002/ dad2. 12293
- Furukawa R, Driessnack M, Colclough Y. A committee approach maintaining cultural originality in translation. Appl Nurs Res.
(2):144-146. doi:10.1016/j.apnr.2013.11.011 - Helmich E, Cristancho S, Diachun L, Lingard L. ‘How would you call this in English?’: being reflective about translations in international, cross-cultural qualitative research. Perspect Med Educ. 2017;6(2):127-132. doi:10.1007/S40037-017-0329-1
- Uysal-Bozkir Ö, Parlevliet JL, de Rooij SE. Insufficient cross-cultural adaptations and psychometric properties for many translated health assessment scales: a systematic review. J Clin Epidemiol. 2013;66(6):608-618. doi:10.1016/j.jclinepi.2012.12.004
- Albach CA, Wagland R, Hunt KJ. Cross-cultural adaptation and measurement properties of generic and cancer-related patient-reported outcome measures (PROMs) for use with cancer patients in Brazil: a systematic review. Qual Life Res. 2018;27(4):857-870. doi:10.1007/s11136-017-1703-5
- Praveen S, Parmar J, Chandio N, Arora A. A systematic review of cross-cultural adaptation and psychometric properties of oral health literacy tools. Int J Environ Res Public Health. 2021;18(19):10422. doi:10.3390/ijerph181910422
- Min SN, Duangthip D, Gao SS, Detsomboonrat P. Quality of the adaptation procedures and psychometric properties of the scale of oral health outcomes for 5-year-old children (SOHO-5): a systematic review. Qual Life Res. 2023;32(6):1537-1547. doi:10.1007/s11136-022-03280-2
- Danielsen AK, Pommergaard H-C, Burcharth J, Angenete E, Rosenberg J. Translation of questionnaires measuring health related quality of life is not standardized: a literature based research study. PLoS One. 2015;10(5):e0127050. doi:10.1371/journal.pone. 0127050
- Øygarden A-MU, Berg RC, Abudayya A, Glavin K, Strøm BS. Measurement instruments for parental stress in the postpartum period: a scoping review. PLoS One. 2022;17(3):e0265616. doi:10.1371/journal.pone. 0265616
- Echevarría-Guanilo ME, Gonçalves N, Romanoski PJ. Psychometric properties of measurement instruments: conceptual basis and evaluation methods: part II. Texto Context – Enferm. 2019;28(e20170311):1-14. doi:10.1590/1980-265x-tce-2017-0311
- Cruchinho P, Teixeira G, Lucas P, Gaspar F. Evaluating the methodological approaches of cross-cultural adaptation of the Bedside Handover Attitudes and Behaviours Questionnaire into Portuguese. J Healthc Leadersh. 2023;15:193-208. doi:10.2147/JHL.S422122
- Slade D, Murray KA, Pun JKH, Eggins S. Nurses’ perceptions of mandatory bedside clinical handovers: an Australian hospital study. J Nurs Manag. 2019;27(1):161-171. doi:10.1111/jonm. 12661
- Freudental-Pederson M, Hartmann-Peterson K, Nielsen LD. Mixing methods in the search for mobile complexity. In: Ficham B, Mark M, Murray L, editors. Mobile Methodologies. Palgrave McMillan; 2010:30.
- Peters M, Passchier J. Translating instruments for cross-cultural studies in headache research. Headache J Head Face Pain. 2006;46(1):82-91. doi:10.1111/j.1526-4610.2006.00298.x
- Weeks A, Swerissen H, Belfrage J. Issues, challenges, and solutions in translating study instruments. Eval Rev. 2007;31(2):153-165. doi:10.1177/0193841X06294184
- Wild D, Grove A, Martin M, et al. Principles of good practice for the translation and cultural adaptation process for patient-reported outcomes (PRO) measures: report of the ISPOR task force for translation and cultural adaptation. Value Heal. 2005;8(2):94-104. doi:10.1111/j.15244733.2005.04054.x
- Reichenheim ME, Moraes CL. Operacionalização de adaptação transcultural de instrumentos de aferição usados em epidemiologia. Rev Saude Publica. 2007;41(4):665-673. doi:10.1590/S0034-89102006005000035
- Sousa V, Rojjanasrirat W. Translation, adaptation and validation of instruments or scales for use in cross-cultural health care research: a clear and user-friendly guideline. J Eval Clin Pract. 2011;17(2):268-274. doi:10.1111/j.1365-2753.2010.01434.x
- Hagell P, Hedin P-J, Meads DM, Nyberg L, McKenna SP. Effects of Method of Translation of Patient-Reported Health Outcome Questionnaires: a Randomized Study of the Translation of the Rheumatoid Arthritis Quality of Life (RAQoL) Instrument for Sweden. Value Heal. 2010;13(4):424-430. doi:10.1111/j.1524-4733.2009.00677.x
- Lee WL, Chinna K, Lim Abdullah K, Zainal Abidin I. The forward-backward and dual-panel translation methods are comparable in producing semantic equivalent versions of a heart quality of life questionnaire. Int J Nurs Pract. 2019;25(1). doi:10.1111/ijn.12715
- Papadakis NM, Aletta F, Kang J, Oberman T, Mitchell A, Stavroulakis GE. Translation and cross-cultural adaptation methodology for soundscape attributes – a study with independent translation groups from English to Greek. Appl Acoust. 2022;200:109031. doi:10.1016/j. apacoust.2022.109031
- van de Vijver FJR, Poortinga YH. On the study of culture in developmental science. Hum Dev. 2002;45(4):246-256. doi:10.1159/000064985
- Petkovic J, Epstein J, Buchbinder R, et al. Toward ensuring health equity: readability and cultural equivalence of OMERACT patient-reported outcome measures. J Rheumatol. 2015;42(12):2448-2459. doi:10.3899/jrheum. 141168
- Bundgaard K, Nisbeth Brøgger M. “Don’t fix bad translations”: a netnographic study of translators’ understandings of back translation in the medical domain. MonTi Monogr Traducción e Interpret. 2018;1(10):205-224. doi:10.6035/MonTI.2018.10.8
- Hawkins M, Cheng C, Elsworth GR, Osborne RH. Translation method is validity evidence for construct equivalence: analysis of secondary data routinely collected during translations of the Health Literacy Questionnaire (HLQ). BMC Med Res Methodol. 2020;20(1):130. doi:10.1186/ s12874-020-00962-8
- Epstein J, Osborne RH, Elsworth GR, Beaton DE, Guillemin F. Cross-cultural adaptation of the Health Education Impact Questionnaire: experimental study showed expert committee, not back-translation, added value. J Clin Epidemiol. 2015;68(4):360-369. doi:10.1016/j. jclinepi.2013.07.013
- Teig CJP, Bond MJ, Grotle M, et al. A novel method for the translation and cross-cultural adaptation of health-related quality of life patient-reported outcome measurements. Health Qual Life Outcomes. 2023;21(1):13. doi:10.1186/s12955-023-02089-y
- Tsai T-I, Luck L, Jefferies D, Wilkes L. Challenges in adapting a survey: ensuring cross-cultural equivalence. Nurse Res. 2018;26(1):28-32. doi:10.7748/nr.2018.e1581
- Jayawickreme E, Jayawickreme N, Goonasekera MA. Using focus group methodology to adapt measurement scales and explore questions of wellbeing and mental health. Intervention. 2012;10(2):156-167. doi:10.1097/WTF.0b013e328356f3c4
- Montenegro M, Valdez D, Crawford B, Turner R, Lo W-J, Jozkowski KN. Using a decentering framework to create English/Spanish surveys about abortion: insights into comparative survey research (CSR) for new survey development and recommendations for optimal use. Soc Sci J. 2022;1-15. doi:10.1080/03623319.2022.2092379
- Vizcaya-Moreno MF, Pérez-Cañaveras RM. Country validation of the CLES-Scale: linguistic and cultural perspectives. In: Saarikoski M, Strandell-Laine C, editors. The CLES-Scale: An Evaluation Tool for Healthcare Education. Springer; 2018:31-46.
- Erkut S. Developing multiple language versions of instruments for intercultural research. Child Dev Perspect. 2010;4(1):19-24. doi:10.1111/ j.1750-8606.2009.00111.x
- Marcondes FB, de Vasconcelos RA, Marchetto A, de Andrade ALL, Filho AZ, Etchebehere M. Translation and cross-cultural adaptation of the Rowe score for Portuguese. Acta Ortop Bras. 2012;20(6):346-350. doi:10.1590/S1413-78522012000600007
- Nepal GM, Shrestha A, Acharya R. Translation and cross-cultural adaptation of the Nepali version of the Rowland Universal Dementia Assessment Scale (RUDAS). J Patient-Reported Outcomes. 2019;3(1). doi:10.1186/s41687-019-0132-3
- Coll-Risco I, Camiletti-Moirón D, Acosta-Manzano P, Aparicio VA. Translation and cross-cultural adaptation of the Pregnancy Physical Activity Questionnaire (PPAQ) into Spanish. J Matern Neonatal Med. 2019;32(23):3954-3961. doi:10.1080/14767058.2018.1479849
- Pereira GIDN, Costa CDDS, Geocze L, Borim AA, Ciconelli RM, Camacho-Lobato L. Cross-cultural adaptation and validation for Portuguese (Brazil) of health related quality of life instruments specific for gastroesophageal reflux disease. Arq Gastroenterol. 2007;44(2):168-177. doi:10.1590/s0004-28032007000200016
- Grundström H, Rauden A, Olovsson M. Cross-cultural adaptation of the Swedish version of Endometriosis Health Profile-30. J Obstet Gynaecol (Lahore). 2020;40(7):969-973. doi:10.1080/01443615.2019.1676215
- Polesello GC, Godoy GF, De Castro Trindade CA, De Queiroz MC, Honda E, Ono NK. Translation and cross-cultural adaptation of the modified Hip outcome tool (mhot) into Portuguese. Acta Ortop Bras. 2012;20(2):88-92. doi:10.1590/S1413-78522012000200006
- Muquith MA, Islam MN, Haq SA, Ten Klooster PM, Rasker JJ, Yunus MB. Cross-cultural adaptation and validation of a Bengali version of the modified fibromyalgia impact questionnaire. BMC Musculoskelet Disord. 2012;13(1):1. doi:10.1186/1471-2474-13-157
- Toma G, Guetterman TC, Yaqub T, Talaat N, Fetters MD. A systematic approach for accurate translation of instruments: experience with translating the Connor-Davidson Resilience Scale into Arabic. Methodol Innov. 2017;10(3):205979911774140. doi:10.1177/2059799117741406
- Poot CC, Meijer E, Fokkema M, Chavannes NH, Osborne RH, Kayser L. Translation, cultural adaptation and validity assessment of the Dutch version of the eHealth Literacy Questionnaire: a mixed-method approach. BMC Public Health. 2023;23(1):1006. doi:10.1186/s12889-023-15869-4
- Hasani L, Santoso H, Junus K. Instrument development for investigating students’ intention to participate in online discussion forums: crosscultural and context adaptation sing SEM. J Educ Online. 2021;18(3). doi:10.9743/JEO.2021.18.3.9
- Bundgaard K, Brøgger MN. Who is the back translator? An integrative literature review of back translator descriptions in cross-cultural adaptation of research instruments. Perspectives (Montclair). 2019;27(6):833-845. doi:10.1080/0907676X.2018.1544649
- Geisinger KF. Cross-cultural normative assessment: translation and adaptation issues influencing the normative interpretation of assessment instruments. Psychol Assess. 1994;6(4):304-312. doi:10.1037/1040-3590.6.4.304
- van Widenfelt BM, Treffers PDA, de Beurs E, Siebelink BM, Koudijs E. Translation and cross-cultural adaptation of assessment instruments used in psychological research with children and families. Clin Child Fam Psychol Rev. 2005;8(2):135-147. doi:10.1007/s10567-005-4752-1
- Son J. Back translation as a documentation tool. Int J Transl Interpret Res. 2018;10(2):89-100. doi:10.12807/ti.110202.2018.a07
- Coster WJ, Mancini MC. Recommendations for translation and cross-cultural adaptation of instruments for occupational therapy research and practice. Rev Ter Ocup da Univ São Paulo. 2015;26(1):50. doi:10.11606/issn.2238-6149.v26i1p50-57
- Ortiz-Gutiérrez S, Cruz-Avelar A. Translation and cross-cultural adaptation of health assessment tools. Actas Dermo-Sifiliográficas (English Ed. 2018;109(3):202-206. doi:10.1016/j.adengl.2018.02.003
- Ramada-Rodilla JM, Serra-Pujadas C, Delclós-Clanchet GL. Adaptación cultural y validación de cuestionarios de salud: revisión y recomendaciones metodológicas. Salud Publica Mex. 2013;55(1):57-66. doi:10.1590/S0036-36342013000100009
- Prakash V, Shah S, Hariohm K. Cross-cultural adaptation of patient-reported outcome measures: a solution or a problem? Ann Phys Rehabil Med. 2019;62(3):174-177. doi:10.1016/j.rehab.2019.01.006
- Fortes CPDD, Araújo de QC AP. Check list para tradução e adaptação transcultural de questionários em saúde. Cad Saúde Coletiva. 2019;27 (2):202-209. doi:10.1590/1414-462
- Hernández A, Hidalgo MD, Hambleton RK, Gómez-Benito J. International test commission guidelines for test adaptation: a criterion checklist. Psicothema. 2020;32(3):390-398. doi:10.7334/psicothema2019.306
- DuBay M, Watson LR. Translation and cultural adaptation of parent-report developmental assessments: improving rigor in methodology. Res Autism Spectr Disord. 2019;62:55-65. doi:10.1016/j.rasd.2019.02.005
- Hall DA, Zaragoza Domingo S, Hamdache LZ, et al. A good practice guide for translating and adapting hearing-related questionnaires for different languages and cultures. Int J Audiol. 2018;57(3):161-175. doi:10.1080/14992027.2017.1393565
- José M, Elosua P, Hambleton RK. Directrices para la traducción y adaptación de los tests: segunda edición. Psicothema. 2013;25(2):151-157. doi:10.7334/psicothema2013.24
- Santo RM, Ribeiro-Ferreira F, Alves MR, Epstein J, Novaes P. Enhancing the cross-cultural adaptation and validation process: linguistic and psychometric testing of the Brazilian-Portuguese version of a self-report measure for dry eye. J Clin Epidemiol. 2015;68(4):370-378. doi:10.1016/j.jclinepi.2014.07.009
- Arafat S, Chowdhury H, Qusar M, Hafez M. Cross cultural adaptation and psychometric validation of research instruments: a methodological review. J Behav Heal. 2016;5(3):129. doi:10.5455/jbh.20160615121755
- Kyriazos TA. Applied psychometrics: the 3-Faced Construct Validation Method, a routine for evaluating a factor structure. Psychology. 2018;09 (08):2044-2072. doi:10.4236/psych.2018.98117
- Plonsky L, Kim Y. Task-based learner production: a substantive and methodological review. Annu Rev Appl Linguist. 2016;36:73-97. doi:10.1017/S0267190516000015
- Peters MD, Godfrey C, McInerney P, Soares CB, Khalil H, Parker D. Development of a scoping review protocol. In: Aromataris E, Munn Z, editors. Joanna Briggs Institute Reviewer’s Manual. : The Joanna Briggs Institute; 2020
- Shekelle PG, Woolf SH, Eccles M, Grimshaw J. Developing clinical guidelines. West J Med. 1999;170(6):348-351.
- OECD Eurostat and World Health Organization. Classification of Health Care Providers (ICHA-HP). In: A System of Health Accounts 2011. OECD Publishing; 2017: 121-152. doi:10.1787/9789264270985-en
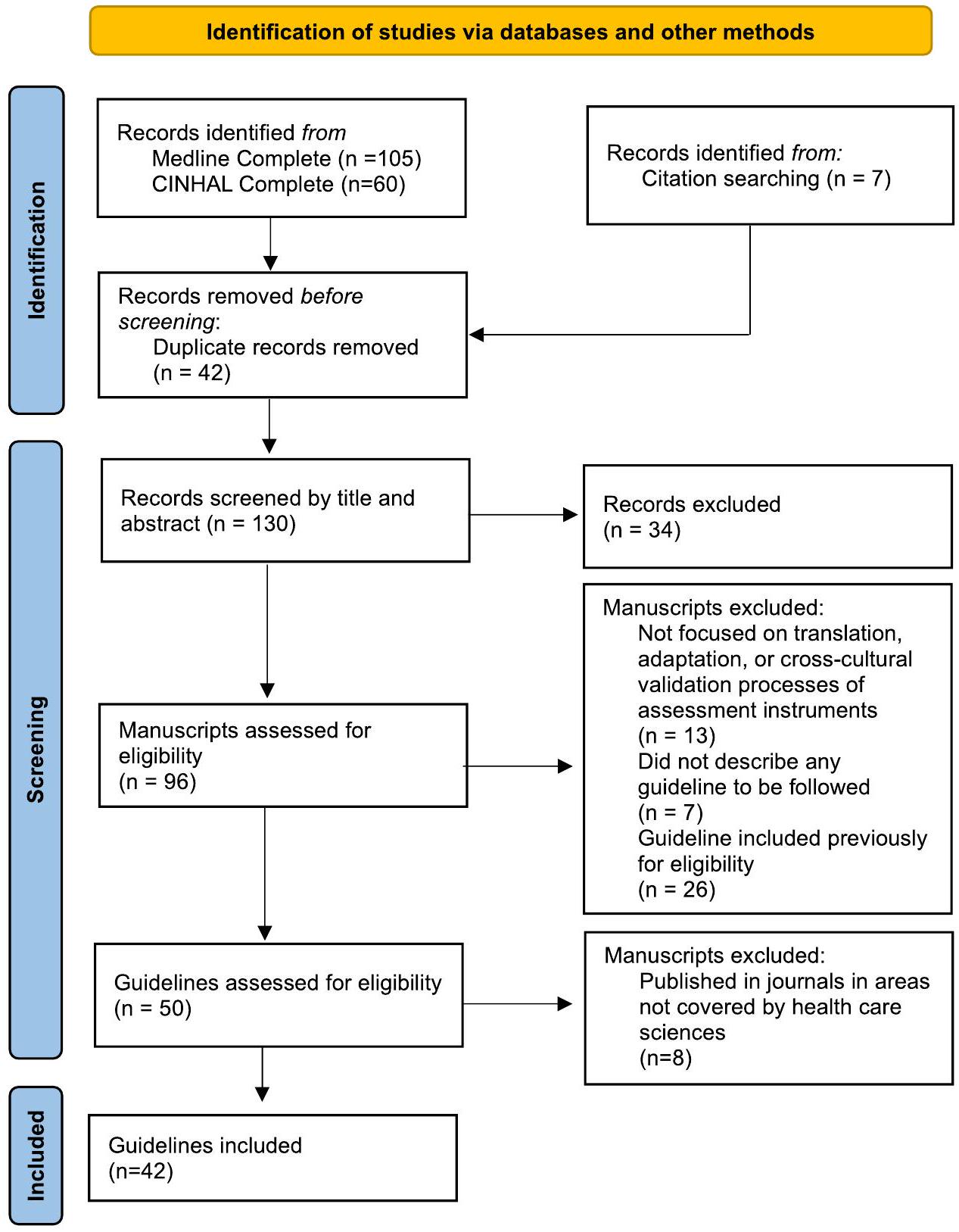
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021: n71. doi:10.1136/bmj.n71
- Schreier M. Qualitative content analysis. In: Flick U, editor. The SAGE Handbook of Qualitative Data Analysis. Sage Publications; 2014:170-183.
- World Health Organization. Introduction. In: WHO Handbook for Guideline Developement. 2nd ed. World Health Organization; 2014:2
- Pernambuco L, Espelt A, Magalhães Junior HV, Lima de KC. Recomendações para elaboração, tradução, adaptação transcultural e processo de validação de testes em fonoaudiologia. CoDAS. 2017;29(3). doi:10.1590/2317-1782/20172016217
- International Test Comission. ITC Guidelines for Translating and Adapting Tests. International Test Comission; 2017.
- Tafforeau J, Cobo ML, Tolonen H, Christa S-N, Tinto A. Guidelines for the Development and Criteria for the Adoption of Health Survey Instruments; 2015. Available from: https://ec.europa.eu/health/ph_information/dissemination/reporting/healthsurveys_en.pdf. Accessed May 16, 2024.
- Høegh MC, Høegh S-M. Trans-adapting outcome measures in rehabilitation: cross-cultural issues. Neuropsychol Rehabil. 2009;19(6):955-970. doi:10.1080/09602010902995986
- McKenna SP. Measuring patient-reported outcomes: moving beyond misplaced common sense to hard science. BMC Med. 2011;9(1):86. doi:10.1186/1741-7015-9-86
- Prinsen CAC, Mokkink LB, Bouter LM, et al. COSMIN guideline for systematic reviews of patient-reported outcome measures. Qual Life Res. 2018;27(5):1147-1157. doi:10.1007/s11136-018-1798-3
- Mokkink LB, Prinsen CAC, Patrick DL, et al. COSMIN Study Design Checklist for Patient-Reported Outcome Measurement Instruments; 2019. Available from: https://www.cosmin.nl/wp-content/uploads/COSMIN-study-designing-checklist_final.pdf. Accessed May 16, 2024.
- Gjersing L, Caplehorn JR, Clausen T. Cross-cultural adaptation of research instruments: language, setting, time and statistical considerations. BMC Med Res Methodol. 2010;10(1):13. doi:10.1186/1471-2288-10-13
- Chávez LM, Canino GC Toolkit on Translating and Adapting Instruments; 2005. Available from: https://www.hsri.org/files/uploads/publica tions/PN54_Translating_and_Adapting.pdf. Accessed May 16, 2024.
- World Health Organization. WHO guidelines on translation: process of translation and adaptation of instruments; 2019. Available from: http:// www.who.int/substance_abuse/research_tools/translation/en/. Accessed May 16, 2024.
- Wild D, Eremenco S, Mear I, et al. Multinational trials-Recommendations on the translations required, approaches to using the same language in different countries, and the approaches to support pooling the Data: the ISPOR patient-reported outcomes translation and linguistic validation good research practices task force Report. Value Heal. 2009;12(4):430-440. doi:10.1111/j.1524-4733.2008.00471.x
- Swami V, Barron D. Translation and validation of body image instruments: challenges, good practice guidelines, and reporting recommendations for test adaptation. Body Image. 2019;31:204-220. doi:10.1016/j.bodyim.2018.08.014
- Kuliś D, Bottomley A, Velikova G, Greimel E, Koller M. EORTC Quality of Life Group Translation Procedure; 2017. Available from: https:// www.eortc.org/app/uploads/sites/2/2018/02/translation_manual_2017.pdf. Accessed May 16, 2024.
- Ohrbach R, Ohrbach J, Jezewski M, John MT, Lobbezoo F. Guidelines for Establishing Cultural Equivalency of Instruments; 2013. Available from: https://ubwp.buffalo.edu/rdc-tmdinternational/wp-content/uploads/sites/58/2017/01/Guidelines-for-Translation-and-Cultural-Equivalency-of-Instruments-2013_05_118608.pdf. Accessed May 16, 2024.
- Baker DL, Melnikow J, Ying Ly M, Shoultz J, Niederhauser V, Diaz-Escamilla R. Translation of health surveys using mixed methods. J Nurs Scholarsh. 2010;42(4):430-438. doi:10.1111/j.1547-5069.2010.01368.x
- Biering-Sørensen F, Alexander MS, Burns S, et al. Recommendations for translation and reliability testing of international spinal cord injury data sets. Spinal Cord. 2011;49(3):357-360. doi:10.1038/sc.2010.153
- Koller M, Aaronson NK, Blazeby J, et al. Translation procedures for standardised quality of life questionnaires: the European Organisation for Research and Treatment of Cancer (EORTC) approach. Eur J Cancer. 2007;43(12):1810-1820. doi:10.1016/j.ejca.2007.05.029
- Eremenco SL, Cella D, Arnold BJ. A comprehensive method for the translation and cross-cultural validation of health status questionnaires. Eval Health Prof. 2005;28(2):212-232. doi:10.1177/0163278705275342
- Dhamani KA, Richter MS. Translation of research instruments: research processes, pitfalls and challenges. Afr J Nurs Midwifery. 2011;13 (1):3-13.
- Sperber AD. Translation and validation of study instruments for cross-cultural research. Gastroenterology. 2004;126:S124-S128. doi:10.1053/j. gastro.2003.10.016
- Aaronson N, Alonso J, Burnam A, et al. Assessing health status and quality-of-life instruments: attributes and review criteria. Qual Life Res. 2002;11(3):193-205. doi:10.1023/a:1015291021312
- Organisation for Economic Co-operation and Development. Translation and adaptation guidelines for PISA 2012; 2012. Available from: https:// www.oecd.org/pisa/pisaproducts/49273486.pdf. Accessed May 16, 2024.
- Swami V, Todd J, Barron D. Translation and validation of body image instruments: an addendum to Swami and Barron (2019) in the form of frequently asked questions. Body Image. 2021;37:214-224. doi:10.1016/j.bodyim.2021.03.002
- Terwee CB, Prinsen CAC, Chiarotto A, et al. COSMIN methodology for evaluating the content validity of patient-reported outcome measures: a Delphi study. Qual Life Res. 2018;27(5):1159-1170. doi:10.1007/s11136-018-1829-0
- Yasir ASM. Cross Cultural Adaptation & Psychometric Validation of Instruments: step-wise Description. Int J Psychiatry. 2016;1(1). doi:10.33140/IJP/01/01/00001
- Regnault A, Herdman M. Using quantitative methods within the Universalist model framework to explore the cross-cultural equivalence of patient-reported outcome instruments. Qual Life Res. 2015;24(1):115-124. doi:10.1007/s11136-014-0722-8
- Mokkink LB, Terwee CB, Knol DL, et al. The COSMIN checklist for evaluating the methodological quality of studies on measurement properties: a clarification of its content. BMC Med Res Methodol. 2010;10(1):22. doi:10.1186/1471-2288-10-22
- Mokkink LB, Terwee CB, Patrick DL, et al. The COSMIN checklist for assessing the methodological quality of studies on measurement properties of health status measurement instruments: an international Delphi study. Qual Life Res. 2010;19(4):539-549. doi:10.1007/s11136-010-9606-8
- Terwee CB, Bot SDM, de Boer MR, et al. Quality criteria were proposed for measurement properties of health status questionnaires.
Clin Epidemiol. 2007;60(1):34-42. doi:10.1016/j.jclinepi.2006.03.012 - Möhler R, Bartoszek G, Köpke S, Meyer G. Proposed criteria for reporting the development and evaluation of complex interventions in healthcare (CReDECI): guideline development. Int
Nurs Stud. 2012;49(1):40-46. doi:10.1016/j.ijnurstu.2011.08.003 - American Educational Research Association. Standards for Educational and Psychological Testing. American Educational Research Association; 2014.
- Gudmundsson E. Guidelines for translating and adapting psychological instruments. Nord Psychol. 2009;61(2):29-45. doi:10.1027/19012276.61.2.29
- Forsyth BH, Kudela MS, Levin K, Lawrence D, Willis GB. Methods for translating an English-language survey questionnaire on tobacco use into mandarin, Cantonese, Korean, and Vietnamese. Field Methods. 2007;19(3):264-283. doi:10.1177/1525822X07302105
- American Psychological Association. Criteria for practice guideline development and evaluation. Am Psychol. 2002;57(12):1048-1051. doi:10.1037/0003-066X.57.12.1048
- Iliescu D. Pre-condition guidelines. In: Adapting Tests in Linguist and Cultural Adaptations. Cambridge University Press; 2017:87.
- Iliescu D. Documentation guidelines. In: Adapting Tests in Linguist and Cultural Adaptations. Cambridge University Press; 2017:105.
- Hambletom RK. Issues, designs and tecnical guidelines for adaptating tests into multiples languages and cultures. In: Hembletom RK, Merenda PF, Spilberg CD, editors. Adapting Educational and Psychological Tests for Cross-Cultural Assessment. Lawrence Erlbaum Associates; 2005:3-38.
- Oakland T. Selected ethical issues relevant to test adaptations. In: Hambletom RK, Merenda PF, Spielberg CD, editors. Adapting Educational and Psychological Tests for Cross-Cultural Assessment. Lawrence Erlbaum Associates; 2005:80.
- Brislin RW, Lonner WJ, Thorndike RM. Cross cultural research methods. In: Transcultural Psychiatric Research Review. Vol. 12. John Wiley ans Sons;1973:7-10. doi:10.1177/136346157501200101
- Beaton D, Bombardier C, Guillemin F, Ferraz MB. Recommendations for the cross-cultural adaptation of the DASH & QuickDASH outcome measure. Inst Work Heal. 2007;45.
- Ozolins U, Hale S, Cheng X, Hyatt A, Schofield P. Translation and back-translation methodology in health research: a critique. Expert Rev Pharmacoecon Outcomes Res. 2020;20(1):69-77. doi:10.1080/14737167.2020.1734453
- Bornman J, Sevcik RA, Romski M, Pae HK. Successfully translating language and culture when adapting assessment measures. J Policy Pract Intellect Disabil. 2010;7(2):111-118. doi:10.1111/j.1741-1130.2010.00254.x
- Simonsen E, Mortensen EL. Difficulties in translation of personality scales. J Pers Disord. 1990;4(3):290-296. doi:10.1521/pedi.1990.4.3.290
- Tanzer NK. Developing Tests for Use in Multiple Languages and Cultures: A Plea for Simultaneous Development. Lawrence Erlbaum Associates; 2005.
- Bennett PM. Reviewing translated scales: backtranslation under the spotlight. Transl Matters. 2022;4(125-144).
- van de Vijver F, Tanzer NK. Bias and equivalence in cross-cultural assessment: an overview. Eur Rev Appl Psychol. 2004;54(2):119-135. doi:10.1016/j.erap.2003.12.004
- de Klerk S, Jerosch-Herold C, Buchanan H, van Niekerk L. Shared decision making and the practice of community translation in presenting a pre-final Afrikaans for the Western Cape Disabilities of the Arm, Shoulder and Hand (DASH) questionnaire: a proposal for improved translation and cross-cultural adaptation. J Patient-Reported Outcomes. 2019;3(1):52. doi:10.1186/s41687-019-0144-z
- Erkut S. Developing multiple language versons of instruments for intercultural research. In: Ji M, editor. Cross-Cultural Health Translation: Exploring Methodological and Digital Tools. Routledge; 2019:9-10.
- Silverblatt A, Zlobin N. Production elements. In: International Communications: A Media Literacy Approach. Routledge; 2015:108.
- St Amant K. A new web for the new millennium. In: Lipson C, Day M, editors. Technical Communication and the World Wide Web. Lawrence Erlbaum Associates Publishers; 2005:148.
- Drasgow F, Probst TM. The psicometrics of adaptation: evaluating measurement equivalance across languages and cultures. In: Hambletom RK, Merenda PF, Spielberg CD, editors. Adapting Educational and Psychological Tests for Cross-Cultural Assessment. Lawrence Erlbaum Associates Publishers; 2005:361.
- Erkut S, Alarcón O, Coll CG, Tropp LR, García HAV. The dual-focus approach to creating bilingual measures. J Cross Cult Psychol. 1999;30 (2):206-218. doi:10.1177/0022022199030002004
- van der Vidjer F, Leung K. Equivalence and bias: a review of concepts, models, and data analytic procedures. In: Matsumoto D, van de Vijver F, editors. Cross-Cultural Research Methods in Psychology. Cambridge University Press; 2011:17-44.
- American Educational Research Association. Test design and development. Standards for Educational and Psychological Testing; American Educational Research Association; 2014. 81-84.
- van Teijlingen ER, Hundley V. Pilot study. In: Lewis-Back MS, Bryman A, Liao TF, editors. The SAGE Encyclopedia of Social Science Research Methods. Vol II. Sage; 2004:823-824.
- Gallagher PM. Pretest. In: Lewis-Back MS, Bryman A, Liao TF, editors. The SAGE Encyclopedia of Social Science Research Methods. Vol II. Sage Publications; 2004:853-854.
- Korabik K, van Rhijn T. Best practices in scale translation and establisment mesurement equivalence. In: Shockey KM, Shen W, Johnson RC, editors. The Cambridge Handbook of the Global Work-Family Interface. Cambridge University Press; 2018:212-229.
- Willis G. Pretesting of health survey questionnaires: cognitive interviewing, usability testing, and behavior coding. In: Johnson TP, editor. Handbook on Health Survey Methods. Willey; 2015:221.
- Rodrigues IB, Adachi JD, Beattie KA, MacDermid JC. Development and validation of a new tool to measure the facilitators, barriers and preferences to exercise in people with osteoporosis. BMC Musculoskelet Disord. 2017;18(1):540. doi:10.1186/s12891-017-1914-5
- Perneger TV, Courvoisier DS, Hudelson PM, Gayet-Ageron A. Sample size for pre-tests of questionnaires. Qual Life Res. 2015;24(1):147-151. doi:10.1007/s11136-014-0752-2
- Holyk GG. Question testing methods. In: Encyclopedia of Survey Research Methods. Vol II. Sage; 2008:658-659.
- van der Zouwen J, Smit JH, et al. Evaluating survey questions by analyzing patterns of behavior codes and question-answer sequences: a diagnostic approach. In: Presser S, Rothgeb JM, Couper MP, editors. Methods for Testing and Evaluating Survey Questionnaires. Wiley Interscience; 2004. 124.
- Wynd CA, Schmidt B, Schaefer MA. Two quantitative approaches for estimating content validity. West J Nurs Res. 2003;25(5):508-518. doi:10.1177/0193945903252998
- Polit DF, Beck CT. Measurement and data quality. In: Nursing Research: Generating and Assessing Evidence for Nursing Practice. 9th ed. Wolker Klumer / Lippincott Williams & Wilkins; 2012:334.
- Linden B, Stuart H. Preliminary analysis of validation evidence for two new scales assessing teachers’ confidence and worries related to delivering mental health content in the classroom. BMC Psychol. 2019;7(1):32. doi:10.1186/s40359-019-0307-y
- Rubio DM, Berg-Weger M, Tebb SS, Lee ES, Rauch S. Objectifying content validity: conducting a content validity study in social work research. Soc Work Res. 2003;27(2):94-104. doi:10.1093/swr/27.2.94
- Almanasreh E, Moles R, Chen TF. Evaluation of methods used for estimating content validity. Res Soc Adm Pharm. 2019;15(2):214-221. doi:10.1016/j.sapharm.2018.03.066
- DesRoches D. Establishment survey. In: Lavrakas PJ, editor. Encyclopedia of Survey Research Methods. Sage; 2008:124.
- Kelly J, Lavrakas PJ. Debriefing. In: Encyclopedia of Survey Research Methods. Vol I. Sage; 2008:181-182.
- Beatty P. Developing measures of health behavior and he alth service utilization. In: Johnson TP, editor. Handbook on Health Survey Methods. Willey; 2015:186-187.
- Polit DF, Beck CT, Owen SV. Is the CVI an acceptable indicator of content validity? Appraisal and recommendations. Res Nurs Health. 2007;30 (4):459-467. doi:10.1002/nur. 20199
- Yusoff MSB. ABC of content validation and content validity index calculation. Educ Med J. 2019;11(2):49-54. doi:10.21315/eimj2019.11.2.6
- Polit DF, Beck CT. Qualitative research design and approaches. In: Nursing Research: Generating and Assessing Evidence for Nursing Practice. 10th ed. Wolters Kluwer; 2017:489.
- Lawshe CH. A quantitative approach to content validity. Pers Psychol. 1975;28(4):563-575. doi:10.1111/j.1744-6570.1975.tb01393.x
- Ayre C, Scally AJ. Critical values for Lawshe’s Content Validity Ratio. Meas Eval Couns Dev. 2014;47(1):79-86. doi:10.1177/ 0748175613513808
- Stone KS, Frazier SK. Evaluation of measurement precision, accuracy, and error in biophysical data. In: Waltz CF, Strickland OL, Lenz ER, editors. Measurement in Nursing and Health Research. 4th ed. Springer Publishing; 2010:387-390.
- Cicchetti DV. On a model for assessing the security of infantile attachment: issues of observer reliability and validity. Behav Brain Sci. 1984;7 (1):149-150. doi:10.1017/S0140525X00026558
- Fleiss JL. Measuring nominal scale agreement among many raters. Psychol Bull. 1971;76(5):378-382. doi:10.1037/h0031619
- Kottner J, Audige L, Brorson S, et al. Guidelines for reporting reliability and agreement studies (GRRAS) were proposed. Int J Nurs Stud. 2011;48(6):661-671. doi:10.1016/j.ijnurstu.2011.01.016
- Osborne JW. Best Practices in Exploratory Factor Analysis. CreateSpace Independent Publishing; 2014.
- Lloret-Segura S, Ferreres-Traver A, Hernández-Baeza A, Tomás-Marco I. El análisis factorial exploratorio de los ítems: una guía práctica, revisada y actualizada. An Psicol. 2014;30(3). doi:10.6018/analesps.30.3.199361
- Field A. Why is my evil lecturer forcing me to learn statistics? In: Discovering Statistics Using IBM SPSS Statistics. 5th ed. Sage Publications; 2018:70-71.
- Kothari CR. Multivariate analysis techniques. In: Research Methodology: Methods and Techniques. 2nd ed. New Age Publisher; 2004:322.
- Haig BD. Exploratory factor analysis, theory generation, and scientific method. In: Method Matters in Psychology: Essays in Applied Philosophy of Science. Springer; 2018:65-88.
- Haig BD. Exploratory factor analysis, theory generation, and scientific method. Multivariate Behav Res. 2005;40(3):303-329. doi:10.1207/ s15327906mbr4003_2
- Råholm M-B. Abductive reasoning and the formation of scientific knowledge within nursing research. Nurs Philos. 2010;11(4):260-270. doi:10.1111/j.1466-769X.2010.00457.x
- Prudon P. Confirmatory factor analysis as a tool in research using questionnaires: a critique. Compr Psychol. 2015;4:03.CP.4.10. doi:10.2466/ 03.CP.4.10
- Eldridge J. Reliability, validity and trustworthiness. In: Boswell C, Cannon S, editors. Introduction to Nursing Research: Incorporating Evidence-Based Practice. 5th ed. Jones & Bartlettt Publishers; 2020:271-294.
- Abu-Bader SH. Working with SPSS. In: Using Statistical Methods in Social Science Research: With a Complete SPS Guide. 2nd ed. Oxford University Press; 2021:56.
- Perrin KM. Reliability and validity. In: Principles of Planning, Evaluation and Research for Health Care Programmes. 2nd ed. Jones &: Bartlett Learning; 2022:141.
- Grove SK. Quantitative measurement concepts. In: Gray JR, Grove SK, editors. Burns & Groves the Practice of Nursing Research: Appraisal, Synthesis, and Generation of Evidence. 9th ed. Elsevier; 2021:462.
- Hair J, Hollingsworth CL, Randolph AB, Chong AYL. An updated and expanded assessment of PLS-SEM in information systems research. Ind Manag Data Syst. 2017;117(3):442-458. doi:10.1108/IMDS-04-2016-0130
- Field A. Exploratory factor analysis. In: Discovering Statistics Using IBM SPSS Statistics (North American Edition). 5th ed. Sage; 2018:1390-1391.
- Clark LA, Watson D. Constructing validity: new developments in creating objective measuring instruments. Psychol Assess. 2019;31 (12):1412-1427. doi:10.1037/pas0000626
- Moreira J. Questionários: Teoria e Prática. Almedina; 2004.
- Streiner DL, Norman GR, Cainey J. Basic concepts. In: Health Measurement Scales: A Practical Guide to Their Development and Use. 5th ed. Oxford University Press; 2015:8.
- Bryant F. Assessing the validity of measurement. In: Grimm LG, Yarnold PR, editors. Reading and Understanding More Multivariate Statistics. American Psychological Association; 2002:99-146.
- Marôco J, Etapas da análise de equações estruturais. 3rd. Análise de Equações Estruturais, 2021:55
- Sommer I, Larsen K, Nielsen CM, Stenholt BV, Bjørk IT. Improving clinical nurses’ development of supervision skills through an action learning approach. Nurs Res Pract. 2020;2020:1-10. doi:10.1155/2020/9483549
- Bowling A. Quality of life: concepts, measurements and patient perception. In: Research Methods in Health: Investigating Health and Health Services. 4th ed. Open University Press; 2014:54.
- Bot SM, Terwee CB, van der Windt DAWM, Boute LM, Dekker J, Vet de HCW. Psychometric evaluation of self-report questionnaires: the development of a checklist. In: Adér HJ, Mellenberg C, editors. Proceedings of the Second Workshop on Research Methodology. Vu University; 2003:161-168.
- Powers BA, Knapp TR. Face validity. In: Dictionary of Nursing Theory and Research. 3rd ed. Spinger Publishing; 2006:63.
- Soeken WKL. Validity of measures. In: Measurement in Nursing and Health Research. 4th ed. Springer Publishing Company; 2010:163.
- Streiner DL, Norman GR, Cairney J. Seleting the itens. In: Health Measurement Scales: A Practical Guide to Their Development and Use. 5th. Oxford University Press; 2015:92.
- Zumbo BD. Validity as contextualized and pragmatic explanation, and its implications for validation practice. In: Lissitz RW, editor. The Concept of Validity: Revisions, New Directions, and Applications. Information Age Publishing; 2009:68.
- Chan EKH. Standards and guidelines for validation practices: development and evaluation of measurement instruments. In: Zumbo BD, Chan EKH, editors. Validity and Validation in Social, Behavioral, and Health Sciences. Springer Publishing; 2014:9-24.
- Mokkink LB, Terwee CB, Knol DL, et al. Taxonomy and definitions. COSMIN Checklist Manual. 2012;9.
انشر عملك في هذه المجلة
DOI: https://doi.org/10.2147/jmdh.s419714
PMID: https://pubmed.ncbi.nlm.nih.gov/38840704
Publication Date: 2024-05-01
Translation, Cross-Cultural Adaptation, and Validation of Measurement Instruments: A Practical Guideline for Novice Researchers
Abstract
Cross-cultural validation of self-reported measurement instruments for research is a long and complex process, which involves specific risks of bias that could affect the research process and results. Furthermore, it requires researchers to have a wide range of technical knowledge about the translation, adaptation and pre-test aspects, their purposes and options, about the different psychometric properties, and the required evidence for their assessment and knowledge about the quantitative data processing and analysis using statistical software. This article aimed: 1) identify all guidelines and recommendations for translation, cross-cultural adaptation, and validation within the healthcare sciences; 2 ) describe the methodological approaches established in these guidelines for conducting translation, adaptation, and cross-cultural validation; and 3) provide a practical guideline featuring various methodological options for novice researchers involved in translating, adapting, and validating measurement instruments. Forty-two guidelines on translation, adaptation, or cross-cultural validation of measurement instruments were obtained from “CINAHL with Full Text” (via EBSCO) and “MEDLINE with Full Text”. A content analysis was conducted to identify the similarities and differences in the methodological approaches recommended. Bases on these similarities and differences, we proposed an eight-step guideline that includes: a) forward translation; 2) synthesis of translations; 3) back translation; 4) harmonization; 5) pre-testing; 6) field testing; 7) psychometric validation, and 8) analysis of psychometric properties. It is a practical guideline because it provides extensive and comprehensive information on the methodological approaches available to researchers. This is the first methodological literature review carried out in the healthcare sciences regarding the methodological approaches recommended by existing guidelines.
Introduction
reliable measuring instruments,
Concepts and Specifics Terms
Types of Equivalence
equivalence of Herdman et al.
Typologies of Biases
Methodological Approaches
Research Rationale and Aims
Materials and Methods
Identification of Existing Guidelines

Content Analysis of Existing Guidelines
Proposal for a Practical Guideline
a practical guideline based from an universalist perspective,
Results
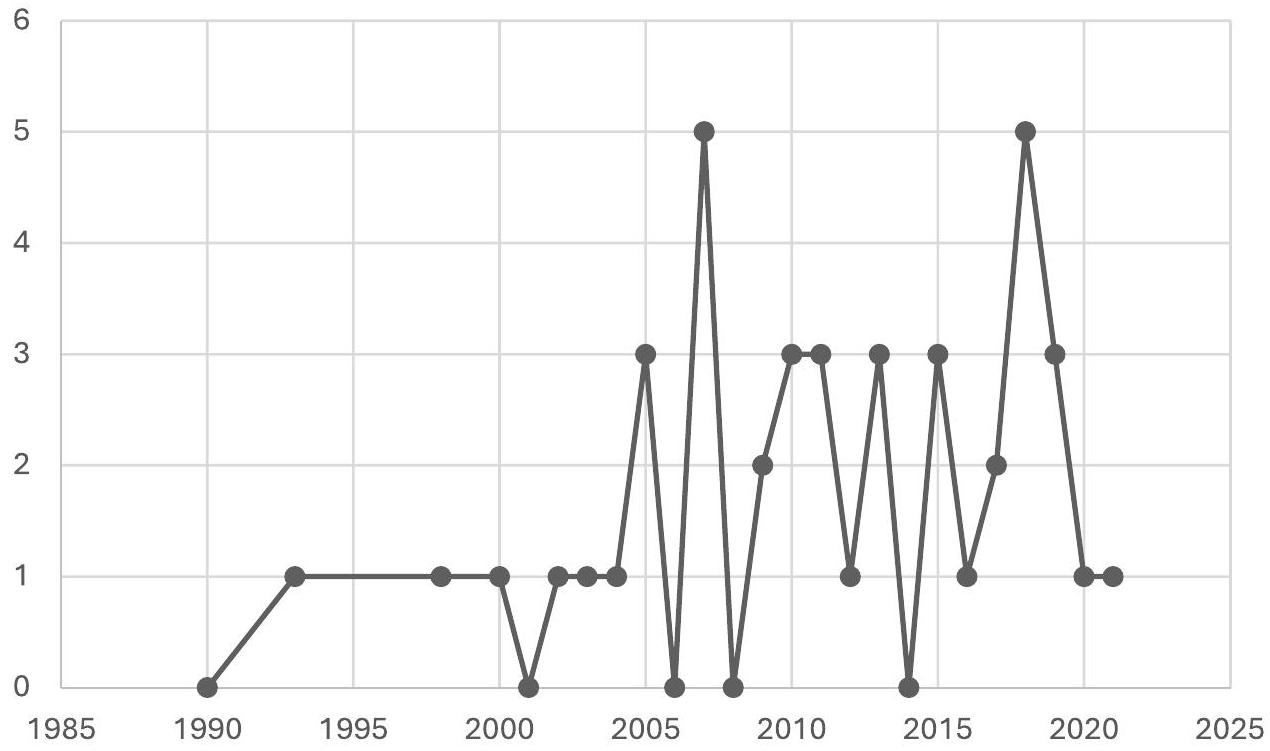
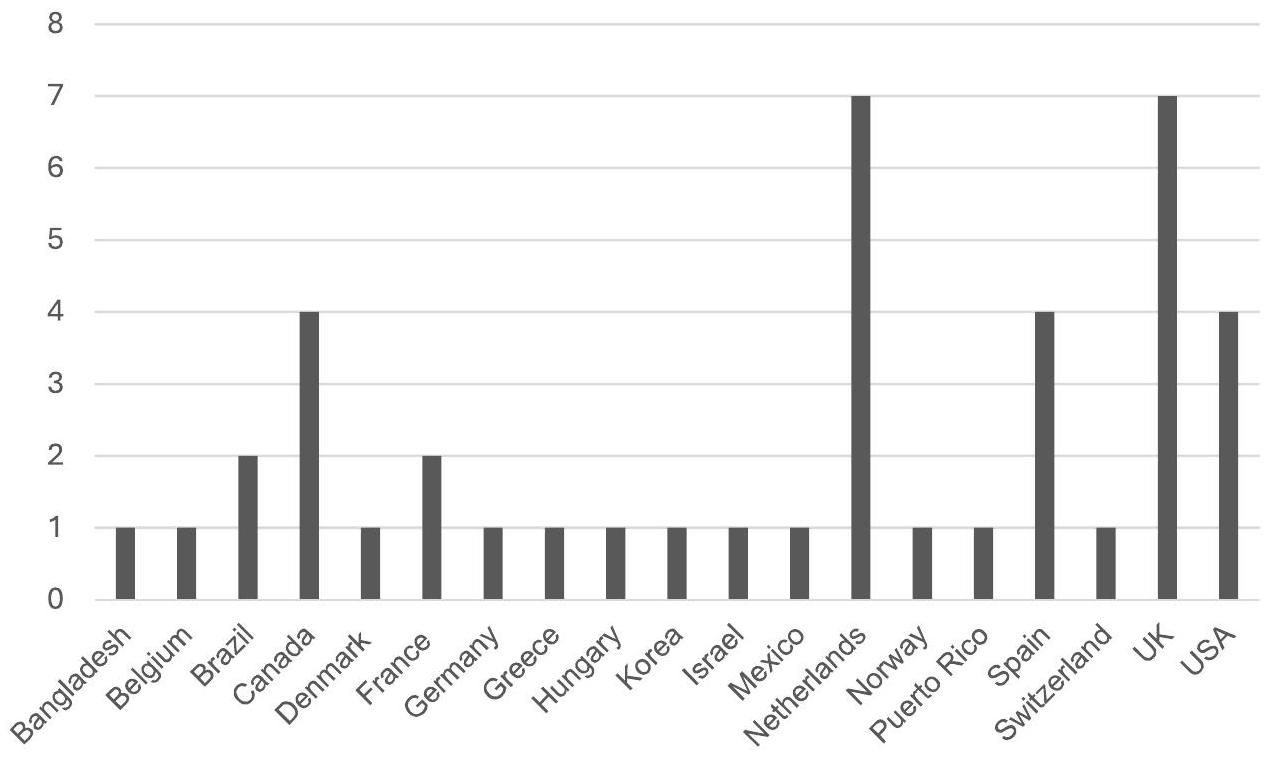
General Information


of the instrument and as supplementary material in the publication of a scientific article reporting on the process of crosscultural adaptation of the measurement instrument.
Cross-Cultural Translation
the constructs with elements not involved in the previous translations but with extensive knowledge of the constructs to be measured.
Cross-Cultural Adaptation
from pre-test participants,
Cross-Cultural Validation
authors refer generically to its evaluation.
Discussion
Process Documentation
written evidence of the probable relevance of the instruments to participants from the “target culture”, as well as of the operational equivalence of the instrument. The documentation of decisions may be empirical but most will be theoretical in nature.
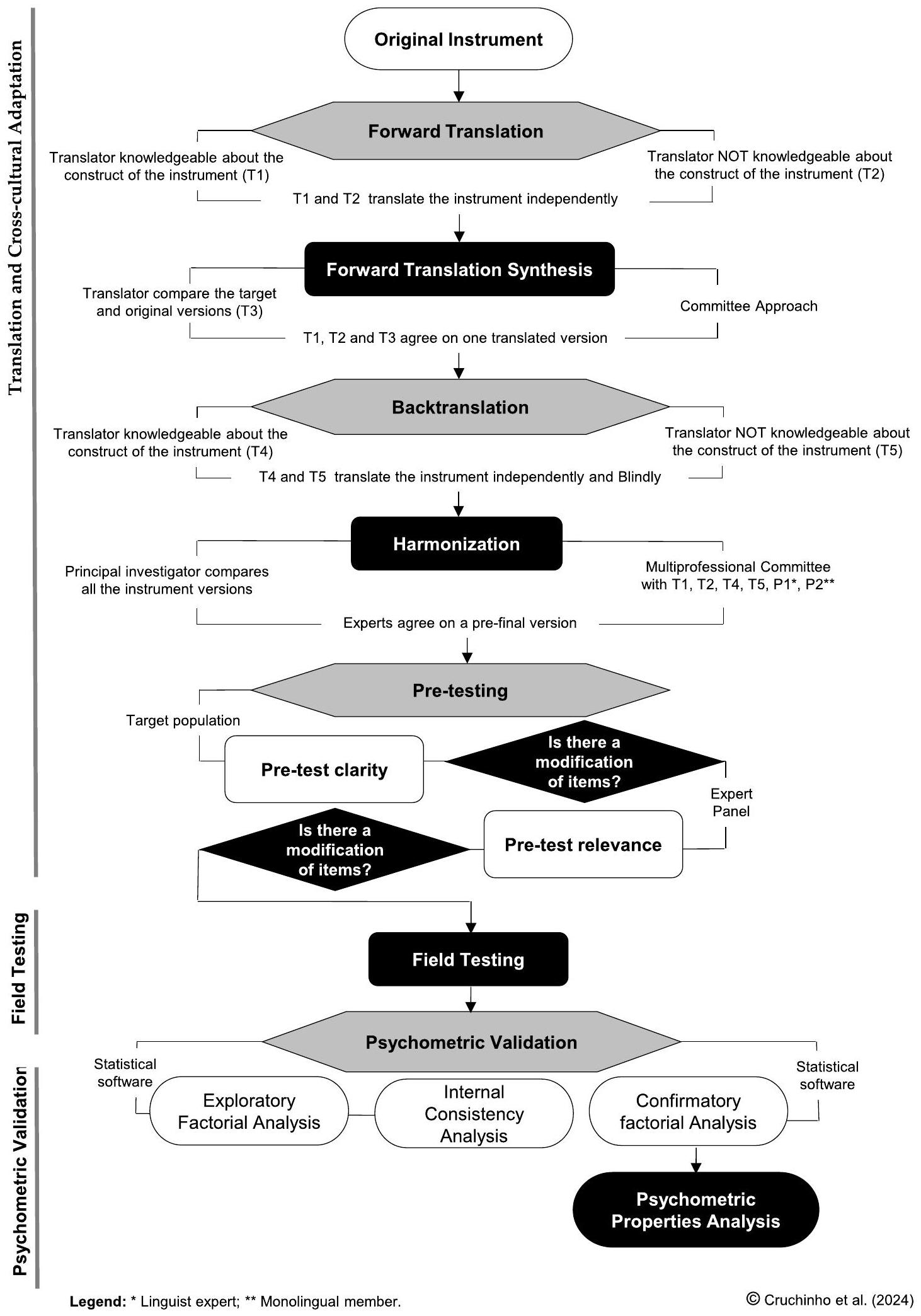
Stages of the Process
Forward Translation

Forward Translation Synthesis
Back Translation
of the original document are generated. Regardless of whether minor discrepancies occur between the two versions, the main aspect that needs to be analyzed in the next step is whether there is a change in meaning between the items in the back translation and the items in the “original instrument”.
Harmonization
Pre-testing
convenience sampling.
Field Testing
Psychometric Validation
the item-total correlation.
Psychometric Properties Analysis
Implications for Practice
allows these researchers to focus on the knowledge and skills about qualitative and quantitative methodological approaches that they need to acquire and develop to be able to conduct a validation study. Finally, it allows novice researchers to develop team leadership skills in the research process. This includes not only coordinating the activities of the team of translators and experts that comprise the research team but also foreseeing the research outputs can be generated with the process of translation, adaptation, and cross-cultural validation. Examples of such outputs include a literature review article on the construct of the instrument, a concept analysis article, a methodological article reporting the process of translation and crosscultural adaptation, and another detailing the psychometric properties’ quality of the instrument.
Conclusion
Acknowledgments
Disclosure
References
- May C, Finch T, Rapley T. Normalization process theory. In: Nielsen P, Birken SA, editors. Handbook of Implementation Science. Edward Elgar Publishing; 2020:157.
- Portney LG. Designing surveys and questionnaires. In: Foundations of Clinical Research: Applications to Evidence-Based Practice. 4th ed. F.A. Davis Company; 2020:141-150.
- Chow S-C, Liu J-P. Basic statistic concepts. In: Design and Analysis of Clinical Trials. 2nd ed. Wiley-Interscience; 2004:47-54.
- Sidani S, Braden CJ. Examination of interventions’ acceptance. In: Nursing and Health Interventions: Design, Evaluation, and Implementation. 2nd ed. Wiley-Blackwell; 2021:218-225.
- Efstathiou G. Translation, adaptation and validation process of research intruments. In: Suohnen R, Stolt M, Papatravou E, editors. Individualized Care: Theory, Measurement, Research and Practice. Springer; 2019.
- Cha E-S, Kim KH, Erlen JA. Translation of scales in cross-cultural research: issues and techniques. J Adv Nurs. 2007;58(4):386-395. doi:10.1111/j.1365-2648.2007.04242.x
- Sutherland S. Outcomes research. In: Gray JR, Grove SK, editors. The Practice of Nursing Research: Appraisal, Synthesis, and Generation of Evidence. 9th ed. Elsevier; 2020:365.
- Miller LA, Lovler RL. How do we assess the psychometric quality of a test? In: Foundation of Psychological Testing: A Practical Approach. 6th ed. Sage Publications; 2020:326.
- Kristjansson EA, Desrochers A, Zumbo B. Translating and adapting measurement instruments for cross-linguistic and cross-cultural research: a guide for practitioners. Can J Nurs Res. 2003;35(2):127-142.
- Alagappan T. The cross-cultural adaptation process of a patient-reported outcome measure. J Sci Soc. 2023;50(1):13. doi:10.4103/jss. jss_136_21
- Sul SIR, Lucas PRMB. Translation and validation of the anticipated turnover scale for the Portuguese cultural context. Nurs Open. 2020;7 (5):1475-1481. doi:10.1002/nop2.521
- Almeida S, Nascimento A, Lucas PB, Jesus É, Araújo B. RN4CAST study in Portugal: validation of the Portuguese version of the Practice Environment Scale of the Nursing Work Index. Aquichan. 2020;20(3):1-10. doi:10.5294/aqui.2020.20.3.8
- Lucas P, Jesus E, Almeida S, Araújo B. Validation of the psychometric properties of the Practice Environment Scale of Nursing Work Index in primary health care in Portugal. Int J Environ Res Public Health. 2021;18(12):6422. doi:10.3390/ijerph18126422
- Anunciada S, Benito P, Gaspar F, Lucas P. Validation of psychometric properties of the Nursing Work Index: revised scale in Portugal. Int J Environ Res Public Health. 2022;19(9):4933. doi:10.3390/ijerph 19094933
- Carvalho M, Gaspar F, Potra T, Lucas P. Translation, adaptation, and validation of the Self-efficacy scale for clinical nurse leaders for the Portuguese culture. Int J Environ Res Public Health. 2022;19(14):8590. doi:10.3390/ijerph19148590
- Sousa E, Lin C-F, Gaspar F, Lucas P. Translation and validation of the indicators of quality nursing work environments in the Portuguese cultural context. Int J Environ Res Public Health. 2022;19(19):12313. doi:10.3390/ijerph191912313
- Cabrita C, Lucas P, Teixeira G, Gaspar F. Translation and validation of the Individual Workload Perception Scale: revised for Portuguese nurses. Healthcare. 2022;10(12):2476. doi:10.3390/healthcare10122476
- Gomes P, Ribeiro S, Silva M, et al. Cross-cultural validation of the Portuguese version of the Quality of Oncology Nursing Care Scale. Cancers (Basel). 2024;16(5):859. doi:10.3390/cancers16050859
- Cunha F, Pinto MR, Riesch S, Lucas P, Almeida S, Vieira M. Translation, adaptation, and validation of the Portuguese version of the Exercise of Self-Care Agency Scale. Healthcare. 2024;12(2):159. doi:10.3390/healthcare12020159
- Waltz CF, Strickland OL, Lenz ER. Other mesurement issues. In: Measurement in Nursing and Health Research. 4th ed. Springer Publishing; 2010:446-448.
- Beaton D, Bombardier C, Guillemin F, Ferraz MB. Guidelines for the process of cross-cultural adaptation of self-report measures. Spine (Phila Pa 1976). 2000;25(24):3186-3191. doi:10.1097/00007632-200012150-00014
- Hedrih V. Test translation. In: Adapting Psychological Tests and Measurement Instruments for Cross-Cultural Research: An Introduction. Routledge; 2020:48-98.
- Nord C. Defining translation functions: the translation brief as a guideline for the trainee translator. Ilha Do Desterro. 1997;33:41-55.
- Montalt V, González-Davies M. Understanding medical communication. In: Medical Translation Step by Step: Learning by Drafting. Routledge; 2006:47.
- Epstein J, Santo RM, Guillemin F. A review of guidelines for cross-cultural adaptation of questionnaires could not bring out a consensus.
Clin Epidemiol. 2015;68(4):435-441. doi:10.1016/j.jclinepi.2014.11.021 - Herdman M, Fox-Rushby J, Badia X. A model of equivalence in the cultural adaptation of HRQoL instruments: the universalist approach. Qual Life Res. 1998;7(4):323-335. doi:10.1023/a:1024985930536
- Peña ED. Lost in translation: methodological considerations in cross-cultural research. Child Dev. 2007;78(4):1255-1264. doi:10.1111/j.14678624.2007.01064.x
- Alonso J, Black C, Norregaard J-C, et al. Cross-cultural differences in the reporting of global functional capacity. Med Care. 1998;36 (6):868-878. doi:10.1097/00005650-199806000-00010
- Muniz J, Hambleton RK, Xing D. Small samples studies to detect flaws in item translations. Int J Test. 2001;1(2):115-135. doi:10.1207/ S15327574IJT0102_2
- Waltz CF, Strickland OL, Lenz ER. Other measurement issues. In: Measurement in Nursing and Health Research. 4th ed. Springer Publishing; 2010:449-452.
- Riccio CA, Yoon H, McCormick AS. Neuropsychological test selection with clients who are Asian. In: Davis JM, D’Amato RC, editors. Neuropsychology of Asians and Asian-Americans: Practical and Theoretical Considerations. Spinger Publishing; 2014:153.
- Smith TW, et al. Developing and evaluating cross national survey instruments. In: Presser S, Rothgeb JM, Couper MP, editors. Methods for Testing and Evaluating Survey Questionnaires. Wiley Interscience; 2004. 439-442.
- van de Vijver F, Portinga YH. Conceptual and methodological issues in adapting tests. In: Hembletom RK, Merenda PF, Spilberg CD, editors. Adapting Educational and Psychological Tests for Cross-Cultural Assessment. Lawrence Erlbaum Associates; 2005:41-47.
- Austin PC, Brunner LJ. Type I error inflation in the presence of a ceiling effect. Am Stat. 2003;57(2):97-104. doi:10.1198/0003130031450
- Holbrook A. Acquiescence response bias. In: Lavrakas PJ, editor. Encyclopedia of Survey Research Methods. Vol. I. Sage; 2008:3.
- van de Vijver FJR, Poortinga YH. Towards an Integrated Analysis of Bias in Cross-Cultural Assessment. Eur J Psychol Assess. 1997; 13 (1):29-37. doi:10.1027/1015-5759.13.1.29
- Bartram D. Increasing validity with forced-choice criterion measurement formats. Int J Sel Assess. 2007;15(3):263-272. doi:10.1111/j.14682389.2007.00386.x
- Wivagg J. Forced choice. In: Lavrakas PJ, editor. Encyclopedia of Survey Research Methods. Sage; 2008:289-290.
- Maitland A. Attitudes measurement. In: Lavrakas PJ, editor. Encyclopedia of Survey Research Methods. Sage; 2008:37-38.
- Hilton A, Skrutkowski M. Translating instruments into other languages: development and testing processes. Cancer Nurs. 2002;25(1):1-7. doi:10.1097/00002820-200202000-00001
- Lino de CR, Brüggemann M, Souza M de L de OM, Barbosa de S, Santos Dos EKA. Adaptação transcultural de instrumentos de pesquisa conduzida pela enfermagem do Brasil: uma revisão integrativa. Texto Context – Enferm. 2018;26(4). doi:10.1590/0104-07072017001730017
- Guillemin F, Bombardier C, Beaton D. Cross-cultural adaptation of health-related quality of life measures: literature review and proposed guidelines. J Clin Epidemiol. 1993;46(12):1417-1432. doi:10.1016/0895-4356(93)90142-N
- Machado da R, Fernandes AD de BF S, Oliveira de ALCB, Soares LS, Gouveia de O MT, Silva da GRF. Métodos de adaptação transcultural de instrumentos na área da enfermagem. Rev Gaúcha Enferm. 2018;39. doi:10.1590/1983-1447.2018.2017-0164
- Acquadro C, Conway K, Hareendran A, Aaronson N. Literature review of methods to translate Health-Related Quality of Life Questionnaires for use in multinational clinical trials. Value Heal. 2008;11(3):509-521. doi:10.1111/j.1524-4733.2007.00292.x
- Farina N, Jacobs R, Sani TP, et al. Description of the cross-cultural process adopted in the STRiDE (STrengthening Responses to dementia in DEveloping countries) program: a methodological overview. Alzheimer’s Dement Diagnosis, Assess Dis Monit. 2022;14(1). doi:10.1002/ dad2. 12293
- Furukawa R, Driessnack M, Colclough Y. A committee approach maintaining cultural originality in translation. Appl Nurs Res.
(2):144-146. doi:10.1016/j.apnr.2013.11.011 - Helmich E, Cristancho S, Diachun L, Lingard L. ‘How would you call this in English?’: being reflective about translations in international, cross-cultural qualitative research. Perspect Med Educ. 2017;6(2):127-132. doi:10.1007/S40037-017-0329-1
- Uysal-Bozkir Ö, Parlevliet JL, de Rooij SE. Insufficient cross-cultural adaptations and psychometric properties for many translated health assessment scales: a systematic review. J Clin Epidemiol. 2013;66(6):608-618. doi:10.1016/j.jclinepi.2012.12.004
- Albach CA, Wagland R, Hunt KJ. Cross-cultural adaptation and measurement properties of generic and cancer-related patient-reported outcome measures (PROMs) for use with cancer patients in Brazil: a systematic review. Qual Life Res. 2018;27(4):857-870. doi:10.1007/s11136-017-1703-5
- Praveen S, Parmar J, Chandio N, Arora A. A systematic review of cross-cultural adaptation and psychometric properties of oral health literacy tools. Int J Environ Res Public Health. 2021;18(19):10422. doi:10.3390/ijerph181910422
- Min SN, Duangthip D, Gao SS, Detsomboonrat P. Quality of the adaptation procedures and psychometric properties of the scale of oral health outcomes for 5-year-old children (SOHO-5): a systematic review. Qual Life Res. 2023;32(6):1537-1547. doi:10.1007/s11136-022-03280-2
- Danielsen AK, Pommergaard H-C, Burcharth J, Angenete E, Rosenberg J. Translation of questionnaires measuring health related quality of life is not standardized: a literature based research study. PLoS One. 2015;10(5):e0127050. doi:10.1371/journal.pone. 0127050
- Øygarden A-MU, Berg RC, Abudayya A, Glavin K, Strøm BS. Measurement instruments for parental stress in the postpartum period: a scoping review. PLoS One. 2022;17(3):e0265616. doi:10.1371/journal.pone. 0265616
- Echevarría-Guanilo ME, Gonçalves N, Romanoski PJ. Psychometric properties of measurement instruments: conceptual basis and evaluation methods: part II. Texto Context – Enferm. 2019;28(e20170311):1-14. doi:10.1590/1980-265x-tce-2017-0311
- Cruchinho P, Teixeira G, Lucas P, Gaspar F. Evaluating the methodological approaches of cross-cultural adaptation of the Bedside Handover Attitudes and Behaviours Questionnaire into Portuguese. J Healthc Leadersh. 2023;15:193-208. doi:10.2147/JHL.S422122
- Slade D, Murray KA, Pun JKH, Eggins S. Nurses’ perceptions of mandatory bedside clinical handovers: an Australian hospital study. J Nurs Manag. 2019;27(1):161-171. doi:10.1111/jonm. 12661
- Freudental-Pederson M, Hartmann-Peterson K, Nielsen LD. Mixing methods in the search for mobile complexity. In: Ficham B, Mark M, Murray L, editors. Mobile Methodologies. Palgrave McMillan; 2010:30.
- Peters M, Passchier J. Translating instruments for cross-cultural studies in headache research. Headache J Head Face Pain. 2006;46(1):82-91. doi:10.1111/j.1526-4610.2006.00298.x
- Weeks A, Swerissen H, Belfrage J. Issues, challenges, and solutions in translating study instruments. Eval Rev. 2007;31(2):153-165. doi:10.1177/0193841X06294184
- Wild D, Grove A, Martin M, et al. Principles of good practice for the translation and cultural adaptation process for patient-reported outcomes (PRO) measures: report of the ISPOR task force for translation and cultural adaptation. Value Heal. 2005;8(2):94-104. doi:10.1111/j.15244733.2005.04054.x
- Reichenheim ME, Moraes CL. Operacionalização de adaptação transcultural de instrumentos de aferição usados em epidemiologia. Rev Saude Publica. 2007;41(4):665-673. doi:10.1590/S0034-89102006005000035
- Sousa V, Rojjanasrirat W. Translation, adaptation and validation of instruments or scales for use in cross-cultural health care research: a clear and user-friendly guideline. J Eval Clin Pract. 2011;17(2):268-274. doi:10.1111/j.1365-2753.2010.01434.x
- Hagell P, Hedin P-J, Meads DM, Nyberg L, McKenna SP. Effects of Method of Translation of Patient-Reported Health Outcome Questionnaires: a Randomized Study of the Translation of the Rheumatoid Arthritis Quality of Life (RAQoL) Instrument for Sweden. Value Heal. 2010;13(4):424-430. doi:10.1111/j.1524-4733.2009.00677.x
- Lee WL, Chinna K, Lim Abdullah K, Zainal Abidin I. The forward-backward and dual-panel translation methods are comparable in producing semantic equivalent versions of a heart quality of life questionnaire. Int J Nurs Pract. 2019;25(1). doi:10.1111/ijn.12715
- Papadakis NM, Aletta F, Kang J, Oberman T, Mitchell A, Stavroulakis GE. Translation and cross-cultural adaptation methodology for soundscape attributes – a study with independent translation groups from English to Greek. Appl Acoust. 2022;200:109031. doi:10.1016/j. apacoust.2022.109031
- van de Vijver FJR, Poortinga YH. On the study of culture in developmental science. Hum Dev. 2002;45(4):246-256. doi:10.1159/000064985
- Petkovic J, Epstein J, Buchbinder R, et al. Toward ensuring health equity: readability and cultural equivalence of OMERACT patient-reported outcome measures. J Rheumatol. 2015;42(12):2448-2459. doi:10.3899/jrheum. 141168
- Bundgaard K, Nisbeth Brøgger M. “Don’t fix bad translations”: a netnographic study of translators’ understandings of back translation in the medical domain. MonTi Monogr Traducción e Interpret. 2018;1(10):205-224. doi:10.6035/MonTI.2018.10.8
- Hawkins M, Cheng C, Elsworth GR, Osborne RH. Translation method is validity evidence for construct equivalence: analysis of secondary data routinely collected during translations of the Health Literacy Questionnaire (HLQ). BMC Med Res Methodol. 2020;20(1):130. doi:10.1186/ s12874-020-00962-8
- Epstein J, Osborne RH, Elsworth GR, Beaton DE, Guillemin F. Cross-cultural adaptation of the Health Education Impact Questionnaire: experimental study showed expert committee, not back-translation, added value. J Clin Epidemiol. 2015;68(4):360-369. doi:10.1016/j. jclinepi.2013.07.013
- Teig CJP, Bond MJ, Grotle M, et al. A novel method for the translation and cross-cultural adaptation of health-related quality of life patient-reported outcome measurements. Health Qual Life Outcomes. 2023;21(1):13. doi:10.1186/s12955-023-02089-y
- Tsai T-I, Luck L, Jefferies D, Wilkes L. Challenges in adapting a survey: ensuring cross-cultural equivalence. Nurse Res. 2018;26(1):28-32. doi:10.7748/nr.2018.e1581
- Jayawickreme E, Jayawickreme N, Goonasekera MA. Using focus group methodology to adapt measurement scales and explore questions of wellbeing and mental health. Intervention. 2012;10(2):156-167. doi:10.1097/WTF.0b013e328356f3c4
- Montenegro M, Valdez D, Crawford B, Turner R, Lo W-J, Jozkowski KN. Using a decentering framework to create English/Spanish surveys about abortion: insights into comparative survey research (CSR) for new survey development and recommendations for optimal use. Soc Sci J. 2022;1-15. doi:10.1080/03623319.2022.2092379
- Vizcaya-Moreno MF, Pérez-Cañaveras RM. Country validation of the CLES-Scale: linguistic and cultural perspectives. In: Saarikoski M, Strandell-Laine C, editors. The CLES-Scale: An Evaluation Tool for Healthcare Education. Springer; 2018:31-46.
- Erkut S. Developing multiple language versions of instruments for intercultural research. Child Dev Perspect. 2010;4(1):19-24. doi:10.1111/ j.1750-8606.2009.00111.x
- Marcondes FB, de Vasconcelos RA, Marchetto A, de Andrade ALL, Filho AZ, Etchebehere M. Translation and cross-cultural adaptation of the Rowe score for Portuguese. Acta Ortop Bras. 2012;20(6):346-350. doi:10.1590/S1413-78522012000600007
- Nepal GM, Shrestha A, Acharya R. Translation and cross-cultural adaptation of the Nepali version of the Rowland Universal Dementia Assessment Scale (RUDAS). J Patient-Reported Outcomes. 2019;3(1). doi:10.1186/s41687-019-0132-3
- Coll-Risco I, Camiletti-Moirón D, Acosta-Manzano P, Aparicio VA. Translation and cross-cultural adaptation of the Pregnancy Physical Activity Questionnaire (PPAQ) into Spanish. J Matern Neonatal Med. 2019;32(23):3954-3961. doi:10.1080/14767058.2018.1479849
- Pereira GIDN, Costa CDDS, Geocze L, Borim AA, Ciconelli RM, Camacho-Lobato L. Cross-cultural adaptation and validation for Portuguese (Brazil) of health related quality of life instruments specific for gastroesophageal reflux disease. Arq Gastroenterol. 2007;44(2):168-177. doi:10.1590/s0004-28032007000200016
- Grundström H, Rauden A, Olovsson M. Cross-cultural adaptation of the Swedish version of Endometriosis Health Profile-30. J Obstet Gynaecol (Lahore). 2020;40(7):969-973. doi:10.1080/01443615.2019.1676215
- Polesello GC, Godoy GF, De Castro Trindade CA, De Queiroz MC, Honda E, Ono NK. Translation and cross-cultural adaptation of the modified Hip outcome tool (mhot) into Portuguese. Acta Ortop Bras. 2012;20(2):88-92. doi:10.1590/S1413-78522012000200006
- Muquith MA, Islam MN, Haq SA, Ten Klooster PM, Rasker JJ, Yunus MB. Cross-cultural adaptation and validation of a Bengali version of the modified fibromyalgia impact questionnaire. BMC Musculoskelet Disord. 2012;13(1):1. doi:10.1186/1471-2474-13-157
- Toma G, Guetterman TC, Yaqub T, Talaat N, Fetters MD. A systematic approach for accurate translation of instruments: experience with translating the Connor-Davidson Resilience Scale into Arabic. Methodol Innov. 2017;10(3):205979911774140. doi:10.1177/2059799117741406
- Poot CC, Meijer E, Fokkema M, Chavannes NH, Osborne RH, Kayser L. Translation, cultural adaptation and validity assessment of the Dutch version of the eHealth Literacy Questionnaire: a mixed-method approach. BMC Public Health. 2023;23(1):1006. doi:10.1186/s12889-023-15869-4
- Hasani L, Santoso H, Junus K. Instrument development for investigating students’ intention to participate in online discussion forums: crosscultural and context adaptation sing SEM. J Educ Online. 2021;18(3). doi:10.9743/JEO.2021.18.3.9
- Bundgaard K, Brøgger MN. Who is the back translator? An integrative literature review of back translator descriptions in cross-cultural adaptation of research instruments. Perspectives (Montclair). 2019;27(6):833-845. doi:10.1080/0907676X.2018.1544649
- Geisinger KF. Cross-cultural normative assessment: translation and adaptation issues influencing the normative interpretation of assessment instruments. Psychol Assess. 1994;6(4):304-312. doi:10.1037/1040-3590.6.4.304
- van Widenfelt BM, Treffers PDA, de Beurs E, Siebelink BM, Koudijs E. Translation and cross-cultural adaptation of assessment instruments used in psychological research with children and families. Clin Child Fam Psychol Rev. 2005;8(2):135-147. doi:10.1007/s10567-005-4752-1
- Son J. Back translation as a documentation tool. Int J Transl Interpret Res. 2018;10(2):89-100. doi:10.12807/ti.110202.2018.a07
- Coster WJ, Mancini MC. Recommendations for translation and cross-cultural adaptation of instruments for occupational therapy research and practice. Rev Ter Ocup da Univ São Paulo. 2015;26(1):50. doi:10.11606/issn.2238-6149.v26i1p50-57
- Ortiz-Gutiérrez S, Cruz-Avelar A. Translation and cross-cultural adaptation of health assessment tools. Actas Dermo-Sifiliográficas (English Ed. 2018;109(3):202-206. doi:10.1016/j.adengl.2018.02.003
- Ramada-Rodilla JM, Serra-Pujadas C, Delclós-Clanchet GL. Adaptación cultural y validación de cuestionarios de salud: revisión y recomendaciones metodológicas. Salud Publica Mex. 2013;55(1):57-66. doi:10.1590/S0036-36342013000100009
- Prakash V, Shah S, Hariohm K. Cross-cultural adaptation of patient-reported outcome measures: a solution or a problem? Ann Phys Rehabil Med. 2019;62(3):174-177. doi:10.1016/j.rehab.2019.01.006
- Fortes CPDD, Araújo de QC AP. Check list para tradução e adaptação transcultural de questionários em saúde. Cad Saúde Coletiva. 2019;27 (2):202-209. doi:10.1590/1414-462
- Hernández A, Hidalgo MD, Hambleton RK, Gómez-Benito J. International test commission guidelines for test adaptation: a criterion checklist. Psicothema. 2020;32(3):390-398. doi:10.7334/psicothema2019.306
- DuBay M, Watson LR. Translation and cultural adaptation of parent-report developmental assessments: improving rigor in methodology. Res Autism Spectr Disord. 2019;62:55-65. doi:10.1016/j.rasd.2019.02.005
- Hall DA, Zaragoza Domingo S, Hamdache LZ, et al. A good practice guide for translating and adapting hearing-related questionnaires for different languages and cultures. Int J Audiol. 2018;57(3):161-175. doi:10.1080/14992027.2017.1393565
- José M, Elosua P, Hambleton RK. Directrices para la traducción y adaptación de los tests: segunda edición. Psicothema. 2013;25(2):151-157. doi:10.7334/psicothema2013.24
- Santo RM, Ribeiro-Ferreira F, Alves MR, Epstein J, Novaes P. Enhancing the cross-cultural adaptation and validation process: linguistic and psychometric testing of the Brazilian-Portuguese version of a self-report measure for dry eye. J Clin Epidemiol. 2015;68(4):370-378. doi:10.1016/j.jclinepi.2014.07.009
- Arafat S, Chowdhury H, Qusar M, Hafez M. Cross cultural adaptation and psychometric validation of research instruments: a methodological review. J Behav Heal. 2016;5(3):129. doi:10.5455/jbh.20160615121755
- Kyriazos TA. Applied psychometrics: the 3-Faced Construct Validation Method, a routine for evaluating a factor structure. Psychology. 2018;09 (08):2044-2072. doi:10.4236/psych.2018.98117
- Plonsky L, Kim Y. Task-based learner production: a substantive and methodological review. Annu Rev Appl Linguist. 2016;36:73-97. doi:10.1017/S0267190516000015
- Peters MD, Godfrey C, McInerney P, Soares CB, Khalil H, Parker D. Development of a scoping review protocol. In: Aromataris E, Munn Z, editors. Joanna Briggs Institute Reviewer’s Manual. : The Joanna Briggs Institute; 2020
- Shekelle PG, Woolf SH, Eccles M, Grimshaw J. Developing clinical guidelines. West J Med. 1999;170(6):348-351.
- OECD Eurostat and World Health Organization. Classification of Health Care Providers (ICHA-HP). In: A System of Health Accounts 2011. OECD Publishing; 2017: 121-152. doi:10.1787/9789264270985-en
- Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021: n71. doi:10.1136/bmj.n71
- Schreier M. Qualitative content analysis. In: Flick U, editor. The SAGE Handbook of Qualitative Data Analysis. Sage Publications; 2014:170-183.
- World Health Organization. Introduction. In: WHO Handbook for Guideline Developement. 2nd ed. World Health Organization; 2014:2
- Pernambuco L, Espelt A, Magalhães Junior HV, Lima de KC. Recomendações para elaboração, tradução, adaptação transcultural e processo de validação de testes em fonoaudiologia. CoDAS. 2017;29(3). doi:10.1590/2317-1782/20172016217
- International Test Comission. ITC Guidelines for Translating and Adapting Tests. International Test Comission; 2017.
- Tafforeau J, Cobo ML, Tolonen H, Christa S-N, Tinto A. Guidelines for the Development and Criteria for the Adoption of Health Survey Instruments; 2015. Available from: https://ec.europa.eu/health/ph_information/dissemination/reporting/healthsurveys_en.pdf. Accessed May 16, 2024.
- Høegh MC, Høegh S-M. Trans-adapting outcome measures in rehabilitation: cross-cultural issues. Neuropsychol Rehabil. 2009;19(6):955-970. doi:10.1080/09602010902995986
- McKenna SP. Measuring patient-reported outcomes: moving beyond misplaced common sense to hard science. BMC Med. 2011;9(1):86. doi:10.1186/1741-7015-9-86
- Prinsen CAC, Mokkink LB, Bouter LM, et al. COSMIN guideline for systematic reviews of patient-reported outcome measures. Qual Life Res. 2018;27(5):1147-1157. doi:10.1007/s11136-018-1798-3
- Mokkink LB, Prinsen CAC, Patrick DL, et al. COSMIN Study Design Checklist for Patient-Reported Outcome Measurement Instruments; 2019. Available from: https://www.cosmin.nl/wp-content/uploads/COSMIN-study-designing-checklist_final.pdf. Accessed May 16, 2024.
- Gjersing L, Caplehorn JR, Clausen T. Cross-cultural adaptation of research instruments: language, setting, time and statistical considerations. BMC Med Res Methodol. 2010;10(1):13. doi:10.1186/1471-2288-10-13
- Chávez LM, Canino GC Toolkit on Translating and Adapting Instruments; 2005. Available from: https://www.hsri.org/files/uploads/publica tions/PN54_Translating_and_Adapting.pdf. Accessed May 16, 2024.
- World Health Organization. WHO guidelines on translation: process of translation and adaptation of instruments; 2019. Available from: http:// www.who.int/substance_abuse/research_tools/translation/en/. Accessed May 16, 2024.
- Wild D, Eremenco S, Mear I, et al. Multinational trials-Recommendations on the translations required, approaches to using the same language in different countries, and the approaches to support pooling the Data: the ISPOR patient-reported outcomes translation and linguistic validation good research practices task force Report. Value Heal. 2009;12(4):430-440. doi:10.1111/j.1524-4733.2008.00471.x
- Swami V, Barron D. Translation and validation of body image instruments: challenges, good practice guidelines, and reporting recommendations for test adaptation. Body Image. 2019;31:204-220. doi:10.1016/j.bodyim.2018.08.014
- Kuliś D, Bottomley A, Velikova G, Greimel E, Koller M. EORTC Quality of Life Group Translation Procedure; 2017. Available from: https:// www.eortc.org/app/uploads/sites/2/2018/02/translation_manual_2017.pdf. Accessed May 16, 2024.
- Ohrbach R, Ohrbach J, Jezewski M, John MT, Lobbezoo F. Guidelines for Establishing Cultural Equivalency of Instruments; 2013. Available from: https://ubwp.buffalo.edu/rdc-tmdinternational/wp-content/uploads/sites/58/2017/01/Guidelines-for-Translation-and-Cultural-Equivalency-of-Instruments-2013_05_118608.pdf. Accessed May 16, 2024.
- Baker DL, Melnikow J, Ying Ly M, Shoultz J, Niederhauser V, Diaz-Escamilla R. Translation of health surveys using mixed methods. J Nurs Scholarsh. 2010;42(4):430-438. doi:10.1111/j.1547-5069.2010.01368.x
- Biering-Sørensen F, Alexander MS, Burns S, et al. Recommendations for translation and reliability testing of international spinal cord injury data sets. Spinal Cord. 2011;49(3):357-360. doi:10.1038/sc.2010.153
- Koller M, Aaronson NK, Blazeby J, et al. Translation procedures for standardised quality of life questionnaires: the European Organisation for Research and Treatment of Cancer (EORTC) approach. Eur J Cancer. 2007;43(12):1810-1820. doi:10.1016/j.ejca.2007.05.029
- Eremenco SL, Cella D, Arnold BJ. A comprehensive method for the translation and cross-cultural validation of health status questionnaires. Eval Health Prof. 2005;28(2):212-232. doi:10.1177/0163278705275342
- Dhamani KA, Richter MS. Translation of research instruments: research processes, pitfalls and challenges. Afr J Nurs Midwifery. 2011;13 (1):3-13.
- Sperber AD. Translation and validation of study instruments for cross-cultural research. Gastroenterology. 2004;126:S124-S128. doi:10.1053/j. gastro.2003.10.016
- Aaronson N, Alonso J, Burnam A, et al. Assessing health status and quality-of-life instruments: attributes and review criteria. Qual Life Res. 2002;11(3):193-205. doi:10.1023/a:1015291021312
- Organisation for Economic Co-operation and Development. Translation and adaptation guidelines for PISA 2012; 2012. Available from: https:// www.oecd.org/pisa/pisaproducts/49273486.pdf. Accessed May 16, 2024.
- Swami V, Todd J, Barron D. Translation and validation of body image instruments: an addendum to Swami and Barron (2019) in the form of frequently asked questions. Body Image. 2021;37:214-224. doi:10.1016/j.bodyim.2021.03.002
- Terwee CB, Prinsen CAC, Chiarotto A, et al. COSMIN methodology for evaluating the content validity of patient-reported outcome measures: a Delphi study. Qual Life Res. 2018;27(5):1159-1170. doi:10.1007/s11136-018-1829-0
- Yasir ASM. Cross Cultural Adaptation & Psychometric Validation of Instruments: step-wise Description. Int J Psychiatry. 2016;1(1). doi:10.33140/IJP/01/01/00001
- Regnault A, Herdman M. Using quantitative methods within the Universalist model framework to explore the cross-cultural equivalence of patient-reported outcome instruments. Qual Life Res. 2015;24(1):115-124. doi:10.1007/s11136-014-0722-8
- Mokkink LB, Terwee CB, Knol DL, et al. The COSMIN checklist for evaluating the methodological quality of studies on measurement properties: a clarification of its content. BMC Med Res Methodol. 2010;10(1):22. doi:10.1186/1471-2288-10-22
- Mokkink LB, Terwee CB, Patrick DL, et al. The COSMIN checklist for assessing the methodological quality of studies on measurement properties of health status measurement instruments: an international Delphi study. Qual Life Res. 2010;19(4):539-549. doi:10.1007/s11136-010-9606-8
- Terwee CB, Bot SDM, de Boer MR, et al. Quality criteria were proposed for measurement properties of health status questionnaires.
Clin Epidemiol. 2007;60(1):34-42. doi:10.1016/j.jclinepi.2006.03.012 - Möhler R, Bartoszek G, Köpke S, Meyer G. Proposed criteria for reporting the development and evaluation of complex interventions in healthcare (CReDECI): guideline development. Int
Nurs Stud. 2012;49(1):40-46. doi:10.1016/j.ijnurstu.2011.08.003 - American Educational Research Association. Standards for Educational and Psychological Testing. American Educational Research Association; 2014.
- Gudmundsson E. Guidelines for translating and adapting psychological instruments. Nord Psychol. 2009;61(2):29-45. doi:10.1027/19012276.61.2.29
- Forsyth BH, Kudela MS, Levin K, Lawrence D, Willis GB. Methods for translating an English-language survey questionnaire on tobacco use into mandarin, Cantonese, Korean, and Vietnamese. Field Methods. 2007;19(3):264-283. doi:10.1177/1525822X07302105
- American Psychological Association. Criteria for practice guideline development and evaluation. Am Psychol. 2002;57(12):1048-1051. doi:10.1037/0003-066X.57.12.1048
- Iliescu D. Pre-condition guidelines. In: Adapting Tests in Linguist and Cultural Adaptations. Cambridge University Press; 2017:87.
- Iliescu D. Documentation guidelines. In: Adapting Tests in Linguist and Cultural Adaptations. Cambridge University Press; 2017:105.
- Hambletom RK. Issues, designs and tecnical guidelines for adaptating tests into multiples languages and cultures. In: Hembletom RK, Merenda PF, Spilberg CD, editors. Adapting Educational and Psychological Tests for Cross-Cultural Assessment. Lawrence Erlbaum Associates; 2005:3-38.
- Oakland T. Selected ethical issues relevant to test adaptations. In: Hambletom RK, Merenda PF, Spielberg CD, editors. Adapting Educational and Psychological Tests for Cross-Cultural Assessment. Lawrence Erlbaum Associates; 2005:80.
- Brislin RW, Lonner WJ, Thorndike RM. Cross cultural research methods. In: Transcultural Psychiatric Research Review. Vol. 12. John Wiley ans Sons;1973:7-10. doi:10.1177/136346157501200101
- Beaton D, Bombardier C, Guillemin F, Ferraz MB. Recommendations for the cross-cultural adaptation of the DASH & QuickDASH outcome measure. Inst Work Heal. 2007;45.
- Ozolins U, Hale S, Cheng X, Hyatt A, Schofield P. Translation and back-translation methodology in health research: a critique. Expert Rev Pharmacoecon Outcomes Res. 2020;20(1):69-77. doi:10.1080/14737167.2020.1734453
- Bornman J, Sevcik RA, Romski M, Pae HK. Successfully translating language and culture when adapting assessment measures. J Policy Pract Intellect Disabil. 2010;7(2):111-118. doi:10.1111/j.1741-1130.2010.00254.x
- Simonsen E, Mortensen EL. Difficulties in translation of personality scales. J Pers Disord. 1990;4(3):290-296. doi:10.1521/pedi.1990.4.3.290
- Tanzer NK. Developing Tests for Use in Multiple Languages and Cultures: A Plea for Simultaneous Development. Lawrence Erlbaum Associates; 2005.
- Bennett PM. Reviewing translated scales: backtranslation under the spotlight. Transl Matters. 2022;4(125-144).
- van de Vijver F, Tanzer NK. Bias and equivalence in cross-cultural assessment: an overview. Eur Rev Appl Psychol. 2004;54(2):119-135. doi:10.1016/j.erap.2003.12.004
- de Klerk S, Jerosch-Herold C, Buchanan H, van Niekerk L. Shared decision making and the practice of community translation in presenting a pre-final Afrikaans for the Western Cape Disabilities of the Arm, Shoulder and Hand (DASH) questionnaire: a proposal for improved translation and cross-cultural adaptation. J Patient-Reported Outcomes. 2019;3(1):52. doi:10.1186/s41687-019-0144-z
- Erkut S. Developing multiple language versons of instruments for intercultural research. In: Ji M, editor. Cross-Cultural Health Translation: Exploring Methodological and Digital Tools. Routledge; 2019:9-10.
- Silverblatt A, Zlobin N. Production elements. In: International Communications: A Media Literacy Approach. Routledge; 2015:108.
- St Amant K. A new web for the new millennium. In: Lipson C, Day M, editors. Technical Communication and the World Wide Web. Lawrence Erlbaum Associates Publishers; 2005:148.
- Drasgow F, Probst TM. The psicometrics of adaptation: evaluating measurement equivalance across languages and cultures. In: Hambletom RK, Merenda PF, Spielberg CD, editors. Adapting Educational and Psychological Tests for Cross-Cultural Assessment. Lawrence Erlbaum Associates Publishers; 2005:361.
- Erkut S, Alarcón O, Coll CG, Tropp LR, García HAV. The dual-focus approach to creating bilingual measures. J Cross Cult Psychol. 1999;30 (2):206-218. doi:10.1177/0022022199030002004
- van der Vidjer F, Leung K. Equivalence and bias: a review of concepts, models, and data analytic procedures. In: Matsumoto D, van de Vijver F, editors. Cross-Cultural Research Methods in Psychology. Cambridge University Press; 2011:17-44.
- American Educational Research Association. Test design and development. Standards for Educational and Psychological Testing; American Educational Research Association; 2014. 81-84.
- van Teijlingen ER, Hundley V. Pilot study. In: Lewis-Back MS, Bryman A, Liao TF, editors. The SAGE Encyclopedia of Social Science Research Methods. Vol II. Sage; 2004:823-824.
- Gallagher PM. Pretest. In: Lewis-Back MS, Bryman A, Liao TF, editors. The SAGE Encyclopedia of Social Science Research Methods. Vol II. Sage Publications; 2004:853-854.
- Korabik K, van Rhijn T. Best practices in scale translation and establisment mesurement equivalence. In: Shockey KM, Shen W, Johnson RC, editors. The Cambridge Handbook of the Global Work-Family Interface. Cambridge University Press; 2018:212-229.
- Willis G. Pretesting of health survey questionnaires: cognitive interviewing, usability testing, and behavior coding. In: Johnson TP, editor. Handbook on Health Survey Methods. Willey; 2015:221.
- Rodrigues IB, Adachi JD, Beattie KA, MacDermid JC. Development and validation of a new tool to measure the facilitators, barriers and preferences to exercise in people with osteoporosis. BMC Musculoskelet Disord. 2017;18(1):540. doi:10.1186/s12891-017-1914-5
- Perneger TV, Courvoisier DS, Hudelson PM, Gayet-Ageron A. Sample size for pre-tests of questionnaires. Qual Life Res. 2015;24(1):147-151. doi:10.1007/s11136-014-0752-2
- Holyk GG. Question testing methods. In: Encyclopedia of Survey Research Methods. Vol II. Sage; 2008:658-659.
- van der Zouwen J, Smit JH, et al. Evaluating survey questions by analyzing patterns of behavior codes and question-answer sequences: a diagnostic approach. In: Presser S, Rothgeb JM, Couper MP, editors. Methods for Testing and Evaluating Survey Questionnaires. Wiley Interscience; 2004. 124.
- Wynd CA, Schmidt B, Schaefer MA. Two quantitative approaches for estimating content validity. West J Nurs Res. 2003;25(5):508-518. doi:10.1177/0193945903252998
- Polit DF, Beck CT. Measurement and data quality. In: Nursing Research: Generating and Assessing Evidence for Nursing Practice. 9th ed. Wolker Klumer / Lippincott Williams & Wilkins; 2012:334.
- Linden B, Stuart H. Preliminary analysis of validation evidence for two new scales assessing teachers’ confidence and worries related to delivering mental health content in the classroom. BMC Psychol. 2019;7(1):32. doi:10.1186/s40359-019-0307-y
- Rubio DM, Berg-Weger M, Tebb SS, Lee ES, Rauch S. Objectifying content validity: conducting a content validity study in social work research. Soc Work Res. 2003;27(2):94-104. doi:10.1093/swr/27.2.94
- Almanasreh E, Moles R, Chen TF. Evaluation of methods used for estimating content validity. Res Soc Adm Pharm. 2019;15(2):214-221. doi:10.1016/j.sapharm.2018.03.066
- DesRoches D. Establishment survey. In: Lavrakas PJ, editor. Encyclopedia of Survey Research Methods. Sage; 2008:124.
- Kelly J, Lavrakas PJ. Debriefing. In: Encyclopedia of Survey Research Methods. Vol I. Sage; 2008:181-182.
- Beatty P. Developing measures of health behavior and he alth service utilization. In: Johnson TP, editor. Handbook on Health Survey Methods. Willey; 2015:186-187.
- Polit DF, Beck CT, Owen SV. Is the CVI an acceptable indicator of content validity? Appraisal and recommendations. Res Nurs Health. 2007;30 (4):459-467. doi:10.1002/nur. 20199
- Yusoff MSB. ABC of content validation and content validity index calculation. Educ Med J. 2019;11(2):49-54. doi:10.21315/eimj2019.11.2.6
- Polit DF, Beck CT. Qualitative research design and approaches. In: Nursing Research: Generating and Assessing Evidence for Nursing Practice. 10th ed. Wolters Kluwer; 2017:489.
- Lawshe CH. A quantitative approach to content validity. Pers Psychol. 1975;28(4):563-575. doi:10.1111/j.1744-6570.1975.tb01393.x
- Ayre C, Scally AJ. Critical values for Lawshe’s Content Validity Ratio. Meas Eval Couns Dev. 2014;47(1):79-86. doi:10.1177/ 0748175613513808
- Stone KS, Frazier SK. Evaluation of measurement precision, accuracy, and error in biophysical data. In: Waltz CF, Strickland OL, Lenz ER, editors. Measurement in Nursing and Health Research. 4th ed. Springer Publishing; 2010:387-390.
- Cicchetti DV. On a model for assessing the security of infantile attachment: issues of observer reliability and validity. Behav Brain Sci. 1984;7 (1):149-150. doi:10.1017/S0140525X00026558
- Fleiss JL. Measuring nominal scale agreement among many raters. Psychol Bull. 1971;76(5):378-382. doi:10.1037/h0031619
- Kottner J, Audige L, Brorson S, et al. Guidelines for reporting reliability and agreement studies (GRRAS) were proposed. Int J Nurs Stud. 2011;48(6):661-671. doi:10.1016/j.ijnurstu.2011.01.016
- Osborne JW. Best Practices in Exploratory Factor Analysis. CreateSpace Independent Publishing; 2014.
- Lloret-Segura S, Ferreres-Traver A, Hernández-Baeza A, Tomás-Marco I. El análisis factorial exploratorio de los ítems: una guía práctica, revisada y actualizada. An Psicol. 2014;30(3). doi:10.6018/analesps.30.3.199361
- Field A. Why is my evil lecturer forcing me to learn statistics? In: Discovering Statistics Using IBM SPSS Statistics. 5th ed. Sage Publications; 2018:70-71.
- Kothari CR. Multivariate analysis techniques. In: Research Methodology: Methods and Techniques. 2nd ed. New Age Publisher; 2004:322.
- Haig BD. Exploratory factor analysis, theory generation, and scientific method. In: Method Matters in Psychology: Essays in Applied Philosophy of Science. Springer; 2018:65-88.
- Haig BD. Exploratory factor analysis, theory generation, and scientific method. Multivariate Behav Res. 2005;40(3):303-329. doi:10.1207/ s15327906mbr4003_2
- Råholm M-B. Abductive reasoning and the formation of scientific knowledge within nursing research. Nurs Philos. 2010;11(4):260-270. doi:10.1111/j.1466-769X.2010.00457.x
- Prudon P. Confirmatory factor analysis as a tool in research using questionnaires: a critique. Compr Psychol. 2015;4:03.CP.4.10. doi:10.2466/ 03.CP.4.10
- Eldridge J. Reliability, validity and trustworthiness. In: Boswell C, Cannon S, editors. Introduction to Nursing Research: Incorporating Evidence-Based Practice. 5th ed. Jones & Bartlettt Publishers; 2020:271-294.
- Abu-Bader SH. Working with SPSS. In: Using Statistical Methods in Social Science Research: With a Complete SPS Guide. 2nd ed. Oxford University Press; 2021:56.
- Perrin KM. Reliability and validity. In: Principles of Planning, Evaluation and Research for Health Care Programmes. 2nd ed. Jones &: Bartlett Learning; 2022:141.
- Grove SK. Quantitative measurement concepts. In: Gray JR, Grove SK, editors. Burns & Groves the Practice of Nursing Research: Appraisal, Synthesis, and Generation of Evidence. 9th ed. Elsevier; 2021:462.
- Hair J, Hollingsworth CL, Randolph AB, Chong AYL. An updated and expanded assessment of PLS-SEM in information systems research. Ind Manag Data Syst. 2017;117(3):442-458. doi:10.1108/IMDS-04-2016-0130
- Field A. Exploratory factor analysis. In: Discovering Statistics Using IBM SPSS Statistics (North American Edition). 5th ed. Sage; 2018:1390-1391.
- Clark LA, Watson D. Constructing validity: new developments in creating objective measuring instruments. Psychol Assess. 2019;31 (12):1412-1427. doi:10.1037/pas0000626
- Moreira J. Questionários: Teoria e Prática. Almedina; 2004.
- Streiner DL, Norman GR, Cainey J. Basic concepts. In: Health Measurement Scales: A Practical Guide to Their Development and Use. 5th ed. Oxford University Press; 2015:8.
- Bryant F. Assessing the validity of measurement. In: Grimm LG, Yarnold PR, editors. Reading and Understanding More Multivariate Statistics. American Psychological Association; 2002:99-146.
- Marôco J, Etapas da análise de equações estruturais. 3rd. Análise de Equações Estruturais, 2021:55
- Sommer I, Larsen K, Nielsen CM, Stenholt BV, Bjørk IT. Improving clinical nurses’ development of supervision skills through an action learning approach. Nurs Res Pract. 2020;2020:1-10. doi:10.1155/2020/9483549
- Bowling A. Quality of life: concepts, measurements and patient perception. In: Research Methods in Health: Investigating Health and Health Services. 4th ed. Open University Press; 2014:54.
- Bot SM, Terwee CB, van der Windt DAWM, Boute LM, Dekker J, Vet de HCW. Psychometric evaluation of self-report questionnaires: the development of a checklist. In: Adér HJ, Mellenberg C, editors. Proceedings of the Second Workshop on Research Methodology. Vu University; 2003:161-168.
- Powers BA, Knapp TR. Face validity. In: Dictionary of Nursing Theory and Research. 3rd ed. Spinger Publishing; 2006:63.
- Soeken WKL. Validity of measures. In: Measurement in Nursing and Health Research. 4th ed. Springer Publishing Company; 2010:163.
- Streiner DL, Norman GR, Cairney J. Seleting the itens. In: Health Measurement Scales: A Practical Guide to Their Development and Use. 5th. Oxford University Press; 2015:92.
- Zumbo BD. Validity as contextualized and pragmatic explanation, and its implications for validation practice. In: Lissitz RW, editor. The Concept of Validity: Revisions, New Directions, and Applications. Information Age Publishing; 2009:68.
- Chan EKH. Standards and guidelines for validation practices: development and evaluation of measurement instruments. In: Zumbo BD, Chan EKH, editors. Validity and Validation in Social, Behavioral, and Health Sciences. Springer Publishing; 2014:9-24.
- Mokkink LB, Terwee CB, Knol DL, et al. Taxonomy and definitions. COSMIN Checklist Manual. 2012;9.
